Disorders in acid-base balance are commonly found in patients admitted to intensive care. Clinicians responsible for these patients need a clear understanding of acid-base pathophysiology in order to provide effective treatment for these disorders. These tutorials are designed to supplement the tutorial in the Basic Sciences section of ATOTW and will concentrate on aspects of metabolic acidosis pertinent to critical care. In part 1 we shall cover the assessment of metabolic acidosis, including determining cause and assessing severity. In part 2 we shall cover specific types of metabolic acidosis commonly encountered in critical illness.
Clinical Scenarios
Case 1.
A known diabetic patient presented with severe diabetic ketoacidosis. He was drowsy, exhibited classical Kussmaul respiration and proceeded to have a respiratory arrest whilst being admitted to ICU. After immediate intubation the trainee ventilated the patient with the ventilator’s default settings (rate 12, tidal volume 500ml, PEEP 5cmH2O FiO2 50%) and attempted to secure arterial and central venous access. Shortly after intubation the patient became asystolic and could not be resuscitated.
Why did this patient have a respiratory arrest?
Why did he deteriorate after intubation?
Case 2
A young woman was admitted to the surgical ward with a history of severe vomiting and increasing right iliac fossa abdominal pain. On examination she was found to have a rigid abdomen, with visible subdiaphragmatic gas on CXR. She was cardiovascularly unstable and her admission bloods showed Hb 17.4, WCC 24.6, Plateletsts 79, Na+ 125, K+ 4.3, Cl– 93, urea 20.4 creatine 310 (mcmol/l) and blood gas analysis showed:
- pH – 7.1
- pCO2 – 5.2 kPa
- pO2 – 29.3 kPa
- HCO3– – 14.3
Interpret these results. What is the likely cause of her acidosis?
How do you interpret her chloride level?
Is her compensation adequate/maximal?
Case 3.
An elderly lady was admitted from a care home with a week long history of severe diarrhoea. She was dehydrated and hypotensive. Admission bloods revealed Na+ 134, K+ 2.5, Cl– 122, urea 15.4, creatine 280, and blood gas analysis revealed:
- pH – 7.21
- pCO2 – 2.9 kPa
- pO2 – 19.5 kPa
- HCO3– – 6.4
Describe the acid base disorder present.
What is the likely aetiology?
Is her compensation adequate?
Introduction
Metabolic acidosis is a common component of critical illness. Evaluation of this component can aid diagnosis, assess severity (and likely outcome) and allow the clinician to determine whether current treatments are proving effective.
Definitions
Metabolic acidosis exists when there is an excess level of fixed or exogenous acids in the blood. Fixed acids include hydrochloric acid from arginine, lysine and histidine metabolism, sulphuric acid from methionine and cystine metabolism, phosphoric acid, ketoacids and lactic acid. This is always accompanied by a drop in plasma bicarbonate concentration (relative to the bicarbonate concentration present prior to the onset of the acidosis). This drop in bicarbonate can either be caused by bicarbonate loss or by the presence of an acid.
It should be noted that the body can accommodate significant alteration in acid burden through buffering (bicarbonate being the primary buffer in blood) and the presence of a metabolic acidosis does not always imply the presence of acidaemia (a low blood pH). Please refer to the basic science acid/base tutorial for further details on buffering in blood. Blood pH will only fall appreciably when the buffering capacity of the blood becomes overwhelmed.
A drop in bicarbonate concentration is the hallmark of metabolic acidosis.
Evaluation
When evaluating a critically ill patient with a metabolic acidosis it is necessary to determine the type of acidosis in order to identify the cause of the acidosis. To classify metabolic acidosis it is useful to calculate the anion gap, delta ratio and, if present, the size of the osmolar gap. These concepts are explained below.
The role of the anion gap
Anions are negatively charged ions (like bicarbonate) and are attracted to anodes. Cations are the opposite; they are positively charged ions and they are therefore attracted to a cathode. The anion gap is defined as the concentration difference between the major (measured) cations and anions within the plasma and is normally 12 to 18 mmol/l. [2] Anionic proteins, phosphate, sulphate and low levels of organic acids, which are not measured, account for the difference (i.e. the ‘gap’). When examining the cause of a metabolic acidosis it is useful to calculate the anion gap.
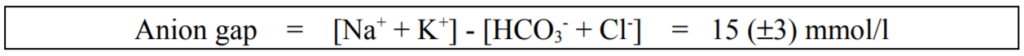
A normal anion gap implies primary bicarbonate loss:
- Plasma bicarbonate is low (the hallmark of acidosis) and chloride concentration is raised.
- This bicarbonate loss may be:
- Gastrointestinal (diarrhoea, fistula)
- Renal (renal tubular acidosis, drug effect).
- Also occurs with rapid intravenous infusion of normal saline (excess chloride) or infusions of parenteral nutrition rich in cationic amino acids (e.g. arginine).
An increased anion gap implies that fixed acids are being retained or an abnormal organic acid is present. Plasma bicarbonate is low and chloride concentration is normal.
Fixed acids may be retained in:
- uraemia
- ketoacidosis (diabetic, alcoholic)
- lactic acidosis.
If fixed acids are normal, exogenous acids should be considered:
- salicylate poisoning
- methanol poisoning
- ethylene glycol poisoning
The role of the Delta Ratio
The delta ratio is defined as:
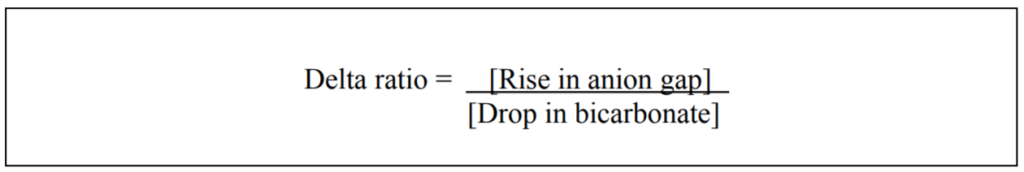
In an increased anion gap acidosis every molecule of fixed acid that dissociates in blood will create one hydrogen ion and one acid anion. The H+ will bind one molecule of bicarbonate (buffering), resulting in a drop in bicarbonate of one for each rise in the anion gap of one. The delta ratio for an increased anion gap acidosis will consequently approach one.
If the delta ratio is very low (less than 0.4) then it supports the diagnosis of a normal anion gap acidosis. If the delta ratio is high (greater than 2) then it suggests that the bicarbonate was high prior to the onset of the acidosis (as seen in compensated respiratory acidosis). A ratio between 0.4 and 2.0 suggests a mixed picture.
Limitations of the Anion Gap and Delta Ratio
Whilst the anion gap is useful in evaluating metabolic acidosis it is not sensitive. The normal range is quoted as 12 to 18 mmol/l which means it is possible for a patient with a normal anion gap of 12 to acquire a severe lactic acidosis (plasma lactate greater than 5 mmol/l) without leaving the normal range. The anion gap is also affected by plasma albumin (an important unmeasured anion) and low albumin levels can significantly offset an anticipated rise in anion gap.
Similarly the theory behind the delta ratio is also flawed. Bicarbonate only accounts for 60% of the buffering capacity of blood and this affects the assumption that one molecule of H+ will bind one molecule of bicarbonate. However when the delta ratio is very high or low it can still prove useful.
Role of the Osmolar Gap
The osmolar gap represents the difference between a sample’s measured osmolality and its calculated osmolarity. It is a useful calculation to perform if alcohol poisoning is suspected as we will discuss in Part B of this tutorial.
Osmolality is measured in the laboratory with an osmometer that either assesses the depression of a sample’s freezing point or the depression of its vapour pressure. It is preferable to use the former as any volatile alcohols in the sample will evaporate as the sample is heated and the results from this method will be inaccurate.
Osmolarity can be calculated using various formulae. One such formula is:
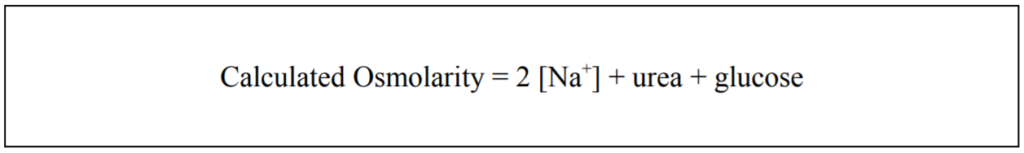
Osmolality and osmolarity differ according to whether the number of osmotically active particles is dissolved in a kilogram or a litre of solvent respectively. The calculated osmolarity utilises the plasma concentration of sodium, glucose and urea. Even though sodium and chloride represent the most important determinants of osmolarity in plasma, chloride concentration is not commonly available. The formula is simplified by taking into account the incomplete dissociation of sodium chloride in plasma. The contribution from sodium and chloride is therefore calculated from the sodium concentration alone by multiplying sodium concentration by the dissociation constant for sodium chloride (1.86) and dividing this by the proportion of water in serum (0.93). Whilst this formulae is quick and easy its accuracy has been questioned and several centres now prefer to use
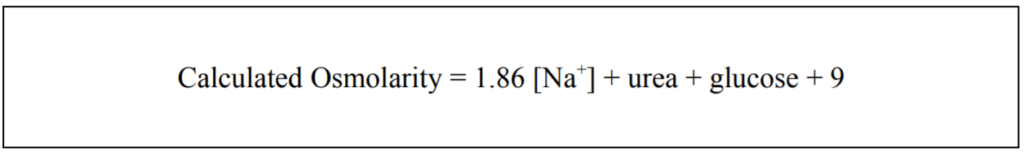
The measured and calculated values should lie within 10 of each other (the difference being created by the inaccuracy of the calculation and the inaccuracy of the osmometer) and if the gap is larger it suggests the presence of unmeasured
osmotically active species.
It is important to realise that the osmolar gap has significant limitations as well. When considering alcohol poisoning the osmolar gap is only present as an early feature and returns towards normal as the alcohol is metabolised and the associated metabolic acidosis develops. Similarly the osmolar gap is not sensitive as the large molecular weight of ethylene glycol accounts for a fatal ingestion, only causing a small rise in osmolar gap.
Compensation for Metabolic Acidosis
When treating critically ill patients with metabolic acidosis it is important to consider the adequacy of their ventilatory response when deciding on treatment priorities. Buffering provides the main means of accommodating a metabolic acidosis. As buffering capacity is exceeded, acidaemia develops. Once this rise in hydrogen ion concentration has reached the CSF it is detected by chemoreceptors and compensation occurs by reducing CO2 levels through hyperventilation (first described by Kussmaul). Detection of low pH in CSF, rather than blood, explains the delay in this compensation and why rapid onset acidosis (seen with convulsions for example) tends not to stimulate respiration in spite of a low blood pH.
Even though respiratory compensation occurs relatively quickly, it can take up to twelve hours to reach maximal capacity. This maximal capacity can be calculated

If the patient’s pCO2 lies within 0.5 kPa of this calculated value, then the respiratory response is appropriate. If the pCO2 is higher then either the compensation is in a very early stage or the patient has a superimposed respiratory acidosis. If this is the case then earlier intervention with respiratory support is indicated.
When providing respiratory support in patients with a metabolic acidosis it is important to remember that respiratory compensation causes an increased minute volume. If you instigate controlled ventilation with a normal minute volume then the pCO2 level will rise rapidly towards normal, the acidaemia will worsen, and the patient will become acutely unstable. Young fit patients with severe diabetic ketoacidosis can generate huge minute volumes (20-30l/min) and drop their pCO2 to below 2 kPa. If you ventilate them with a minute volume of 5 l/min then their acidosis will deteriorate profoundly.
Pitfalls in Assessing Metabolic Acidosis
It is impossible to interpret arterial gases accurately without considering the history and presentation first. Consider the following arterial gas:
- pH – 7.1
- pCO2 – 10.5 kPa
- pO2 – 29.3 kPa
- HCO3– – 24.3
The interpretation of this result will vary according to the clinical presentation:
- If the gas sample was taken from a young unconscious patient, admitted through the emergency room, with pinpoint pupils then you would interpret the gas as showing primary respiratory acidosis from opiate overdose.
- If the gas was taken from an elderly man with severe COAD presenting with sepsis then the gas interpretation will be different.
- Full compensation for a chronic respiratory acidosis should raise the bicarbonate by 3 x (pCO2 – 5.3). If the pCO2 level is chronically raised then you would expect the bicarbonate to be 36 mmol/l.
- This bicarbonate is significantly lower and this suggests a metabolic acidosis superimposed on the background of a compensated respiratory acidosis.
This serves to highlight the importance of basing gas interpretation on clinical details. Other pitfalls arise from failing to recognise the limitations of the anion gap, delta ratio and osmolar gap, as discussed earlier in the tutorial.
Summary
Metabolic acidosis is commonly encountered in intensive care and correct management requires confirmation that an acidosis is present, determination of its nature and then determination of the probable cause. Identification of the root cause will then allow specific treatment with improved efficacy.
Case Examples
Case 1.
In this case the patient presented with a metabolic acidosis caused by loss of diabetic control, relative insulin deficiency and consequently ketoacidosis. (This will be covered in more detail in part B of the tutorial). Kussmaul respiration implies respiratory compensation through hyperventilation. The rise in minute volume can be substantial and maintaining this compensation is very strenuous. As time passes patients tire, the minute volume decreases and the acidosis worsens rapidly. Young patients in particular can maintain full respiratory compensation almost to the point of respiratory arrest, as in this example. Elderly patients tend to tire more slowly and this allows timely recognition and support.
When taking over a patient’s ventilation, in the setting of a metabolic acidosis, it is vital that the minute volume used is raised appropriately. If standard ventilation settings are applies, as in this case example, the drop in alveolar ventilation postinduction will remove the respiratory compensation and the acidosis will deteriorate dramatically. This is likely to be the cause of this patient’s cardiovascular collapse.
Case 2.
The clinical presentation is of an acute abdomen in a previously fit patient. The raised haemoglobin, urea and creatinine suggest that the patient is dehydrated and this raises the possibility of an associated lactic acidosis due to inadequate tissue perfusion. The arterial gasses confirm that acidaemia is present (low pH) and the low bicarbonate implies that the acidosis is metabolic.
The anion gap is raised at 35 and this strongly suggests the presence of an increased anion gap acidosis. The delta ratio is calculated then this is raised at 3.5 and this suggests a pre-existing metabolic alkalosis. While it is impossible to be absolutely sure this would seem to fit clinically. The vomiting that occurred in the days leading up to admission may have caused a metabolic alkalosis (loss of hydrogen ions and chloride) that was confounded by a superimposed lactic acidosis on admission. This would also explain why a severe increased anion gap acidosis only dropped the pH to 7.1 in this example, when you would expect the pH to be lower.
In terms of her respiratory compensation, it is possible to calculate the expected pCO2 from the bicarbonate level and this would suggest that maximal compensation should drop the pCO2 to 3.8 kPa. The fact that her pCO2 is higher should serve as a warning of imminent fatigue and ventilation should be supported as quickly as possible in order to prevent cardiorespiratory collapse.
Case 3
This lady presented with a low pH on arterial gases and low bicarbonate – this implies that a metabolic acidosis is present. The extent of the dehydration raises the possibility of a lactic acidosis however this would create an increased anion gap. The anion gap in this instance is low (8.5) and the delta ratio is 0.5. This strongly supports the presence of a normal anion gap acidosis secondary to bicarbonate loss from the gut.
With a bicarbonate of 6.4 you would expect the patient to drop her pCO2 to 2.28 and in this instance her respiratory compensation is appropriate.
References
- Cohen RD, Woods HF. Disturbances of acid-base homeostasis. In : Weatherall DJ, Ledingham JGG, Warrell DA, eds. Oxford Textbook of Medicine. Oxford: Oxford University Press, 1987; 9.174
- Hoffman RS, Smilkstein MJ, Howland MA, Goldfrank LR. Osmol gaps revisited. Normal values and limitations. Journal of Toxicology – Clinical Toxicology. 1993; 31(1): 81-93.
- Salem MM, Mujais SK. Gaps in the anion gap. Archives of Internal Medicine. 1992;152: 1625-1629.
- Brandis K. Acid-base physiology. From http://www.anaesthesiamcq.com/AcidBaseBook/ABindex.php
- Stewart PA. How to understand acid-base. A quantitative acid-base primer for biology and medicine. From http://www.acidbase.org/



