Intensive Care Medicine
QUESTIONS
Self assessment
- List 5 common causes of pancreatitis. What percentage of episodes are left unexplained despite investigation?
- What are the characteristics features of the pain in acute pancreatitis?
- Which radiological investigation is most useful? When should it be performed? What features of this investigation can be regarded as severe prognostic indicators?
- A patient with severe acute pancreatitis is likely to have an unstable circulation – what monitoring and measures can be taken to correct this?
- What organisms commonly cause local pancreatic infection as a complication of pancreatitis?
- Name three potential methods of reducing infection in pancreatitis?
- Does surgery have a role in the management of severe acute pancreatitis?
The answer to the questions appear in the text below.
Overview
- Introduction
- Causes and pathogenesis
- Clinical features
- Investigations and severity scoring systems
- Principles of general management
Introduction
Acute pancreatitis is characterized by upper abdominal pain and elevated levels of pancreatic enzymes in the blood. This is associated with varying degrees of endocrine and exocrine dysfunction.
The disease may be mild and self-limiting or may rapidly progress to multi-organ dysfunction (MODS) with or without sepsis, and ultimately to death. It is usually possible to establish a cause that is treatable.
Management of the more severe forms is labour and resource-intensive and very timeconsuming, but long-term follow-up reveals a good quality of life, although many suffer from long-term exocrine and endocrine problems.
Causes and pathogenesis
The pathogenesis of pancreatitis is not fully understood. Of the numerous conditions known to cause pancreatitis (see Table 1), gallstones and chronic alcohol abuse account for 75 percent of cases in the United States. There is great geographical variation and the degree of inflammatory response to certain causative agents may have genetic predisposition, making prognosis different in various ethnic groups. For example, alcoholic pancreatitis in Soweto, South Africa has a high mortality.
The mortality in the first two-week period is usually due to systemic inflammatory response syndrome (SIRS) and organ failure; thereafter it is usually due to sepsis and its complications.
Advances in diagnostic and therapeutic interventions have lead to a decrease in mortality from acute pancreatitis, especially in those with severe, often necrotizing, pancreatitis. While the overall mortality in all hospitalized patients with acute pancreatitis is approximately 10 percent, the mortality in the subset with severe acute pancreatitis may be as high as 30 percent.
Mechanical obstruction to the ampulla can be caused by a number of different conditions, but is most commonly caused by the passage of a gallstone through from the common bile duct. Cholecystectomy and clearing the common bile duct of stones prevents the recurrence of the disease.
Approximately 10 percent of chronic alcoholics develop attacks of clinically acute pancreatitis that are indistinguishable from other forms of acute pancreatitis. Alcohol may act by increasing the synthesis of enzymes by pancreatic acinar cells to synthesize the digestive and lysosomal enzymes that are thought to be responsible for acute pancreatitis.
Causes such as hyperlipidaemia (often co-existing with alcoholism) and drug induced pancreatitis can usually be identified from the history, but the diagnosis should only made having carefully excluded biliary disease.
Up to 30% of attacks may still be unexplained following normal biochemical and ultrasound investigation. The recurrence rate in this group of patients is rare. Further extensive investigation is probably not indicated.
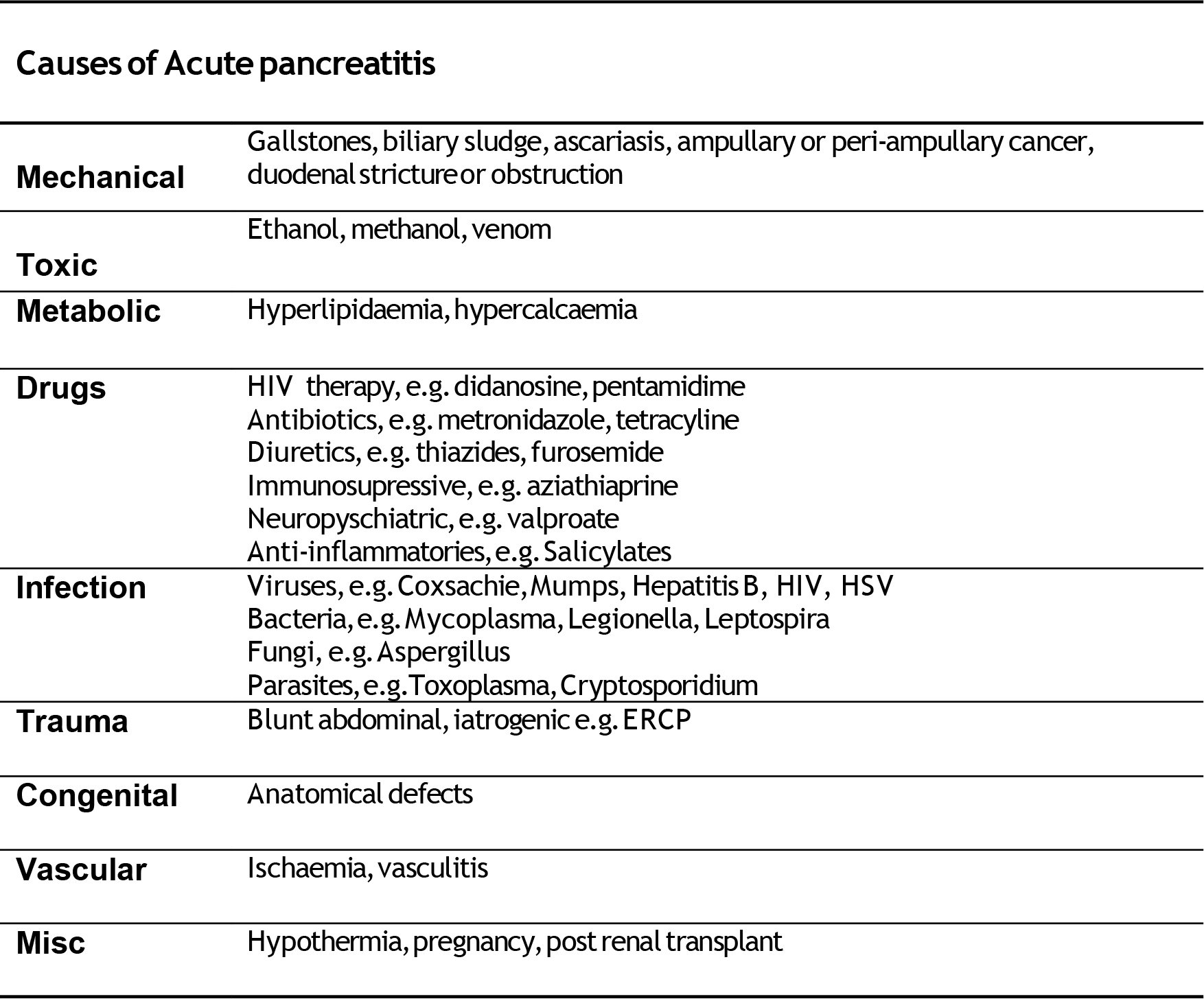
Table 1.
Clinical features
Acute pancreatitis is an important cause of acute upper abdominal pain. Because its clinical features are similar to a number of other acute illnesses, it is difficult to base a diagnosis only on symptoms and signs. The disease varies in severity and the diagnosis is often missed.
Demographics
- Acute gallstone pancreatitis is more common in women, acute alcoholic pancreatitis is more common in men
- Incidence increases with age
- In the first two decades indicates infection (mumps) or a hereditary cause such as hyperlipidaemia.
Symptoms
- a) Acute upper abdominal pain – epigastric, diffuse or right-sided
- b) Often preceded by some episodes of biliary colic after a meal
- c) Often starts two to three days after cessation of drinking alcohol heavily
- d) Onset can be quite rapid but not as rapid as a perforated viscus, and can last for days
- e) 50% patients have a band-like radiation to the back
- f) Associated with nausea and vomiting, agitation and restlessness
- g) Can be relieved by bending forward
- h) Can present as coma and shock
- i) Sufferers usually seek medical help.
Physical signs
- Varying clinical signs depend on disease severity
- From mild epigastric tenderness to distension, guarding and rebound. Often seems less impressive than degree of patients discomfort
- Discolouration in the flank (Grey-Turner’s sign) or the periumbilical region (Cullen’s sign) occurs in 1 % of cases but is not diagnostic. These signs reflect retroperitoneal haemorrhage and are associated with a poor prognosis.
- Obstruction of the common bile duct, due to gallstones or oedema of the head of the pancreas, can lead to jaundice
- An epigastric mass due to pseudocyst formation may become palpable in the course of the disease
- There may also be findings of underlying disorders such as hepatomegaly in alcoholic pancreatitis, xanthomas in hyperlipidemic pancreatitis, and parotid swelling associated with mumps
- Focal chest signs for pleural effusions.
Investigations and severity scoring systems
Acute pancreatitis can be suspected clinically, but requires biochemical and radiology tests to confirm the diagnosis. Clinical, biochemical and radiological features need to be considered together since none of them alone is diagnostic of acute pancreatitis.
Most attacks of acute pancreatitis are mild, with recovery occurring within five to seven days. Death is unusual in such patients. In contrast, severe necrotizing pancreatitis is associated with a high rate of complications and significant mortality. A subgroup of patients develop early severe acute pancreatitis, characterized by extended pancreatic necrosis with organ failure at admission. Identification of this group is vital.
Early detection of this high-risk group has led to the development of scoring systems to help identify those who will require a higher degree of intervention.
Biochemical tests
Pancreatic enzymes. Early in the acute phase of an attack, there is continued synthesis of pancreatic enzymes, but their exocrine secretion is obstructed. As a result these enzymes are released into the systemic circulation. Other organs also release these digestive enzymes, resulting in false positives tests.
Serum amylaserises within 6 –12 hours of an acute attack; and is rapidly cleared from the circulation. It is usually elevated for 3-5 days during an acute attack. It is also elevated in other conditions such as visceral perforation, so cannot be used alone in the diagnosis. It is also known that daily measurement of these enzymes is a poor predictor of clinical progress or prognosis. In severe cases, with marked necrosis production and serum levels of amylase may be normal.
Other enzymes are elevated but have no real advantage over amylase. They include serum lipase, urinary amylase.
Markers of immune activation/inflammation
A C-reactive protein level greater than 150 mg/dl at 48 hrs can discriminate severe from mild disease.
Radiological features
Abdominal plain X-ray
- Important as can exclude bowel obstruction or perforation
- Often unremarkable in mild disease
- Localised ileus in small bowel represents “sentinel loop” sign
- “Colon cut-off” in severe disease represents spasm of descending colon from localised pancreatic inflammation at splenic flexure.
- ‘Ground glass’ appearance may represent ascites.
Chest X-ray
- a. Look for elevated hemidiaphragm, pleural effusion, atelectasis or features of ARDS
- b. Bilateral pleural effusions are indicators of severe disease
Abdominal USS
- Excellent for the detection of gallstones and biliary tree obstruction
- Often limited by presence of overlying bowel gas
- Unable to accurately assess degree of pancreatitic inflammation
CT Scan
- Most important test for establishing diagnosis of pancreatitis, establishing severity and detecting complications.
- Patients should be scanned after 48 hrs, with oral and intravenous contrast. This enables an estimation of degree of unenhanced pancreatic necrosis.
Alcoholic versus gallstone pancreatitis
The differentiation between alcoholic and gallstone pancreatitis has important therapeutic implications, since elimination of the cause in either condition may prevent further attacks. Recurrent attacks of acute pancreatitis suggest an alcoholic aetiology, but patients who have unrecognized gallstones may have recurrent biliary colic, pancreatitis, or cholecystitis. Thus, removing the gallbladder in biliary pancreatitis is imperative. For this reason, abdominal ultrasonography should be performed in every patient with a first attack of acute pancreatitis, to search for gallstones in the gallbladder.
Severity scoring systems
A number of disease-specific scoring systems have been developed, but none are ideal.
Clinical assessment of severity
Clinical assessment looking for signs of shock, peritonitis and respiratory failure is just as effective, although this only detects approximately 40-50% of cases of severe acute pancreatitis. The two most popular scoring systems are the Ranson and Glasgow scales, which can only be completed 48 hours after onset of symptoms and can only be used once.
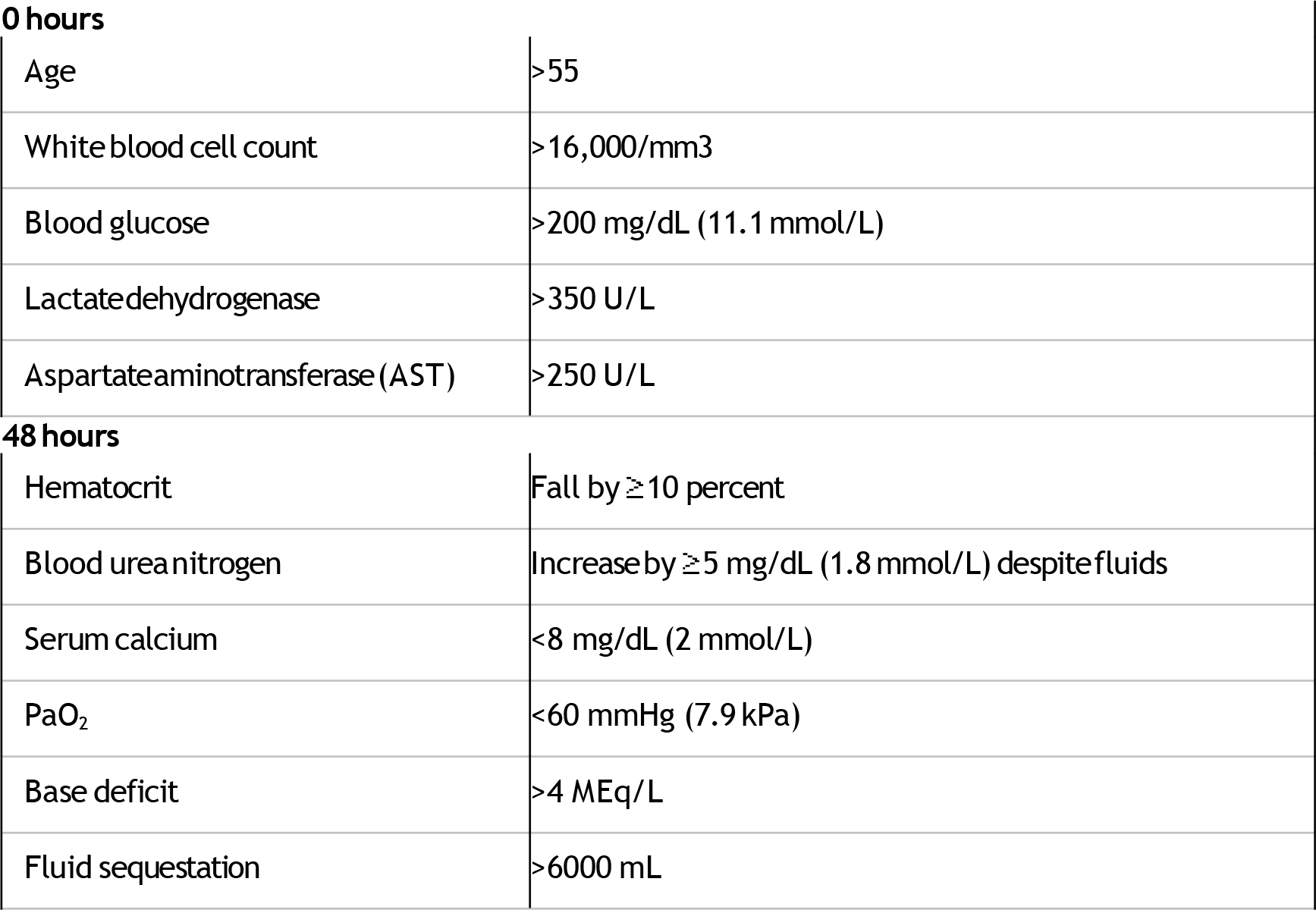
Table 2: Ranson criteria to predict severity of acute pancreatitis
The presence of 1 to 3 criteria represents mild pancreatitis; the mortality rate rises significantly with four or more criteria. Adapted from Ranson JHC, Rifkind KM, Roses DF,et al. Surg Gynecol Obstet 1974; 139: 69.
Poor prognostic factors in patients with acute pancreatitis
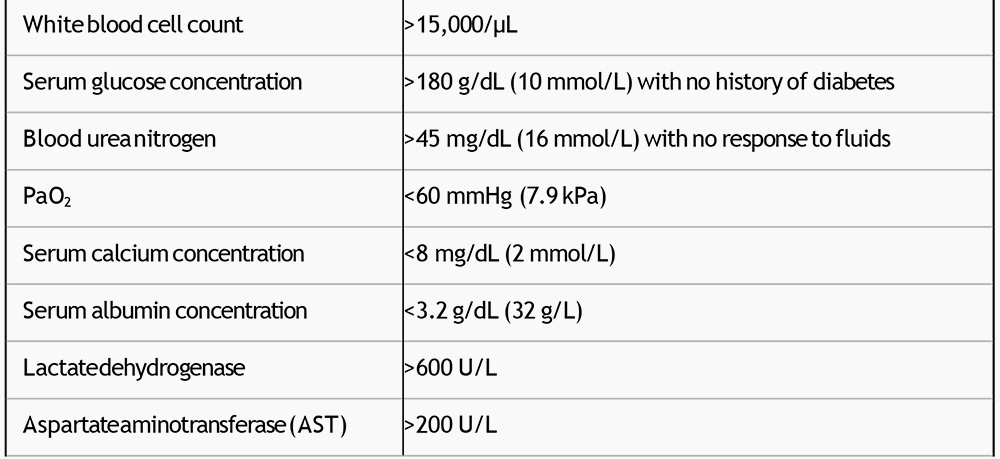
Table 3: Glasgow system to predict severity of acute pancreatitis
The presence of three or more of these criteria within the first 48 hours is indicative of severe pancreatitis. Adapted from: Corfield AP, Williamson RCN, McMahon MJ et al. Lancet 1985; 24: 403
CT Scan assessment of severity
Grading based upon findings on unenhanced CT

Necrosis score based upon contrast enhanced CT

Table 4: CT findings and grading of acute pancreatitis (CT severity index – CTSI)
CT severity index equals unenhanced CT score plus necrosis score: maximum = 10, 6 = severe disease. Adapted from Balthazar EJ, Robinson DL, Megibow,AJ, Ranson JH. Radiology 1990; 174: 331.
Clinical Management
The clinical management of severe acute pancreatitis (SAP) relies on a number of key points:
- Early recognition of severe disease, which has a higher mortality
- Fluid resuscitation and organ support with appropriate monitoring.
- Pain management.
- Removal of underlying predisposing cause e.g. gallstones.
- Prevention and early recognition of complications.
- Nutrition.
- Novel Therapies.
- Surgical intervention where indicated.
Many organisations have issued guidelines to simplify the management of SAP, most are very similar although some have different views on specific issues relating to the prevention if infection (see below).
• Early recognition of severe disease.
The main determinants of poor outcome are the degree of necrosis (may be limited by better fluid management) and the development of ongoing organ failure. Clinical assessment should focus on the early detection of organ dysfunction with frequent monitoring of vital signs. This should be directed at evaluation of intravascular volume status and the early detection of hypoxemia with pulse oximetry or blood gas analysis.
Although there are a number of disease-specific scoring systems (described in tables 2-4 above), frequent clinical assessment is the gold standard. Elevated serum biomarkers such as C-reactive protein do seem to be associated with disease severity or prognosis. However there is a delay of approximately 48 hrs, limiting its role as an early predictor.
Finally, appropriate radiological investigations are important. Ultrasound will determine whether gallstones are present or whether the stone has passed with the detection of a dilated biliary tree. CT scan with contrast will allow an estimation of the degree of pancreatic necrosis. A degree of necrosis greater than 30% is associated with a higher rate of complications and a worse outcome.
• Fluid resuscitation and organ support with appropriate monitoring.
SAP causes disruption of the pancreatic ducts. There is subsequent exudation of nonspecific serine proteases into the peritoneum and subsequently the plasma. This causes activation of the kinin, coagulation, and cytokine cascades, inducing a marked systemic pro-inflammatory response. The microcirculation is disrupted, exudation of intravascular fluids and inflammatory cells occurs, and severe hypovolemia follows. In the absence of adequate fluid resuscitation and cardiovascular optimisation this will result in tissue ischaemia and regional hypo-perfusion and end-organ damage.
Invasive monitoring of the arterial and central venous pressures to guide fluid resuscitation is considered mandatory, if available. Regular measurement of oxygen saturations and hourly urine volume may enable early detection of pulmonary and renal dysfunction. Fluid resuscitation is particularly important because patients with necrotizing pancreatitis may accumulate vast amounts of fluid in the injured pancreatic bed. Approximately 250 to 300 ml of intravenous fluids per hour are required for 48 hours if the patient’s cardiac status permits. Inadequate fluid can lead to a worsened degree of pancreatic necrosis. Direct myocardial depression can result from the disease process, occasionally necessitating the introduction of dobutamine to aid splanchnic perfusion, which is particularly susceptible to hypoperfusion in SAP, although there is no evidence of a mortality benefit.
The optimal resuscitation fluid for acute pancreatitis, crystalloid (salt) or colloid (protein- or starch-containing), isotonic or hypertonic, has not been clarified from existing trials.
• Effective pain control.
Severe abdominal pain is the main symptom and adequate analgesia is essential. This can be provided by systemic opioids (usually using patient controlled analgesia – PCA) or thoracic epidural. Traditionally there has been some concern over sphincter of Oddi spasm with morphine, but recently this has been shown to be clinically irrelevant. Either morphine or fentanyl delivered as a PCA or continuous infusion is acceptable
• Removal of underlying predisposing cause.
Patients with gallstone pancreatitis with obstructive jaundice or biliary sepsis should have ERCP (endoscopic retrograde cholangio-pancreatography) and an endoscopic papillotomy.
• Prevention and early detection of complications.
- Infection. The most serious complication is local pancreatic infection, occurring in approximately 30 % of patients with SAP. It is generally regarded as a late complication. The important organisms causing infection in necrotizing pancreatitis are predominantly gut-derived, including Escherichia coli, Pseudomonas, Klebsiella, and Enterococcus. The majority of infections (about 75 percent) are monomicrobial. Fungal and gram- positive organisms are uncommon, and have been associated with prolonged antibiotic use.It is not clear whether fungal infection has worse outcomes than gram negative infection.
- Common strategies to limit infection include:
- Enteral feeding, which limits line sepsis, maintains gut integrity and limit bacterial translocation.
- Selective decontamination (SDD) of the digestive tract with non-absorbable antibiotics
- Prophylactic systemic antibiotics
The role of systemic antibiotics remains controversial despite a large number of studies. Results have been inconclusive, probably due to the heterogeneity of the patient population, and due to poor tissue penetration of some of the antibiotics used in earlier trials. Current practice is to give intravenous antibiotics with good pancreatic penetration, such as meropenem, in the presence of organ failure or shock.
The duration of therapy tends to be 7 days. Clinicians should be aware of the possibility of a higher rate of infection with fungal or resistant organisms, with prolonged courses of systemic antibiotics.
• Nutrition.
Adequate nutrition is vital in the critically patient with severe disease. In SAP most patients have abdominal pain and ileus is common. There is a concern that enteral feeding may stimulate pancreatic secretion, therefore worsening auto-digestion. Studies are revealing that enteral feeding is safe and may reduce complications. Pancreatic enzyme release is inversely proportional to the distance from the pylorus, when patients are fed distal to the ligament of Treitz (connects the duodenum to the diaphragm at the junction of the 2nd and 3rd parts of duodenum). There is some evidence that jejunal feeding does not stimulate the pancreas at all.
The advantages of enteral feeding are the maintenance of gut integrity, reducing bacterial translocation and the avoidance of intravascular dwelling catheters.
Placement of naso-jejunal tubes can be difficult due to pancreatic swelling and obstruction, and may require endoscopic or fluoroscopic guidance.
A recent study has suggested the concern over gastric feeding causing pancreatic secretion is unjustified and nasogastric feeding may well be as safe and beneficial, if tolerated. [1]
• Novel medical therapies.
The protease inhibitor gabexate mesilate and the somatosatin analogue octreatide were both the subject of recent meta-analyses.[2] They both showed a modest benefit, but these therapies are unlikely to be regarded as cost effective.
In randomized studies, a number of other measures have been shown to be ineffective. These include nasogastric decompression, histamine H2-receptor antagonists, anticholinergics, glucagon, and plasma and peritoneal lavage.[3] A platelet activating factor antagonist, lexipafant, improved mortality in an initial study but not in a subsequent larger trial and is therefore not currently recommended.[4]
• Surgical or radiological drainage
If a patient remains unwell or clinically deteriorates, developing organ failure and signs of sepsis the patient must undergo CT scanning with contrast to exclude infection of the pancreatic bed or a significant collection. There may be another source of sepsis.
The former practice of early open debridement or necrosectomy is longer considered optimal. The current trend is for conservative management with repeated CT-guided microbiological sampling with subsequent antimicrobial therapy or attempted radiological drainage. Often these long-term patients have multiple abdominal drains.
Some patients, treated conservatively, may still require delayed surgical intervention for management of disconnected duct (disruption of pancreatic duct due to necrosis) which may lead to recurrent pancreatitis and/or fluid collections. Growing experience suggests that such an approach may be associated with a reduction in mortality.
Summary
Severe acute pancreatitis can be a devastating illness. The principles of management are mainly supportive therapy, however it is vital that the patient benefits from strict attention to detail in their daily critical care management. Rapid recognition and instigation of therapy for complications, such as infection, bleeding or obstruction can lead to good outcomes.[5-7]
References and further reading
- Eatock, F.C., et al., A randomized study of early nasogastric versus nasojejunal feeding in severe acute pancreatitis. Am J Gastroenterol, 2005. 100(2): p. 432-9.
- Andriulli, A., et al., Meta-analysis of somatostatin, octreotide and gabexate mesilate in the therapy of acute pancreatitis. Aliment Pharmacol Ther, 1998. 12(3): p. 237-45.
- Leach, S.D., F.S. Gorelick, and I.M. Modlin, New perspectives on acute pancreatitis. Scand J Gastroenterol Suppl, 1992. 192: p. 29-38.
- Kingsnorth, A.N., S.W. Galloway, and L.J. Formela, Randomized, doubleblind phase II trial of Lexipafant, a platelet-activating factor antagonist, in human acute pancreatitis. Br J Surg, 1995. 82(10): p. 1414-20.
- UK guidelines for the management of acute pancreatitis. Gut, 2005. 54 Suppl3: p. iii1-9.
- Wyncoll, D.L., The management of severe acute necrotising pancreatitis: an evidence-based review of the literature. Intensive Care Med, 1999. 25(2): p. 146-56.
- Nathens, A.B., et al., Management of the critically ill patient with severe acute pancreatitis. Crit Care Med, 2004. 32(12): p. 2524-36.



