Obstetrics Anaesthesia
KEY POINTS
- Accidental awareness during general anaesthesia is more common in obstetric practice than in any other discipline of anaesthesia.
- Factors contributing to an increased risk of accidental awareness during general anaesthesia include maternal stress or anxiety, emergency surgery and the “intravenous-inhalational” interval.
- Accidental awareness during general anaesthesia can be associated with considerable physiological and psychological harm.
- Strategies to minimise the risk of accidental awareness during general anaesthesia include an appropriate dose of anaesthetic induction agent, and use of nitrous oxide to achieve an adequate depth of anaesthesia
INTRODUCTION
Accidental awareness during general anaesthesia (AAGA) is defined as the unintended experience and subsequent recall of intra-operative events.
In recent decades, the incidence of AAGA has been reported as between 1–2:1,000 general anaesthetics (1). The landmark NAP5 report (Fifth National Audit Project) – a national survey conducted by The Royal College of Anaesthetists (RCOA) which investigated spontaneous reports of AAGA – demonstrated an incidence of 1:19,000 in the general population. NAP5 further highlighted the incidence of AAGA in the obstetric population is significantly higher at 1:670 for caesarean section (CS) under general anaesthesia (GA) (2).
This tutorial summarises some of the possible reasons for this and aims to provide ways to reduce the risk of AAGA in obstetric patients.
AAGA IN OBSTETRIC ANAESTHESIA
Obstetric anaesthesia is high-risk for AAGA due to the anatomical and physiological changes of pregnancy, as well as the technique of administering GA for an emergency procedure. Risk factors are summarised in Table 1 and are explored further in this tutorial.
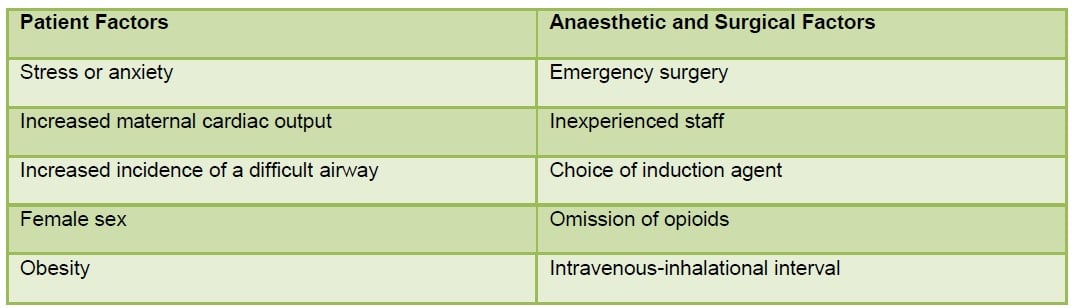
Table 1. Risk factors for awareness in obstetric anaesthesia
AAGA is a major patient safety concern with significant implications for the patient and anaesthetist. For the patient, these include physiological effects such as an exaggerated stress response to laryngoscopy and surgery, as well as psychological sequelae ranging from poor sleep and nightmares to post-traumatic stress disorder (PTSD) (1). The incidence of psychological sequelae varies with studies reporting up to 71% of patients who experience AAGA suffer PTSD or late psychological symptoms (2, 3).
For the anaesthetist, being involved in a case of AAGA can also be a distressing event so it is important that support is available. Moreover, AAGA can be a source of litigation; one study of litigation in obstetric anaesthesia in England found over 80% of lawsuits were because of AAGA (4).
Emergency surgery and staff experience level
GA for obstetrics is often for a time-critical procedure (e.g. emergency CS performed either for maternal or fetal indications). Induction of anaesthesia is traditionally performed using a “classical” rapid sequence induction (RSI) technique consisting of pre-oxygenation, administration of a predetermined dose of thiopental and suxamethonium, application of cricoid pressure followed by tracheal intubation with a definitive airway.
A pregnant woman undergoing emergency CS under GA is likely to experience anxiety and stress and this can contribute to the risk of awareness (2). In addition, stress on the part of the anaesthetist (due to concern about the increased incidence of failed intubations and difficult airway management in obstetric practice) with the attendant effects on clinical performance in an emergency situation may also contribute to the risk of AAGA.
Currently in developed countries, the number of GAs being administered in maternity units continues to decline for a variety of reasons, with the main factor being the increased use of neuraxial techniques for labour analgesia and surgical anaesthesia. This has resulted in fewer training opportunities in administering GAs in obstetric patients. NAP5 identified this as a risk factor for AAGA, and recommended all obstetric anaesthetists maintain their airway skills by regularly practising drills to include critical incidents such as failed intubation (2, 5). This is especially pertinent in the United Kingdom (UK) where trainee anaesthetists manage obstetric patients on-call, without direct supervision.
Choice of induction agents and the effects of maternal cardiac output
Despite widespread use of propofol in anaesthetic practice, thiopental remains an appropriate alternative induction agent in obstetric anaesthesia as practiced in the UK. This was highlighted in a 2013 survey that stated 93% of UK Consultants use thiopental for obstetric GA (6). The reasons cited for doing so were “historical”, as well as concerns about awareness and neonatal effects (propofol has a relatively slower onset time, a shorter distribution half-life and a greater potential for hypotension which may adversely affect placental perfusion) (6).
One of the major advantages of thiopental over propofol is its rapid onset of action (in one arm-brain circulation time; approximately 30 seconds) which has been thought to reduce the likelihood of awareness (propofol in contrast has a comparatively slower onset time because of its larger volume of distribution). However, the increased cardiac output in late pregnancy reduces the duration of action of an intravenous bolus of induction agent and the same mechanism also prolongs the time taken to achieve an effective partial pressure of volatile agent.
Appropriate dosing of induction agents is critical as inappropriately low dosing has been identified as contributing to increased risk of AAGA as demonstrated in examples from the NAP5 report (2). NAP5 recommended that where thiopental is used, a minimum dose of 5 mg kg-1 be administered.
Whilst the properties of thiopental and its proven safety record are major strengths, the potential for drug administration errors (most notably syringe swaps when concurrently drawn up with prophylactic antibiotics which have a similar solution colour – highlighted as the cause for multiple cases of AAGA in NAP5) as well as supply and cost issues (US$8.72 for a 500 mg vial of thiopental as opposed to US$4.65 for a 200 mg vial of propofol) are important considerations (2, 7, 8).
There is a paucity of clinical trials comparing the efficacy of propofol with thiopentone in obstetric practice. More recently, there has been a trend towards propofol as the preferred induction agent in obstetric practice in the UK (8, 9) driven in part by concerns about awareness and also diminishing experience with thiopentone as an induction agent (both of these factors were highlighted as being significant by NAP5 and subsequent editorials that followed its publication [2, 7]). The trend towards using propofol is in keeping with practice in North America (where thiopental is not available) and therefore considerable experience has been developed using propofol for RSI in obstetric practice.
Alternative induction agents such as ketamine (at a dose of 0.5 – 1.5 mg kg-1) remain important options for the anaesthetist. The inherent cardiac stability of ketamine (due to it sympathomimetic effect) affords greater safety margins in unstable patients. The same property makes it well suited to general anaesthesia in resource-poor environments (unusually for an induction agent, ketamine has little effect on central respiratory drive and airway reflexes generally remain well preserved although co-administration of anti-muscarinic drugs is recommended to manage hypersalivation).The onset of action of ketamine however is comparatively slower (90-120 seconds) and there is no definitive endpoint as seen with propofol (loss of verbal responsiveness) or thiopentone (eye closure with loss of eyelash reflex) at induction of anaesthesia.
Difficult airways
The incidence of failed intubation in obstetrics (1:224) is almost 10-fold higher than in the non-obstetric population (1:2230) (10). Contributing factors include increased body habitus, inadequate patient positioning, enlarged breasts which may hinder laryngoscopy, and the presence of laryngeal oedema. When a difficult airway (or failed intubation) is encountered there can be a delay in initiating administration of a volatile agent for maintenance and this can lead to an increased risk of AAGA. It is recommended that a second syringe of induction agent be readily available to be administered as boluses if necessary (2).
The “intravenous-inhalational interval”
The intravenous-inhalational interval is a phenomenon that describes the interval between the administration of an intravenous anaesthetic induction agent (and subsequent redistribution) and an adequate depth of anaesthesia (DOA) using an inhalational (volatile) anaesthetic agent. The unique nature of obstetric anaesthesia is such that the patient is prepped and draped prior to induction of anaesthesia in order to minimise the duration of fetal exposure to the effects of general anaesthesia prior to delivery by CS. The short time interval between induction of anaesthesia and surgical stimulus increases the risk of not achieving an adequate DOA if the “intravenous-inhalational interval” has a long duration (Figure 1) (2).
This is recognised as a key contributor to the development of AAGA in obstetric practice where surgical stimulus usually commences immediately after confirmation of correct placement of the endotracheal tube (2).
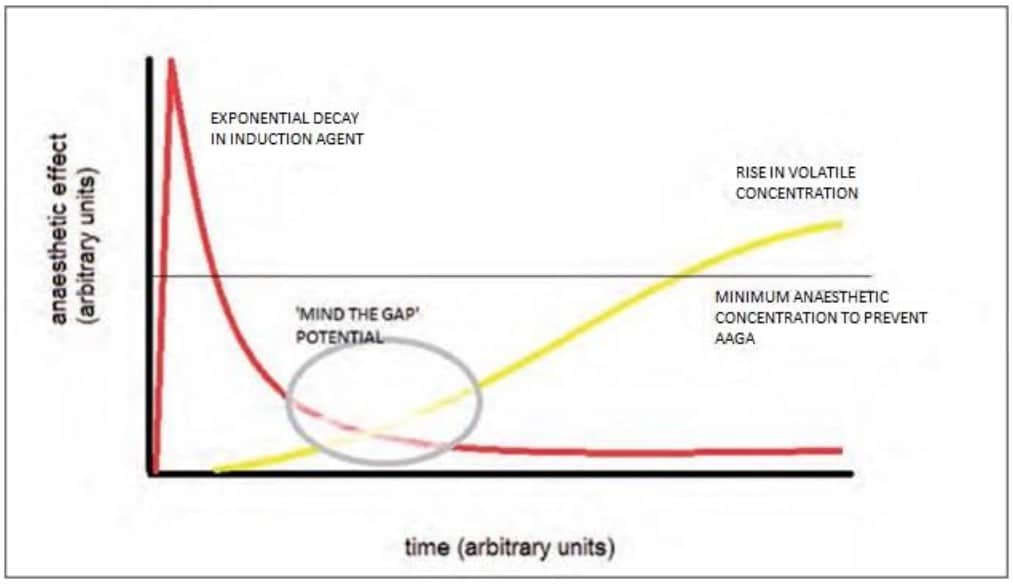
Figure 1. The intravenous-inhalational interval (2). Diagrammatic representation of a ‘gap’ in delivery of anaesthetic when the volatile agent (yellow) is turned on too late, at a low rate or is interrupted, as the effect of the initial intravenous bolus (red) is in decline. The thin black line represents the minimum agent concentration required to prevent AAGA. Reproduced with permission from The National Institute of Academic Anaesthesia.
It is recommended that after induction of anaesthesia and intubation of the trachea, volatile anaesthetics be administered in addition to nitrous oxide (nitrous oxide: oxygen [50:50)] prior to delivery) to target a MAC (minimum alveolar concentration) of between 0.7 – 0.8. This has been shown to consistently achieve mean Bispectral Index (BIS) scores of <60 (suggesting adequate depth of general anaesthesia). Because of its rapid uptake, nitrous oxide remains an important adjunct to reduce the risk of awareness (2, 5, 7).
The pharmacokinetic properties of inhalational agents such as sevoflurane, desflurane and nitrous oxide make them ideal for use in obstetric anesthesia primarily because of their low blood:gas partition coefficients (sevoflurane 0.68, desflurane 0.4, nitrous oxide 0.47, isoflurane 1.4) rendering them less soluble in blood, therefore permitting a rapid increase in the alveolar partial pressure as less is extracted by the pulmonary circulation. By way of comparison, halothane has a blood:gas partition coefficient of 2.4 rendering the agent much more soluble in blood compared to newer agents; the onset of anaesthesia is therefore slower.
Volatile agents are not without risks as halogenated agents have tocolytic effects which can be associated with increased blood loss from uterine atony (at MAC >1). Higher concentrations can cause maternal hypotension that can lead to reduced placental perfusion pressure that can contribute to fetal acidosis. Despite the theoretical reduction in MAC in pregnancy (up to 30%) as a result of the influence of progesterone and endogenous endorphins, the use of volatile-sparing agents such as nitrous oxide as well as opioids and benzodiazepines post-delivery is encouraged to limit the atonic effects of volatile agents (5).
MINIMISING THE RISK AND COMPLICATIONS OF AAGA
Consent
It has been suggested that consent for AAGA in obstetric practice should be considered for all patients undergoing GA (2, 5, 7). This may not always be practical given the emergency nature of anaesthesia. It is important nonetheless to emphasise to the patient what sensations they may be aware of, i.e. cricoid pressure and potentially some other sensations during induction of anaesthesia (5). This approach has been shown to reduce the likelihood and severity of psychological sequelae following AAGA as highlighted by NAP5 where patient understanding of “what was happening or what had happened” appeared to mitigate against immediate and long-term psychological distress (2).
Nitrous oxide
Nitrous oxide is an inhalational agent with a long and established role in obstetric anaesthesia and analgesia due to its volatile-sparing properties.
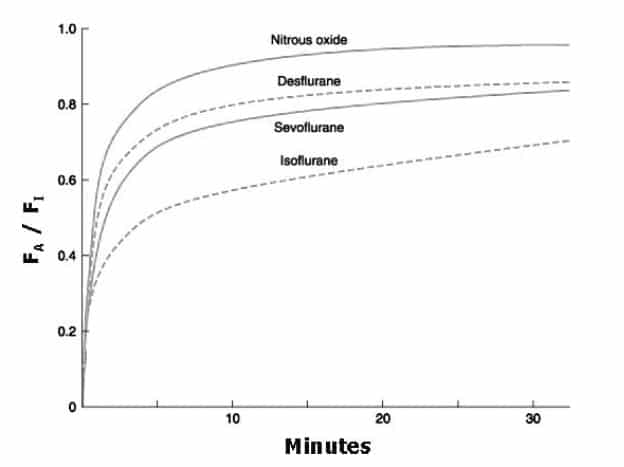
Figure 2. Alveolar concentration of volatile agent (FA)/Fraction inspired of volatile agent (FI) over duration of administration. Reproduced with permission from Allen Press Publishing Services (11).
Figure 2 demonstrates that due to the low blood:gas solubility coefficient of nitrous oxide, it achieves steady state faster than isoflurane, sevoflurane or desflurane (that is FA [alveolar concentration] rises towards FI [Fraction inspired] much faster). Halothane remains in use in some centres around the world. Due to its high solubility in blood (blood:gas coefficient 2.4 versus 0.47 for nitrous oxide and 1.4 for isoflurane) the alveolar concentration (FA) rises towards the fraction inspired (FI) much slower accounting for the slower onset of anaesthesia.
After preoxygenation with 100% oxygen, induction of anaesthesia and confirmation of correct placement of the endotracheal tube, the “concentration” effect can be employed to rapidly achieve an adequate DOA and reduce the likelihood of AAGA due to the intravenous-inhalational interval. An “overpressure” technique can also be used to reduce the intravenous-inhalational interval, which involves setting the vaporiser to deliver a high fractional concentration of inspired inhalational agent (FI) with a high flow rate to more rapidly achieve steady state (e.g. isoflurane and nitrous oxide combination with oxygen). End-tidal agent monitoring is essential, although it is worth noting that the expired endtidal agent concentration may not necessarily be reflective of effect site concentration during the onset of volatile anaesthesia.
Opioids
Concerns around placental transfer of opioids and respiratory depression of the fetus have historically limited the use of opioids during RSI in obstetric patients.
The use of opioids at induction of anaesthesia in obstetrics has a number of advantages for a small cohort of patients, such as those with preeclampsia or significant cardiac disease, as they can suppress the sympathetic response to laryngoscopy, improve intubating conditions, and contribute to a reduced risk of awareness (2, 5).
NAP5 highlighted that whilst the “appropriate use of opiates” may reduce the risk of AAGA in obstetric practice, this was an area warranting further research to clarify optimum dosing and timing of administration during anaesthesia for CS. Some anaesthetists in the UK advocate the use of short acting opioids for induction of GA in pregnant patients to reduce the risk of AAGA, but this is not widely practised.
Recent studies have demonstrated that whilst bolus doses of alfentanil (10 μg kg-1 ) (12) and remifentanil (1 μg kg-1) (13) at induction of anaesthesia attenuate maternal stress response to laryngoscopy, they can also predispose the neonate to transient respiratory depression which may require assisted manual ventilation for a short period and rarely naloxone administration (13). In patients with pre-eclampsia or cardiac disease however, the risk-benefit ratio of the mother and infant needs to be assessed.
Depth of anaesthesia monitors
Depth of anaesthesia (DOA) and end-tidal agent monitors can be used to aid clinical assessment and ensure adequate DOA. This is especially important when muscle relaxants are used.
In the UK, The National Institute for Health and Clinical Care Excellence (NICE) recommends the use of DOA monitors to reduce the risk of awareness in high-risk groups, which includes obstetric patients (14).
BIS is one example of a DOA monitoring technology. It uses aggregate electroencephalogram (EEG) data to calculate a dimensionless number (ranging from 0 – 100); a reading of 40 – 60 correlates with an adequate depth of GA (and a low probability of awareness) and <20 with burst suppression (which consists of alternate periods of slow waves of high amplitude with periods of no activity; this is seen in general anesthesia or coma and may be suggestive of excessive DOA).
Studies in obstetric practice suggest a MAC of 0.75 should be targeted to minimise the risk of awareness. This correlates with a BIS value of <60 which has been demonstrated to not adversely affect uterine tone or fetal outcomes (15).
MANAGING PATIENTS WHO EXPERIENCE AWARENESS
It is vitally important that patients who experience AAGA are followed up thoroughly, and it is recommended that departments have guidelines and pathways for managing such patients (2). Patients should be offered immediate empathetic explanations to help decrease long-term psychological harm and should be offered follow-up in an anaesthetic clinic with access to counselling services.
Anaesthetists (and other staff members) who have been involved with a patient who experienced AAGA may benefit from support and counselling.
Root-cause-analysis (RCA) of AAGA events can be traced back to organisational or system errors, as highlighted in the NAP5 study, so it is imperative that thorough RCAs are performed to identify and amend systems errors to improve patient care through delivery of safe anaesthesia practice, and prevent future patient harm.
SUMMARY
- Obstetric anaesthesia is a high-risk area for accidental awareness during general anaesthesia (AAGA)
- Risk factors include: emergency surgery; inexperienced anaesthetists; the “intravenous-inhalational interval”; difficult or failed intubation
- Strategies to reduce the incidence of awareness include: appropriate dose of anaesthetic induction agent; use of nitrous oxide for maintenance of anaesthesia; and use of depth of anaesthesia (DOA) monitors
- Patients who experience AAGA are at risk of long-term sequelae including post-traumatic stress disorder (PTSD) and should be offered appropriate counselling
REFERENCES AND FURTHER READING
- Avidan MS, Zhang L, Burnside BA, et al. Anaesthesia awareness and the bispectral index. N Engl J Med 2008;358: 1097-1108.
- Pandit JJ, Cook TM, the NAP5 Steering Panel. NAP5. Accidental Awareness During General Anaesthesia. London: The Royal College of Anaesthetists and Association of Anaesthetists of Great Britain and Ireland 2014 ISBN 978-1-900936-11-8.
- Leslie K, Chan MT, Myles PS et al. Posttraumatic stress disorder in aware patients from the B-aware trial. Anesth Analg 2010; 110:823-8.
- Ashpole KJ, Cook TM. Litigation in obstetric general anaesthesia: an analysis of claims against the NHS in England 1995–2007. Anaesthesia 2010:65; 529-30.
- Chaggar RS, Campbell JP. The future of general anaesthesia in obstetrics. BJA Education 2017;17:79-83
- Murdoch H, Scrutton M, Laxton CH. Choice of anaesthetic agents for caesarean section: A UK survey of currentpractice. Int J Obstet Anesth 2013; 22;31–5.
- Lucas DN, Yentis SM. Unsettled weather and the end for thiopental? Obstetric general anaesthesia after the NAP5 and MBRRACE-UK reports. Anaesthesia 2015; 70:375–9.
- Rucklidge, M. Up-to-date or out-of-date: does thiopental have a future in obstetric general anaesthesia? Int J Obstet Anesth 2013; 22:175–8.
- Levy DM. Emergency Caesarean section: best practice. Anaesthesia 2006;61:786-91
- Quinn AC, Milne D, Columb M Et al. Failed tracheal intubation in obstetric anaesthesia: 2 yr national case–control study in the UK. Br J Anaesth 2013; 110:1; 74–80, https://doi.org/10.1093/bja/aes320
- Becker DE, Rosenberg M. Nitrous Oxide and the Inhalation Anesthetics. Anesthesia Progress 2008; 55:4; 124- 131. doi: 10.2344/0003-3006-55.4.124.
- Gin T, Ngan-Kee WD, Siu YK et al. Alfentanil given immediately before the induction of anaesthesia for elective Cesarean delivery. Anesth Analg 2000; 90:1167–72.
- Ngan Kee W, Khaw K, Kwok CM et al. Maternal and Neonatal Effects of Remifentanil at Induction of General Anaesthesia for Cesarean Delivery. Anesthesiology 2006;104:14-20
- National Institute for Health and Care Excellence (NICE). Depth of anaesthesia monitors – Bispectral Index (BIS), E-Entropy and Narcotrend-Compact M. Diagnostics guidance [DG6]. Available from: https://www.nice.org.uk/Guidance/DG6 (accessed 2 Feb 2018).
- Chin KJ, Yeo SWA. BIS-guided study of sevoflurane requirements for adequate depth of anaesthesia in Caesarean section. Anaesthesia 2004;59:1064–8



