Basic Sciences
QUESTIONS
Answer true or false:
- Acute pain
- Is usually proportionate to the injury sustained
- Is usually self-limiting
- Can affect distant organ systems
- Is influenced by a patients psychological state
- Central sensitisation
- Is not seen in acute pain
- Can cause hyperalgesia
- Can cause allodynia
- Is not reversible
- Nociceptors
- Are low threshold nerve endings
- Can be activated by electrical stimulus
- Have a fixed threshold for activation
- Contain phospholipase A2
Introduction
Acute pain is a common reason for presentation to hospital for many medical and surgical pathologies as well as being a key component of successful anaesthetic management. Despite this fact, it is only within the past 20 years that the importance of acute pain has been fully recognised. This followed publication of the first guidelines and studies of best practice in Australia in 1988. Subsequent research has made significant advances in identifying the mechanisms that underpin acute pain; its physiological and clinical consequences and the multifactorial nature of pain perception.
This series of tutorials aims to explain current knowledge of the basic science underlying pain mechanisms and treatment, with later tutorials covering the clinical management of specific pain problems. They are written for a broad audience and consequently will start from a basic level.
Research has consistently demonstrated that pain is very commonly under-recognised and under-treated. This finding has been repeated throughout the world in different cultures and healthcare settings. It is the hope of the authors that this series will help to equip the reader with the knowledge to improve their ability to recognize and treat pain more effectively.
Acute pain
In evolutionary terms, acute pain serves an essential role for the individual as it informs of actual or impending tissue damage. In most circumstances, acute pain is self-limiting and proportionate to the degree of injury sustained. Physical, thermal or chemical stimuli initiate a cascade of events that lead to predictable physiological, psychological and behavioural responses. Nociceptive signals exert profound changes within the peripheral nervous system in response to injury but also extend to affect the spinal cord, higher centres within the brain and remote organ systems through the neuro-humeral and endocrine response.
Acute pain is detected in the peripheral nervous system by specialised primary nerve fibres known as nociceptors. These nerve endings transduce physical and chemical stimuli in a logarithmic scale depending on the intensity of stimulus applied. Once a threshold has been reached, an action potential is produced that travels along a peripheral nerve, through the sensory cell bodies of the dorsal root ganglion and into the spinal cord before ascending to the brain. However, the steps in conveying the nociceptive signal from the peripheral nerves to the brain should not be regarded as a simple relay system. Rather, there is a complex interaction with other neural processes that may augment or diminish the signal at different stages.
The overall perception of acute pain is however not always entirely determined by, or proportionate to, the degree of tissue damage. It is not uncommon in anaesthetic practice to encounter patients who have very different analgesic requirements despite undergoing identical surgical procedures. For example, some patients may only gain pain relief from doses of opioids that render them unconscious while others may not require strong opioids at all. The majority of patients will fall between these two extremes but demonstrates that good pharmacological management and procedural skills, though essential elements of pain management, can be inadequate if used in isolation.
Pain has strong sensory and emotional components that interact together to produce an overall ‘pain experience’. For example, a patient may have previously experienced severe post-operative pain that can lead to fear and anxiety of subsequent surgery. An expectation of pain may in itself lead to an increase in perceived pain and a limited response to analgesics. This gives rise to an altered affective state where patients may be irritable, anxious and afraid and this can feedback to further exacerbate the problem. Attention must therefore be paid to the psychological and emotional state of the patient and address these issues at an early stage with appropriate preparation, explanation and reassurance to ensure optimal pain control.
- Emotion can have a significant impact on pain perception
- Addressing the patient’s anxieties can reduce the severity of pain suffering
Nociceptors and Nerve fibres
Acute pain perception begins with activation of specific sensory nerves, termed nociceptors. These are un-encapsulated free nerve endings that are present in the skin, deep somatic tissue and viscera. Providing the stimulus is suitably intense, high threshold nociceptors will still activate in the absence of actual tissue damage. Nociceptors are probably activated by mechanical distortion of the nerve leading to an increase in H+ and K+ concentration. Nociceptors can be divided into two main classes; A-delta and C fibres. The main properties of these fibres are summarised in the table below. A-beta fibres are included for comparison.
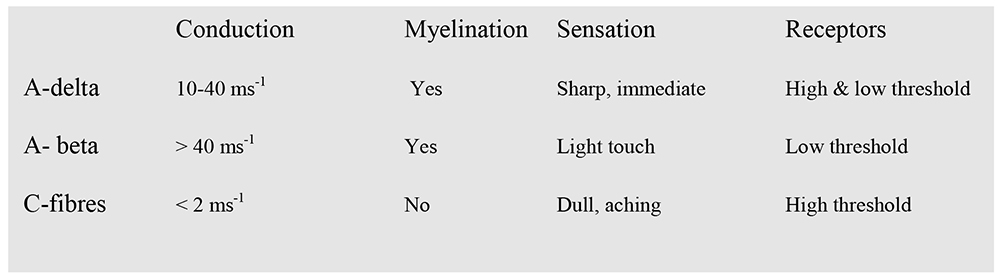
Table 1: Classification and properties of neurones
In contrast to A-delta and C-fibres, A-beta fibres respond to light tactile as well as noxious stimuli. However, A-beta fibres do not increase their rate of firing when the intensity of stimulus increases. The result is that they do not encode noxious stimuli.
Sodium Channels
Sodium channel activation underpins the initiation of an action potential and ultimately the perception of acute pain. Voltage gated sodium channels are the site of action of local anaesthetics that further demonstrate their crucial role in pain signaling and as a site of clinical importance. Recent basic science research reveals that sodium channels are not a single homologous group; rather multiple sub-types exist with differing physiological properties.
Peripheral sensitisation
Any injury to body tissue leads to the release of a multitude of chemical mediators and immune cells that result in the common clinical signs that are seen in acute pain and inflammation. The mechanism behind this initially involves the synthesis of arachidonic acid as a result of the action of phospholipase A2 on membrane lipids. Arachidonic acid is then acted upon by cyclo-oxygenase to synthesise prostaglandin that in-turn directly lowers the activation threshold for A-delta and C-fibres.
At the site of injury, inflammatory mediators such as histamine, bradykinin and leukotrienes are released in addition to prostaglandins. These factors are collectively regarded as the ‘inflammatory soup’ that surrounds peripheral nociceptors resulting in a further reduction in their membrane threshold and activation of dormant receptors. Sensitised receptors display an increased basal (unstimulated) rate of discharge and a supra-normal increase in discharge strength in response to any increase in stimulus. This is easily demonstrated clinically as an area of hyperalgesia extending beyond the boundary of a surgical incision.
Hypersensitivity is also mediated by the Capsaicin/vanilloid receptor TRPV-1. The physiological changes seen within inflamed or infected tissue i.e. heat and acidic pH are known to activate the TPRV-1 receptor and enhance pain sensitivity. Other molecules within nociceptors that are known to enhance pain transmission include substance-P and Calcitonin Gene related peptide (CGRP). These two factors are often released simultaneously within the cell. Substance P activates second order neurones that send a ‘pain’ signal to the brain while CGRP contributes to neurogenic inflammation by causing vasodilation and hence warmth, redness and swelling.
- Peripheral sensitisation: A variety of mediators are released in response to tissue damage which decrease the threshold for nociceptor activation
Central sensitisation
Nociceptive signals from peripheral nerves are conveyed from afferent nerve fibres through the dorsal horn of the spinal cord for relay to the thalamus, limbic and cortical areas. This leads to the perception of acute pain, withdrawal reflexes and behavioural responses. Considerable modulation of afferent nociceptive signals occur within the spinal cord to either enhance or attenuate the signal. As with the peripheral nervous system (PNS), nociceptive input can change the structure and function of the central nervous system (CNS) to respond in an exaggerated fashion such that even innocuous stimuli may be felt as painful i.e. allodynia. This is termed central sensitisation.
Fundamental to the process of central sensitisation is the N-methyl, D-adenine (NMDA) receptor. This receptor is found widely within the CNS but features prominently in the dorsal root ganglion (DRG). In response to an afferent signal, glutamate is released from a primary afferent neurone that binds to the NMDA receptor resulting in an influx of calcium and potassium. This leads to nerve de-polarisation and heightened sensitivity to circulating neurotransmitters. At the molecular level, nociception induces alteration in second order neurones such as phosphorylation of NMDA and AMPA receptors. These processes contribute to the process of central sensitisation.
The processes outlined above are normal physiological consequence of an injury and should return to their pre-injury state after a period of time, usually before complete healing has occurred. However, in some patients this does not occur and the nervous system remains in a heightened, or sensitised, state after tissue healing has completed. In this regard, it is well established that the severity of acute post-operative pain has implications for the development of chronic pain. The reasons for this remain obscure but may be due to a failure within endogenous analgesic pathways to reset the central nervous system.
- Central sensitisation: Changes occur within both the spinal cord and brain which enhance nociceptive transmission and perception
- Failure of this state to return to normal is implicated in the development of chronic pain states
NEURO-ANATOMY
Multiple tracts and centres exist within the central nervous system which are responsible for the transmission, modulation and perception of noxious stimuli. It is important to realise that these areas should not be considered as fixed or functioning in isolation. Rather, they are subject to change from both descending and ascending pathways and can alter or expand their connections to interact with adjacent nerves.
Spinal Cord
Dorsal horn neurones
Cell bodies of afferent nerves lie in the DRG with fibres synapsing in the dorsal horn of the spinal cord. The output from the dorsal horn is however dependent on other neuronal input to the synapse. Afferent neurones may divide prior to entering the cord and send branches cephalad or caudad in the longitudinal tract of Lissauer before synapsing with dorsal horn neurones. The result of this being that a single C-fibre afferent may be responsible for innervating dorsal horn neurons at multiple spinal levels.
Wide Dynamic Range neurones
Neurones in the 3rd, 4th and 5th layers of the spinal cord can respond to noxious input as well as light touch. They are therefore termed wide dynamic range neurones (WDR). These neurones display the important properties of ‘wind-up’ and ‘convergence’. Wind-up is the state where neuronal output increases in response to continuous, low frequency input whereas ‘convergence’ describes how somatic and visceral afferents converge on the same neurone. This accounts for the phenomenon of referred pain where visceral pain is experienced in parts of the body remote from the primary site of pain but are supplied by the same dorsal horn neurone.
Rexed Laminae and Ascending Tracts
The gray matter of the spinal cord can be divided into ten physiologically and histologically distinct layers known as rexed laminae. Laminae 1 to 6 and 10 are the sites that sensory nerves synapse with dorsal horn cells and are important in pain transmission. Laminae 7-9 are involved with motor function.
Lamina 1: Also known as the marginal layer. Cells in this area respond almost exclusively to noxious stimuli and send axons to the spinothalamic tract or parabrachial nucleus.
Lamina 2: More commonly known as the substantia gelatinosa. Cells of lamina 2 receive afferent input directly from small diameter fibres and synapse with many inter-neurones. Cells of lamina 2 display considerable cross-linking with cells in lamina 1 and 5 modulating their function.
Laminae 3 and 4: respond only to innocuous stimuli.
Lamina 5: These cells receive input from large and small diameter afferent fibres from all superficial and deep structures. They respond to noxious as well as innocuous stimulus and receive signals from a large area of the body. It is for this reason they are termed wide-dynamic range cells.
Following a nociceptive impulse traversing and undergoing modulation in the spinal cord the signals then ascend in a number of different tracts. These are summarised below.
Spinothalamic tract
Cells of the STT originate in Laminae 1, 4 and 5 of the spinal cord before crossing the cord at the anterior commisure and ascending in the contralateral anterolateral quadrant. This tract is divided during cordotomy in the treatment of intractable cancer pain
Spinoreticular tract
This is located in the anterolateral quadrant of the spinal cord. Axons from the SRT interact with neurones involved in descending pain modulation and may explain the phenomenon of counter-irritation. This involves one type of pain reducing the severity of another, the idea of ‘rubbing it better’.
Spinomesencephalic tract
This tract supplies sensory input to the superior colliculus and periaqueductal grey (PAG). The PAG is involved in the autonomic and somatomotor reactions to pain whereas the superior colliculus is involved in the integration, orientation and behavioural response to pain.
- Nociceptive pathways are complex, dynamic and influenced by descending pathways from higher cortical centres
Conclusion
Acute pain management is a fundamental component of anaesthetic practice. The anatomy and physiology of acute pain is complex with multiple neuro-chemical and receptor mediated effects. Acute pain perception is not only a product of the magnitude of tissue damage but is influenced by the psychological and emotional state of the patient. Understanding of these concepts is important in formulating an individualised, rational analgesic plan for a patient.
Summary
- Acute pain affects multiple organ systems
- Acute pain causes changes to the peripheral and central nervous system that can increase sensitivity to further stimulus, i.e. sensitisation
- The emotional and psychological state of the patient have a major influence on acute pain perception and analgesic requirements
- Severe acute pain can lead to chronic pain
- The dorsal root ganglion is a key area for pain transmission and modulation
MCQ ANSWERS
- T, T, T, T
- F, T, T, F
- F, T, F, T
WEBLINKS
REFERENCES
- Writing Committee of the WHO Consultation Aspects of Pandemic (H1N1) 2009 Influenza. Clinical aspects of pandemic 2009 influenza A(H1N1) virus infection. NEJM 2010; 362: 1708—19
- Kumar A, Zarychanski R, Pinto R, Cook DJ, Marshall J, Lacroix J et al. Critically ill Patients with 2009 Influenza A (H1N1) Infection in Canada. JAMA 2009; 302(17) 1872—1879
- Pandemic Influenza Infection Control guidance for critical care, DH, April 2008 (available on DH website: www.dh.gov.uk/en/Publicationandstatistics/PublicationsPolicyAndGuidance/DH_084178)
- Clare D. Ramsey, Duane Funk, Russel Miller 111, A Kumar: Ventilator Management of hypoxaemic respiratory failure attributable to H1N1 novel swine origin influenza virus. Crit.Care Med 2010 Vol. 38(4) Suppl
- Song Mi Moon, Sung-Han Kim, Min Hee Jeong, Eun Hyee Lee, Tae-Sung Ko. Acute Encephalopathy and Pandemic (H1N1) 2009 Emerging Infections Diseases. www.cdc.gov/eid. vol.16.No.11, November 2010 1811-1813
- Luis Miguel Noriega, Renato J. Verdugo, Rafael Araos, Jose Manuel Munita, Violeta Diaz, Alejandra Marcotti et al .Pandemic Influenza A (H1N1) 2009 with neurological manifestations, a case series. 2010 Blackwell Publishing Ltd, Influenza and Respiratory Viruses, 4, 117-120
FURTHER READING
- Wall P.D, McMahon S.B., Koltzenburg M. Wall and Melzack’s textbook of Pain. Elsevier/Churchill Livingstone, 2006
- Carr D.B; Goudas L.C. Acute Pain. The Lancet. 1999; 353: p 2051- 2058
- Sorkin L.S, Wallace M.S. Acute Pain Mechanisms. Surgical Clinics of North America.1999; 79 (2):213-229
- Ekman E.F, Koman L.A. Acute Pain Following Musculoskeletal Injuries and Orthopaedic Surgery. Mechanisms and Management. Journal of Bone and Joint Surgery. 2004;86:1316-127



