Paediatric Anaesthesia
KEY POINTS
- Anaesthetists should be aware of the indications and contraindications for propofol-based total intravenous anaesthesia (TIVA) in children.
- Pharmacological and nonpharmacological strategies can be used to establish intravenous (IV) access prior to induction.
- Children can still benefit from conversion to propofol-based TIVA after volatile anaesthetic induction.
- Adjuncts to propofol-based TIVA can greatly enhance immobility and haemodynamic response during surgery.
- Standard safe TIVA practices used in adults should be applied to children.
- Processed EEG monitoring is recommended in children > 1 year undergoing propofol-based TIVA.
INTRODUCTION
Propofol-based total intravenous anaesthesia (TIVA) has been widely used in adult practice for many years and its use in paediatrics is becoming more common. There are numerous clinical indications and benefits of TIVA; however, many anaesthetists are unfamiliar with how to provide propofol-based TIVA for their paediatric patients. While many aspects of TIVA can be translated from adults to children, there are some important considerations and limitations that anaesthetists should be aware of. This tutorial will discuss a practical approach to providing propofol-based TIVA for children. Anaesthetic practitioners who are less familiar with the general principles of TIVA and target-controlled infusions (TCI) are encouraged to read ATOTW 75, ‘‘Target controlled infusions in anaesthetic practice,’’1 and BJA Education, ‘‘Principles of total intravenous anaesthesia,’’2,3 prior to reading this tutorial.
WHEN TO CONSIDER PROPOFOL-BASED TIVA
Figure 1 outlines a simple approach to assist anaesthetists when considering propofol-based TIVA in a child. There are numerous reasons to choose TIVA, some applicable to both adults and children (eg, malignant hyperthermia, postoperative nausea and vomiting, PONV), and some that are more specific to children (eg, fear of face masks, emergence delirium). There
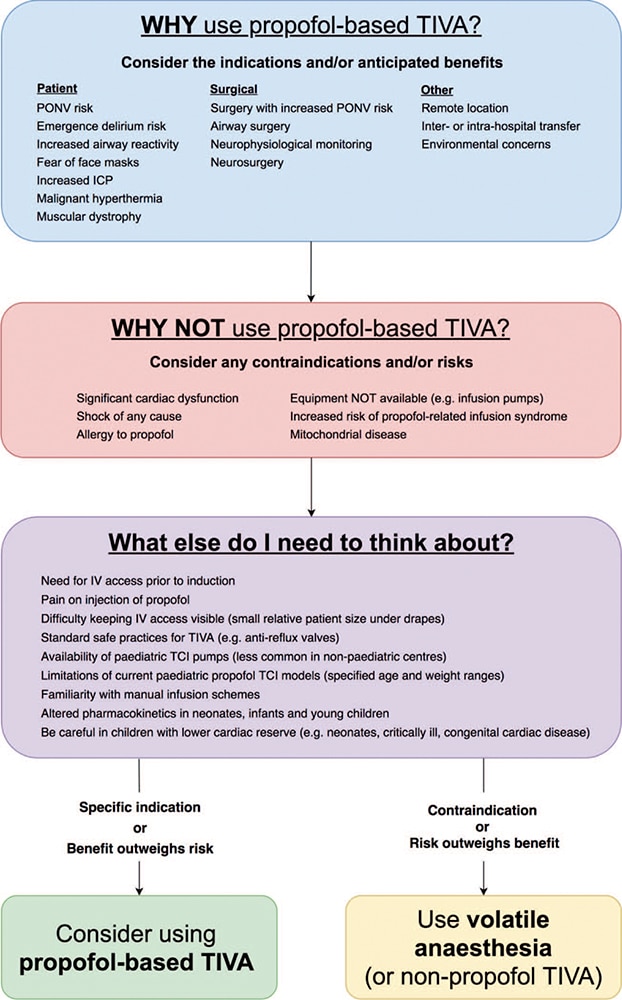
Figure 1. When to consider propofol-based total intravenous anaesthesia. TIVA indicates total intravenous anaesthesia; ICP, intracranial pressure; TCI, target-controlled infusions; and PONV, postoperative nausea and vomiting.
are several reasons more specific to children that deserve special mention. Children commonly present for surgery with concurrent viral upper respiratory tract infections that result in increased airway reactivity. In the child with a mild upper respiratory tract infection who needs to proceed with surgery, propofol induction is associated with a significant reduction in perioperative respiratory adverse events compared with sevoflurane induction.4 Also, children with some types of muscular dystrophies (eg, Duchenne and Becker) are at risk of anaesthesia-induced rhabdomyolysis. When faced with the undiagnosed ‘‘floppy child’’ presenting for urgent surgery, a reasonable approach is to provide a trigger-free anaesthetic using propofolbased TIVA.5
When considering reasons NOT to use propofol-based TIVA, there are routine concerns such as drug allergy and equipment availability (eg, infusion pumps), and there are also specific concerns relating to potential propofol-induced adverse effects in certain patient groups. Due to the significant myocardial depressant and vasodilatory effects of propofol, it should not be used in children with severe unresuscitated shock or significant cardiac dysfunction, for example severe sepsis and some congenital cardiac conditions. Propofol-based TIVA should also be avoided in patients at increased risk of propofol-related infusion syndrome, which is a life-threatening condition characterised by acute refractory bradycardia progressing to asystole and one or more of the following: metabolic acidosis, rhabdomyolysis, hyperlipidaemia, or enlarged or fatty liver. It is typically associated with prolonged infusions (> 48 hours) at high infusion rates (> 4 mg/kg/h), but cases have been reported with lower infusion rates and also high infusion rates of shorter duration.6,7 Risk factors include acute neurological injury, high endogenous or exogenous catecholamine and glucocorticoid levels, and low carbohydrate intake (eg, prolonged fasting). Propofol should also be avoided in patients with known or suspected mitochondrial disease (eg, mitochondrial myopathy) due to potentially increased risk of developing propofol-related infusion syndrome.
Aside from specific indications/contraindications, there are other factors that need to be considered (see Figure 1), many of which are more practical concerns, such as establishing intravenous access and determining how to administer propofol infusions in children. These will be discussed later.
One important factor that requires special mention is the altered pharmacokinetics of intravenous drugs in children. Young children have an increased central compartment size and volume of distribution, which requires larger induction bolus doses (eg, propofol 3 to 5 mg/kg in children versus 1.5 to 2 mg/kg in adults) and higher initial infusion rates. Once peripheral compartments are filled, lower infusion rates more similar to those in adults are needed to maintain anaesthesia. In neonates, immaturity of hepatic enzyme systems also results in slower metabolism, requiring lower maintenance infusion rates; however, there can also be marked pharmacokinetic variability between patients. These alterations in pharmacokinetic handling of drugs change as children get older, eventually becoming more similar to adults once they reach adolescence. Obesity is also becoming more prevalent in the paediatric population and this has pharmacokinetic implications. The suggested approach in obesity is to base dose calculations on lean body weight for induction and total body weight for maintenance of anaesthesia,8,9 although there is evidence that pharmacokinetic models utilising allometric scaling of total body weight more accurately characterise propofol volumes of distribution and clearances in obese patients.10
ESTABLISHING INTRAVENOUS ACCESS
Figure 2 provides an approach for establishing intravenous (IV) access in children. There are various nonpharmacological and pharmacological strategies that can be utilised to help establish IV access prior to induction. Any approach needs to be individualised for each child to ensure the greatest chance of success whilst minimising any distress for the child and parents. Distraction techniques and topical local anaesthetic are often effective; however, anxious and uncooperative or behaviourally challenging children may require premedication. ATOTW 367, ‘‘Paediatric anaesthesia: challenges with induction,’’ provides a useful approach to managing these patients.11
For some children, it may be more appropriate to perform a volatile anaesthetic induction and then establish IV access afterwards, allowing conversion to propofol-based TIVA for maintenance of anaesthesia. With the exception of circumstances where volatile agents are absolutely contraindicated, children can still derive much of the benefit of propofol-based TIVA with this approach. For example, when aiming to reduce emergence delirium, propofol-based TIVA for maintenance of anaesthesia is more effective than volatile anaesthesia alone and may also be more effective than volatile maintenance anaesthesia with the addition of 1 to 3 mg/kg of propofol prior to emergence.12
Transitioning from volatile anaesthetic induction to propofol-based TIVA maintenance needs to be done carefully in order to maintain adequate depth of anaesthesia during the transition while also avoiding the negative haemodynamic effects associated with overanaesthetising patients. The aim is to spread the initial propofol bolus dose over the expected washout of the volatile agent. When using a propofol TCI pump, this can be done by adjusting the initial TCI target and/or the duration of time that the initial bolus is delivered.
There are a number of strategies that can be used to minimise injection pain associated with propofol. Intravenous lidocaine (lignocaine) and cannulas in large proximal veins are common methods for reducing injection pain. Preinduction nitrous oxide and/or preinjection of a fast-onset opioid, such as fentanyl, can also be effective.
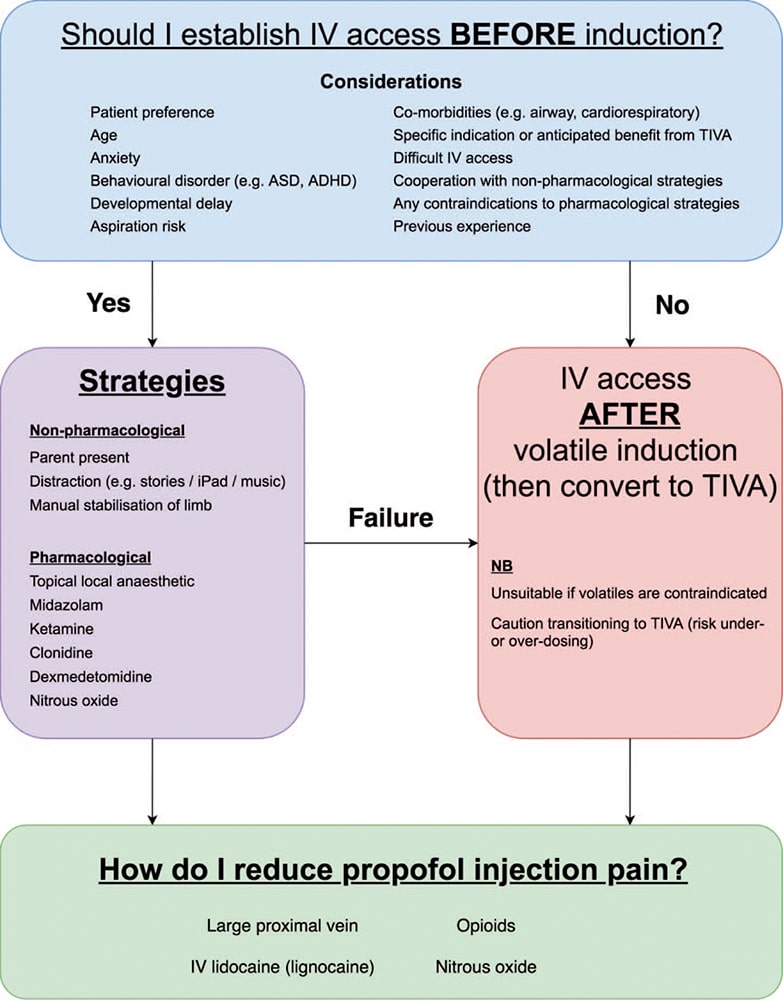
Figure 2. Establishing intravenous access. ASD indicates autism spectrum disorders; ADHD, attention deficit hyperactivity disorder; and TIVA, total intravenous anaesthesia.
ADMINISTERING PROPOFOL-BASED TIVA
Figure 3 outlines how propofol-based TIVA can be administered by either TCI or manual infusion. The pharmacokinetic threecompartment model for propofol is well described in pharmacology textbooks and the key pharmacokinetic alterations in children have been discussed above. The clinical implications in children are larger induction boluses and higher initial infusion rates relative to weight compared to adults, followed by lower maintenance infusion rates. Due to greater accumulation in peripheral compartments during prolonged infusions, failure to appropriately reduce maintenance infusion rates can lead to significantly delayed emergence from anaesthesia.13
Propofol-based TIVA is ideally administered using a TCI pump with an appropriate paediatric model. The following two paediatric TCI models are commonly used:
- Paedfusor: suitable for ages 1 to 16 years and weight 5 to 61 kg
- Kataria: suitable for ages 3 to 16 years and weight 15 to 61 kg.
It should be noted that paediatric TCI models work on plasma site concentration prediction (Cp) rather than effect site concentration prediction (Ce), so practitioners need to be mindful of accounting for time to allow Cp-Ce equilibration when increasing or decreasing target levels. Typical propofol target steady-state Cp values include the following:
- Propofol (sole agent) →Cp = 4 to 6 mcg/mL
- Propofol + adjuncts* (eg, remifentanil infusion or regional anaesthesia) → Cp = 3 to 4 mcg/mL. *This excludesneuromuscular-blocking drugs (NMBDs).
Adult TCI models can potentially be applied to some limited paediatric use. For example, teenagers weighing >61 kg can potentially be managed using the Marsh adult model (NB: there is no age covariate even though the TCI pump requires age>16 years to be programmed). Recently Eleveld et al developed a pharmacokinetic-pharmacodynamic model for propofol TCI that predicted propofol concentrations and bispectral index (BIS) for a broad population ranging from neonates to elderly patients.14 Although there were limitations with the pharmacodynamic model in supporting young children and adolescents, it is encouraging that there may eventually be a single propofol TCI model that can be applied to both paediatric and adult patients.
Manual infusion schemes are used in countries where TCI does not have regulatory approval. They can also be used as an alternative to TCI when appropriate paediatric TCI pumps are not available, or for patients outside the age/weight range of available models (eg, children < 1 year). McFarlan proposed an infusion scheme for children 3 to 11 years (see Table 1).15 An induction bolus dose (2.5 mg/kg) is followed by a maintenance infusion with subsequent adjustments in the infusion rate at prescribed time intervals, with the aim of targeting a steady-state Cp = 3mcg/mL. In clinical practice, aiming for a steady-state Cp = 3mcg/mL generally requires the addition of an adjunct to achieve adequate depth of anaesthesia and optimal surgical conditions. Steur et al proposed a manual infusion scheme for children < 3 years (see Table 2).16 After the induction bolus, infusion rates vary between the age subgroups accounting for altered pharmacokinetics for different ages. Note that Steur’s scheme suggests very high infusion rates in children < 6 months (> 20 mg/kg/h) and should be used with caution in this age group. For signs of reduced depth of anaesthesia, propofol boluses of 1 mg/kg ± adjuncts such as opioids can be given.
Manual infusion schemes only provide suggested doses and require titration to patient response with the aim of achieving appropriate clinical anaesthesia. They also need to be adjusted to account for various factors such as preoperative anxiety, use of adjuncts, required depth of anaesthesia, interindividual variability, and patient comorbidities. Target-controlled infusion models also vary in their accuracy due to the same factors mentioned and can even vary in their performance at different stages of infusion (bolus, maintenance, recovery).17,18 Using processed electroencephalography (pEEG) monitoring, such as BIS, to guide management is the preferred approach for dealing with any inaccuracies in TCI models or manual infusions, and other factors such as interindividual variability.
ADJUNCTS TO PROPOFOL-BASED TIVA
Propofol as a sole anaesthetic agent is extremely poor at maintaining patient immobility. Adjuncts can be used to significantly improve surgical conditions and haemodynamic response to surgical stimulus. Many adjuncts also have propofol-sparing effects, resulting in lower propofol target requirements. Some typical adjuncts used in propofol-based TIVA are discussed below.
Remifentanil
Remifentanil is an ultra–fast-acting opioid that is easily titratable due to its rapid effect-site equilibration. Its advantages include fast onset/offset and short context-sensitive half-time. Its disadvantages include respiratory depression, potential hyperalgesia, and thoracic wall rigidity. Remifentanil is effective in reducing propofol requirements by up to 40%. The Minto model is a commonly used remifentanil TCI algorithm suitable for patients age ≥ 12 years and weight ≥ 30 kilograms.19 Below this age/ weight, manual infusions are used with rates ranging from 0.1 to 0.5 mcg/kg/min. Infusion rates of 0.1 to 0.3 mcg/kg/min generally provide adequate intraoperative analgesia. Higher infusion rates (eg, 0.4 to 0.5 mcg/kg/min) or small boluses (eg, 0.5

Figure 3. Administering propofol-based total intravenous anaesthesia. TIVA indicates total intravenous anaesthesia; TCI, target-controlled infusions.

Table 1. McFarlan propofol infusion scheme for children 3-11 years (doses in mg/kg/hr)^15
to 1mcg/kg) can be given during particularly stimulating periods (eg, laryngoscopy), however this should be used cautiously due to potential bradycardia.
Ketamine
Ketamine is a N-methyl-D-aspartate (NDMA)-receptor antagonist with sedative and analgesic properties. Its uses include premedication, sedation, and perioperative analgesia. Advantages include less respiratory depression, bronchodilation, and excellent analgesia. Ketamine also better maintains sympathetic tone resulting in improved haemodynamic stability. This is extremely useful in patients who are critically ill or have cardiac comorbidities. Ketamine can also be used as a sole anaesthetic agent for TIVA in limited-resource environments (eg, prehospital and developing countries) or when propofol-based TIVA is unsuitable (see Figure 1).20,21 Disadvantages include increased airway secretions, nausea, hallucination, and interference with pEEG monitoring. Manual infusion rates range from 0.1 to 2.5 mg/kg/h depending on factors such as procedure, patient status, and other agents used.22,23 To improve wake-up, infusion rates are typically reduced towards the end of the procedure and ceased at least 30 minutes prior to emergence.20
Dexmedetomidine
Dexmedetomidine is an ∝–2 agonist with sedative, anxiolytic, and analgesic effects. Its uses include premedication and postoperative sedation. Advantages include less respiratory depression and an opioid-sparing effect. Dexmedetomidine can cause an initial hypertension and then subsequent hypotension and bradycardia, which is exacerbated by higher infusion rates. A loading dose of 0.5 to 1mcg/kg over 10 to 20 minutes can be given, followed typically by an intraoperative infusion rate of 0.2 to 0.5 mcg/kg/h. Due to the long context-sensitive half-time of dexmedetomidine (up to 250 minutes after an 8-hour infusion), reduction in the infusion rate of dexmedetomidine and/or other agents used may be required to avoid prolonged time to emergence and extubation.24,25
Other Adjuncts
Opioids such as fentanyl and alfentanil can be administered as either continuous infusions or titrated intermittent boluses. They also reduce propofol requirements and have an advantage over remifentanil of smoother transition to postoperative opioidbased analgesia. Their disadvantage is longer context-sensitive half-times compared with remifentanil, which could cause delay in emergence after prolonged infusions. Neuromuscular-blocking drugs are effective in improving surgical conditions, but do not reduce propofol requirements and need to be used cautiously during TIVA due to risk of awareness under general anaesthesia. It should also be noted that propofol does not have the same potentiating effect on NMBDs that volatile anaesthetics have, consequently larger and/or more frequent doses of NMBDs may be required. Midazolam and clonidine are anxiolytic and sedative agents that can be used as premedication prior to TIVA, as well as in the management of emergence delirium. Regional anaesthesia can also be used in combination with propofol-based TIVA. Peripheral nerve blocks and neuraxial techniques, such as epidurals and caudal blocks, are extremely beneficial in reducing opioid and anaesthetic dose requirements.
AVOIDING DRUG DELIVERY PROBLEMS
The issues related to ensuring appropriate drug delivery during TIVA are the same in adults and children. Standard safepractices for administering TIVA should always be used (see Figure 4). Infusion pumps need to be correctly programmed and
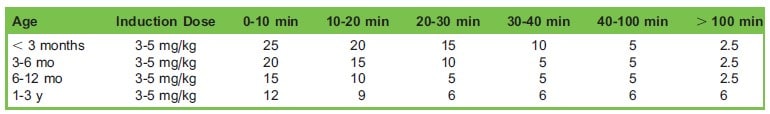
Table 2. Steur propofol infusion scheme for children ≤ 3 years (doses in mg/kg/h)^16
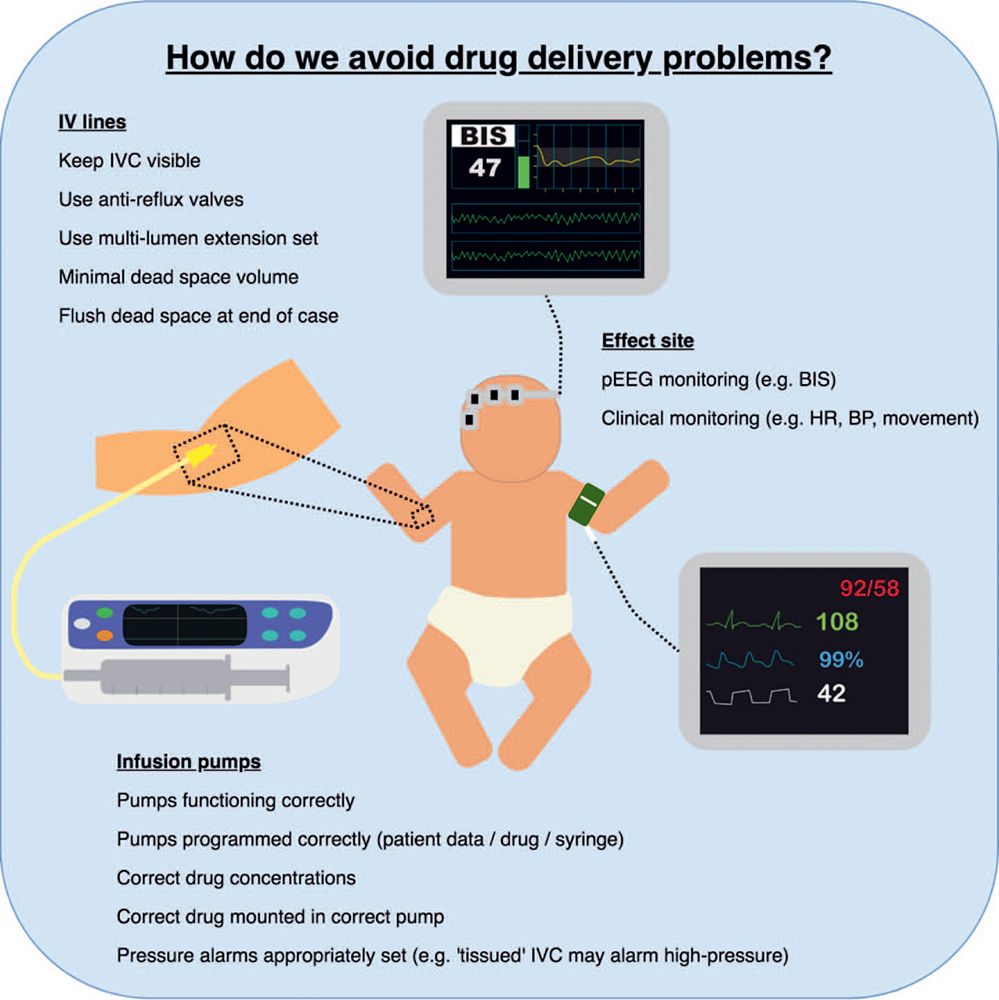
Figure 4. Avoiding drug delivery problems. IVC indicates intravenous catheter; pEEG, processed electroencephalography; and BIS, bispectral index.
drug concentrations need to be appropriate for weight-based dosing. Many manual infusion pumps are able to perform weightbased dose-rate calculations; however, the actual infusion rate should always be checked to confirm that an adequate rate is being delivered. Very low infusion rates (eg, < 0.5 mL/h) may not provide reliable and consistent drug delivery, and drugs can be prepared in lower dose concentrations to avoid this problem. Antireflux valves should be used to prevent backflow of drugs in the IV line. Intravenous access and drug lines should always be kept visible so that any problems, such as disconnections and IV cannula failure, can be detected early and rectified. In infants and younger children this can be more challenging due to their smaller relative size underneath sterile drapes. At the end of all cases, drug lines must be flushed to prevent accidental administration of any residual anaesthetic drugs in the line dead space.26
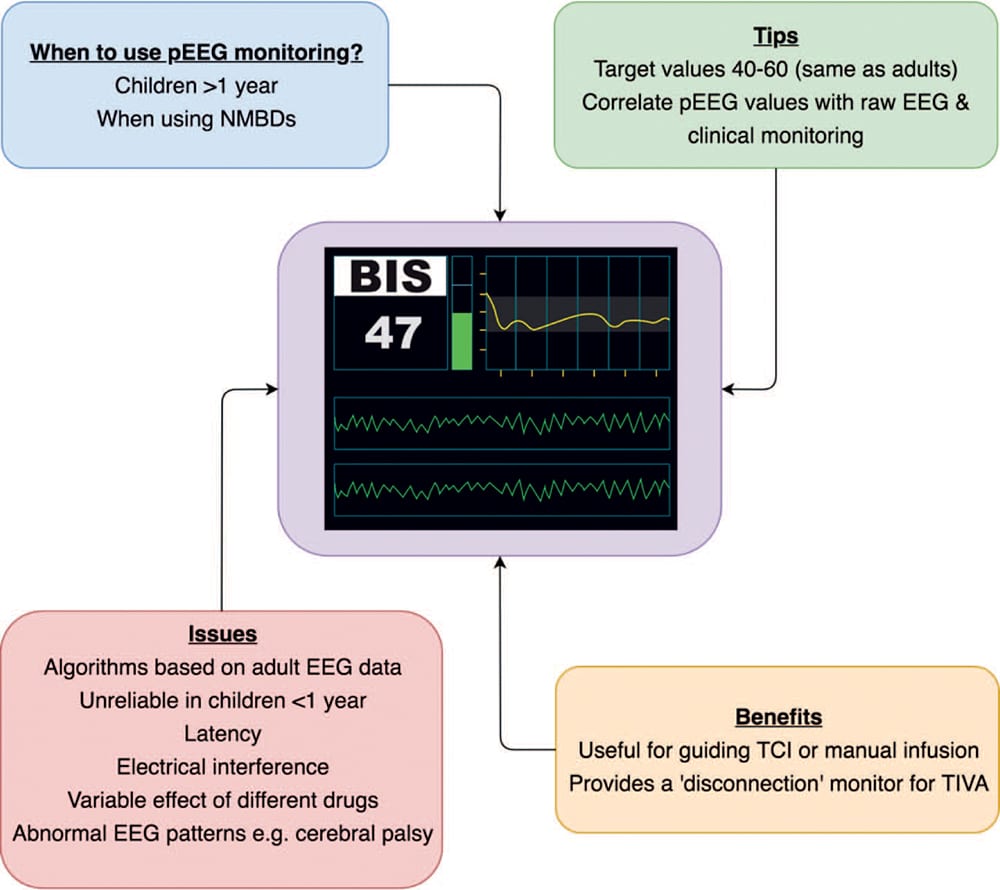
Figure 5. Using processed electroencephalography monitoring in children during propofol-based total intravenous anaesthesia. pEEG indicates processed electroencephalography; NMBDs, neuromuscular-blocking drugs; and TCI, target controlled infusions.
Processed electroencephalography provides a valuable safety mechanism by acting as a ‘‘disconnection’’ monitor during TIVA (see Figure 5). Propriety pEEG algorithms, such as BIS and Entropy, were developed using adult electroencephalography (EEG) data and have been extrapolated for use in children. As such, pEEG in paediatric anaesthesia is still being researched. For children > 1 year, BIS does provide values consistent with anaesthetic concentration relationships as seen in adults.27 However, for infants and neonates (ie, children < 1 year), BIS values have not been found to be consistent, possibly due to the immaturity of the developing brain and its effect on the EEG.28 Currently pEEG monitoring is recommended in children >1 year undergoing TIVA, particularly if NMBDs are administered. Target values are similar to adults (40 to 60), and if values are maintained below 50 then awareness is very unlikely to occur.29
There are known limitations of pEEG such as latency, electrical interference, and variable drug effects.30 Abnormal EEG patterns are another limitation of pEEG. These include normal genetic variants,31 as well as pathological changes such as seizures, cerebral hypoperfusion, and hypothermia. Neurological disorders can also have an effect on pEEG; for example, lower BIS values are observed in children with cerebral palsy.32 Due to these limitations, pEEG values should not be solely relied upon during propofol-based TIVA and should be regularly correlated with the raw EEG data and clinical monitoring.
SUMMARY
There are numerous reasons for using propofol-based TIVA in children, such as concurrent upper respiratory tract infection and reducing emergence delirium. It is important to also consider any relevant reasons for not using propofol, such as cardiovascular compromise and risk of propofol-related infusion syndrome. After evaluating the factors relevant to any specific case, when the anticipated benefits outweigh the risks, anaesthetists should strongly consider using propofol-based TIVA. It is possible to establish IV access in children prior to induction using a variety of pharmacological and nonpharmacological strategies. However, if a volatile anaesthetic induction is performed, many patients can still benefit from conversion to propofol-based TIVA after induction, particularly children at increased risk of emergence delirium.
Propofol-based TIVA can be administered using either paediatric TCI algorithms or manual infusion schemes. The use of adjuncts can greatly enhance immobility and haemodynamic response to surgical stimulus during propofolbased TIVA. It is crucial to implement standard safe TIVA practices for ensuring appropriate drug delivery to patients, including correct drug concentrations, working infusion pumps, and reliable IV access. Processed electroencephalography monitors, such as BIS, provide a valuable ‘‘disconnection’’ monitor during propofol-based TIVA and should be used in children >1 year.
The general principles of propofol-based TIVA in adults can be applied to paediatric practice. Anaesthetists and trainees should be encouraged to gain experience in providing propofol-based TIVA to children.
REFERENCES
- Sivasubramaniam S. ATOTW 75. Target controlled infusions in anaesthetic practice. WFSA ATOTW. 2007; https://www. resources.wfsahq.org/resources/anaesthesia-tutorial-of-the-week. Accessed October 5, 2018.
- Al-Rifai Z, Mulvey D. Principles of total intravenous anaesthesia: basic pharmacokinetics and model descriptions. BJA Educ. 2016;16(3):92-97.
- Al-Rifai Z, Mulvey D. Principles of total intravenous anaesthesia: practical aspects of using total intravenous anaesthesia. BJA Educ. 2016;16(8):276-280.
- Regli A, Becke K, von Ungern-Sternberg BS. An update on the perioperative management of children with upper respiratory tract infections. Curr Op Anaesth. 2017;30(3):362-367.
- Hayes J, Veyckemans F, Bissonnette B. Duchenne muscular dystrophy: an old anesthesia problem revisited. Pediatr Anesth. 2007;18(2):100-106.
- Felleiter P. Propofol infusion syndrome—a fatal case at a low infusion rate. Anesth Analg. 2006;103(4):1050.
- Liolios A, Gue´ rit JM, Scholtes JL, Raftopoulos C, Hantson P. Propofol infusion syndrome associated with short-term largedose infusion during surgical anesthesia in an adult. Anesth Analg. 2005;100(6):1804-1806.
- Mortensen A, Lenz K, Abildstrøm H, Lauritsen TLB. Anesthetizing the obese child. Pediatr Anesth. 2011;21(6):623-629.
- Diepstraten J, Chidambaran V, Sadhasivam S, Esslinger HR, Cox SL, Inge TH, Knibbe CA, Vinks AA. Propofol clearance in morbidly obese children and adolescents: influence of age and body size. Clin Pharmacokinet. 2012;51(8):543-551.
- Cortinez LI, Anderson BJ, Penna A, Olivares L, Munoz HR, Holford NH, Struys MMRF, Sepulveda Pl. Influence of obesity on propofol pharmacokinetics: derivation of a pharmacokinetic model. Br J Anaesth. 2010;105(4):448-456.
- Kelly L, Cooper M. ATOTW 367. Paediatric anaesthesia: Challenges with induction. WFSA ATOTW. 2017; https://resources.wfsahq.org/resources/anaesthesia-tutorial-of-the-week. Accessed May 29, 2018.
- Mason KP. Paediatric emergence delirium: a comprehensive review and interpretation of the literature. Br J Anaesth. 2017;118(3):335-343.
- Short TG, Aun CS, Tan P, Wong J, Tam YH, Oh TE. A prospective evaluation of pharmacokinetic model controlled infusion of propofol in paediatric patients. Br J Anaesth. 1994;72(3):302-306.
- Eleveld DJ, Colin P, Absalom AR, Struys MMRF. Pharmacokinetic-pharmacodynamic model for propofol for broad application in anaesthesia and sedation. Br J Anaesth. 2018;120(5):942-959.
- McFarlan CS, Anderson BJ, Short TG. The use of propofol infusions in paediatric anaesthesia: a practical guide. Pediatr Anesth. 1999;9(3):209-216.
- Steur RJ, Perez RS, DeLange JJ. Dosage scheme for propofol in children under 3 years of age. Pediatr Anesth. 2004;14(6):462-467.
- Rigouzzo A, Servin F, Constant I. Pharmacokinetic-pharmacodynamic modelling of propofol in children. Anesthesiology. 2010;113(2):343-352.
- Sepulveda P, Cortinez LI, Saez C, Penna A, Solari S, Guerra I, Absalom AR. Performance evaluation of paediatric propofol pharmacokinetic models in healthy young children. Br J Anaesth. 2011;107(4):593-600.
- Minto CF, Schnider TW, Egan TD, Youngs E, Lemmens HJM, Gambus PL, Billard V, Hoke JF, Moore KHP, Hermann DJ, Muir KT, Mandema JW, Shafer SL. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil: I—model development. Anesthesiology. 1997;86(1):10-23.
- Stevenson C. Ketamine: a review. WFSA Update Anaesth. 2005; https://resources.wfsahq.org/resources/update-in-anaesthesia. Accessed May 11, 2018.
- Gales A, Maxwell S. ATOTW 381. Ketamine: recent evidence and current uses. WFSA ATOTW. 2018; https://resources.wfsahq.org/resources/anaesthesia-tutorial-of-the-week. Accessed July 1, 2018.
- Shann F. Drug Doses. 17th ed. Melbourne, Australia: University of Melbourne; 2017.
- Weatherall A, Venclovas R. Experience with a propofol-ketamine mixture for sedation during pediatric orthopedic surgery. Pediatr Anesth. 2010;20(11):1009-1016.
- Mahmoud M, Mason KP. Dexmedetomidine: review, update, and future considerations of paediatric perioperative and periprocedural applications and limitations. Br J Anaesth. 2015;115(2):171-182.
- Kim DJ, Kim SH, So KY, Jung KT. Effects of dexmedetomidine on smooth emergence from anaesthesia in elderly patients undergoing orthopaedic surgery. BMC Anesthesiol. 2015;15:139.
- Bowman S, Raghavan K, Walker IA. Residual anaesthesia drugs in intravenous lines—a silent threat? Anaesthesia. 2013;68(6):557-561.
- Whyte SD, Booker PD. Bispectral index during isoflurane anaesthesia in paediatric patients. Anesth Analg. 2004;98(6):1644-1649.
- Davidson AJ, Huang GH, Rebmann CS, Ellery C. Performance of entropy and bispectral index as measures of anaesthesia effect in children of different ages. Pediatr Anesth. 2005;95(5):674-679.
- Davidson AJ, Kwok T. Performance of BIS in children using the paediatric BIS Quattro sensor. Anaesth Intensive Care. 2008;36(6):807-813.
- Duarte LT, Saraiva RA. When the bispectral index can give false results. Rev Bras Anestesiol. 2009;59(1):99-109.
- Schnider TW, Luginbuehl M, Petersen-Felix S, Mathis J. Unreasonably low bispectral index values in a volunteer with genetically determined low-voltage electroencephalographic signal. Anesthesiology. 1998;89(6):1607-1608.
- Choudhry DK, Brenn BR. Bispectral index monitoring: a comparison between normal children and children with quadriplegic cerebral palsy. Anesth Analg. 2002;95(6):1582-1585.



