Covid-19
KEY POINTS
- Acute respiratory distress syndrome is a syndrome of hypoxaemic respiratory failure associated with noncardiogenic pulmonary oedema which occurs in 10% of intensive care unit (ICU) patients.
- It is characterised by an acute inflammatory reaction followed by a proliferative stage. This may then progress to fibrosis or resolution.
- Principles of management aim to avoid exacerbating lung injury by ventilating at low tidal volumes with controlled pressure, appropriate use of positive end-expiratory pressure, and a permissive approach to hypercapnoea.
- Adjuncts include a conservative fluid management strategy and, in moderate to severe cases, neuromuscular blockade and prone-position ventilation.
- Extracorporeal membrane oxygenation can be used as a rescue therapy for severe cases.
INTRODUCTION
First described in 1967, acute respiratory distress syndrome (ARDS) is a syndrome of hypoxaemic respiratory failure associated with noncardiogenic pulmonary oedema. It is the result of diffuse inflammatory damage to the alveoli and pulmonary capillaries as a result of a local or systemic insult. ARDS is often associated with multiple organ dysfunction and carries a high mortality, morbidity, and financial cost.
DEFINITION
The previous definition was updated in 2012 to try and improve its reliability and predictive value. ARDS is now described by the ‘‘Berlin definition’’ as an ‘‘acute diffuse, inflammatory lung injury, leading to increased pulmonary vascular permeability, increased lung weight, and loss of aerated lung tissue…[with] hypoxemia and bilateral radiographic opacities, associated with increased venous admixture, increased physiological dead space, and decreased lung compliance.’’1
The following are the four key diagnostic criteria:
- Acute onset: within 7 days of a defined trigger (e.g. pneumonia).
- Degree of hypoxia: a PaO2:FiO2 ratio of <300 mm Hg (< 40 kPa) on at least 5 cm H2O of positive end-expiratory pressure (PEEP).
- Bilateral lung infiltrates seen on either chest x-ray or computed tomography scan.
- Not fully explained by cardiac failure or fluid overload.
ARDS is further subcategorised into mild, moderate, or severe according to P/F ratio (Table 1).

Table 1. Subcategories of Acute Respiratory Distress Syndrome
EPIDEMIOLOGY
The LUNG SAFE study2 was a prospective observational study of the epidemiology and management of ARDS encompassing 29 000 patients in over 50 countries. It found that ARDS criteria were fulfilled in 10% of all intensive care admissions and in 23% of cases requiring mechanical ventilation. ARDS was mild in 30%, moderate in 47%, and severe in 23% of cases. The median period of mechanical ventilation was 8 days with median ICU stay of 10 days. ICU and hospital mortality were 34% and 40% respectively. The study concluded that ARDS was underrecognised and undertreated with a high mortality.
AETIOLOGY
There are a number of potential precipitating conditions and these can be classified as pulmonary or extrapulmonary (Table 2).
PATHOPHYSIOLOGY
ARDS is characterised by an acute inflammatory response with release of cytokines such as tumor necrosis factor ∝, IL-1, and IL-6. These stimulate neutrophil migration to the alveoli. Here, they become activated and release reactive oxygen species, resulting in free radical production that damages endothelial cells and pneumocytes. This damage to the alveolar and capillary endothelium leads to an accumulation of protein-rich inflammatory exudate in the alveoli and interstitium (‘‘noncardiogenic pulmonary oedema’’). The same plasma proteins denature alveolar surfactant, causing alveolar collapse. The collapsed or fluid-filled alveoli cause a shunt leading to hypoxia.
Following this initial acute inflammation there is a proliferative phase (from approximately 72 hours to 7 days) characterised by proliferation of type 2 pneumocytes, fibroblasts, and myofibroblasts with deposition of new matrix. This is followed by either a fibrotic stage or resolution and reconstitution of normal pulmonary parenchyma. Interstitial and intra-alveolar fibrosis results from excessive disordered deposition of collagen and is associated with a poor outcome, but the factors determining the progression to fibrosis versus resolution are not well understood.3
MANAGEMENT: GENERAL PRINCIPLES
ARDS is one of the most studied areas in intensive care and thus there is a relatively strong evidence base to draw upon. Two recent major guidelines in this area are the American Thoracic Society/European Society of Intensive Care Medicine guidelines (2017) and the (UK) Intensive Care Society’s guidelines on the management of ARDS (2018).4,5
The principles of management, collectively known as lung-protective ventilation, aim to minimise associated lung injury by ventilating at low tidal volumes with controlled pressure, appropriate use of PEEP, rationalised FiO2, and a permissive approach to hypercapnoea. Adjuncts include a conservative fluid-management strategy plus neuromuscular blockade and

Table 2. Potential Precipitating Conditions for Acute Respiratory Distress Syndrome. aPercentages are from the LUNG SAFE study.2
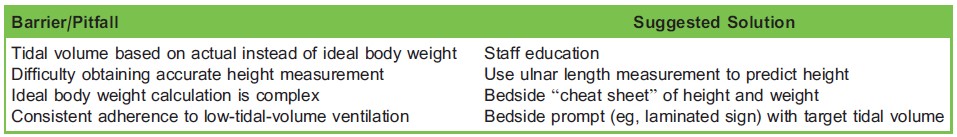
Table 3. Commn Barriers to Low-Tidal-Volume Ventilation
ventilation in the prone position where indicated. These basic principles require minimal specialised equipment and should be achievable in any ICU. Despite this, and the fact that much of the evidence is almost 20 years old, the LUNG SAFE study found ARDS to be underdiagnosed (recognised in only 60% of cases) with poor adherence to best practice.
VENTILATORY STRATEGIES
Low-Tidal-Volume Ventilation
The cornerstone of ARDS management is a strategy of low-tidal-volume ventilation pioneered by the ARDSnet trial in 2000.6 This involves the following:
- Tidal volume 6 ml/kg based on ideal body weight
- Plateau pressure <30 cm H2O
- Permissive hypercapnoea tolerated as long as pH >7.2
The lower tidal volumes were strongly associated with a decrease in mortality and an increase in ventilator-free days. This was thought to be as a result of reducing excessive distension or stretch of the aerated lung, which could be associated with the release of proinflammatory cytokines that could contribute to lung (volutrauma) and other organ injury.
Common barriers to successful implementation and suggested solutions are discussed in Table 3.
Ideal body weight in kilograms can be calculated from height:
Males = 50 + 0.91(height [cm] – 152.4)
Females = 45.5 + 0.91(height [cm] – 152.4)
Positive End-Expiratory Pressure
PEEP prevents alveolar derecruitment, minimising atelectrauma. It increases functional residual capacity, displaces fluid from the alveoli into the interstitium, and moves the lung to the steeper part of the compliance curve. However, this has to be balanced against potential hyperinflation and decreased venous return, and thus cardiac output.
PEEP is patient-specific and determining the ‘‘optimal’’ PEEP for each patient is challenging. A number of different strategies (none of which have proven superiority) have been suggested to set PEEP:
- Use of PEEP/FiO2 tables
- Titrating based on plateau pressure
- Stepwise increase until maximal compliance
- Recruitment manoeuvre followed by decremental titration
- Based upon the inflection point of the pressure/volume curve
Guidelines recommend use of high PEEP (defined variably, but on average 15 cm H2O) on patients with moderate and severe ARDS, in whom a mortality benefit was demonstrated in clinical trials.
Prone-Position Ventilation
Although this has long been known to improve oxygenation, evidence of mortality benefit has been lacking until recently. There are a number of postulated physiological benefits: improved recruitment of atelectatic dorsal lung tissue leading to its increased compliance of lungs (total compliance may actually decrease), more even distribution of tidal volume, and improved V/Q (V= pulmonary ventilation; Q= pulmonary perfusion) matching. Prone ventilation also decreases alveolar hyperinflation, reduces cardiac compression of the underlying lung, and improves drainage of secretions. There is experimental evidence suggesting it may influence the generation and evolution of ventilator-induced lung injury.7
In recent years, evidence (including the landmark ‘‘Proning severe ARDS patients’’ trial) has shown a mortality benefit from ventilating in the prone position when used for >12 hours each day in the subgroup of severe ARDS patients only.8 This must be carried out with caution and appropriate training as there is a proven increased risk of both endotracheal tube obstruction and pressure sores.
Airway Pressure Release Ventilation
Airway pressure release ventilation (APRV), sometimes known as ‘‘inverse ratio ventilation’’ is a ventilatory mode in which the majority of the respiratory cycle (usually around 90%) is spent at a high pressure with a brief drop to lower pressure to allow expiration to occur. Crucially, the time spent at low pressure is set to only allow the expiratory flow rate to decrease to 70% to 75% of its peak value, preventing alveolar derecruitment. Spontaneous breathing without any additional support is encouraged.
The rationale for APRV is that the higher mean airway pressure opens the alveoli and then holds them open, improving oxygenation and decreasing lung injury. This is known as the ‘‘open lung’’ concept and has some support from small trials and animal studies. The evidence for APRV in ARDS is currently weak with only 1 single-centre randomised control trial (RCT), which showed improved gas exchange and more ventilator-free days in the APRV group.9 This minimal evidence base means neither the American Thoracic Society nor the Intensive Care Society guidelines offered an opinion on the
subject.
High-Frequency Oscillatory Ventilation
High-frequency oscillatory ventilation is a ventilatory mode, more commonly used in paediatrics, where the lung is held open at a high mean airway pressure (typically 25-30 cm H2O) and small tidal volumes of 1 to 3 ml/kg are delivered at a frequency between 3 and 15 Hz. Two trials published in 2013 have led to high-frequency oscillatory ventilation falling out of favour. OSCAR (High Frequency OSCillation in ARDS) showed no difference in mortality between high-frequency oscillatory ventilation and control; OSCILLATE (High-Frequency Oscillation in Early Acute Respiratory Distress Syndrome) demonstrated increased mortality with oscillation.10,11 As such, guidelines recommend strongly against its use in the management of ARDS.
Recruitment Manoeuvres
Recruitment manoeuvres aim to use a transient increase in ventilatory pressure to open up (‘‘recruit’’) collapsed alveoli, reversing the atelectasis seen in ARDS and increasing the lung area available for gas exchange. Following the recruitment manoeuvre, PEEP is generally increased to hold open the newly recruited alveoli. There are many proposed methods:
- Sustained inflation (eg, continuous positive airway pressure at 30-40 cm H2O for 30-40 seconds)
- Stepwise increase in peak pressure and/or PEEP
- ‘‘Extended sigh’’ in which PEEP is incrementally increased and tidal volume incrementally decreased aiming to keep the plateau pressure constant. This is followed by a sustained inflation at continuous positive airway pressure of 30 cm H2O and the sequence is carried out in reverse to return to baseline.
There is no doubt that recruitment manoeuvres improve oxygenation and compliance at least temporarily. However they can cause haemodynamic instability and barotrauma and the physiological benefit may not be sustained. The American Thoracic Society guidelines give a weak conditional recommendation for the use of recruitment manoeuvres. However, since their publication in 2017, the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial showed increased mortality with their strategy of recruitment manoeuvre and PEEP titration.2 The place of the recruitment manoeuvre is thus currently unclear.
Extracorporeal Membrane Oxygenation
Extracorporeal membrane oxygenation (ECMO) describes the use of large-bore central venous access to remove blood, pass it through a gas exchanger, and return it to the (usually venous) circulation. It is an invasive, expensive, and complex technique, which is a highly effective method of oxygenation but associated with a number of complications relating to vascular access, anticoagulation, and serious bleeding. As a specialist therapy its use is limited to respiratory failure centres.
The CESAR (Conventional ventilatory support vs Extracorporeal membrane oxygenation for Severe Adult Respiratory failure) trial (2009) showed a mortality benefit in those assigned to the ECMO treatment group but was criticised for being a trial of good conventional treatment at a respiratory failure centre rather than a trial of ECMO specifically, as 24% of patients assigned to the ECMO group did not receive it.13 More recently the EOLIA (ECMO to Rescue Lung Injury in Severe ARDS) trial (2018) of ECMO for severe ARDS showed an 11% reduction in mortality in the treatment group, which failed to reach statistical significance.14 The trial was underpowered and complicated by a high rate of crossover from control to ECMO. The present role of ECMO is as a rescue therapy in those with severe ARDS only.
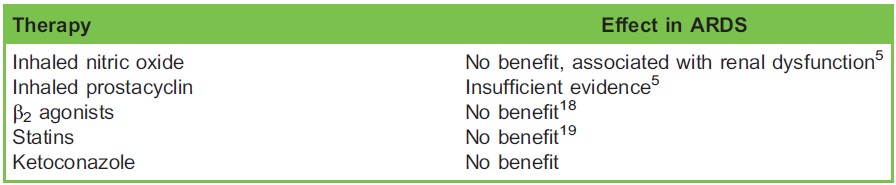
Table 4. Effectiveness of Adjunct Therapies for Acute Respiratory Distress Syndrome (ARDS)
NONVENTILATORY STRATEGIES
Conservative Fluid Therapy
Given that ARDS is a pathological state of noncardiogenic pulmonary oedema, conservative fluid management appears intuitive. The FACTT (Fluid And Catheters Treatment Trial) study compared conservative and liberal fluid strategies and found no difference in mortality but a significant improvement in ventilator-free days and ICU-free days in favour of the conservative arm.15 Despite the lack of strong evidence, guidelines give a weak recommendation for conservative fluid management, which did not appear to be associated with an increase in acute kidney injury.
Neuromuscular Blockade
Neuromuscular blocking agents have the potential to improve patient-ventilator synchrony by eliminating spontaneous respiratory effort and coughing. This may facilitate compliance with lung-protective ventilation protocols and reduce barotrauma. Their use has been shown to improve oxygenation as measured by PaO2:FiO2 ratio, which, in addition to better control of ventilation, may be in part due to a decrease in oxygen consumption. Paralysis clearly necessitates a deep level of sedation, which may cause additional problems. There are also concerns over their role in causing increased ICU-acquired weakness.
The Intensive Care Society guideline concludes that there is weak evidence from systematic reviews to support the use of 48 hours of a cisatracurium infusion in patients with moderate to severe ARDS. All trials to date have been of cisatracurium, which may have a direct anti-inflammatory action in addition to its neuromuscular effect. It is unclear whether these findings can be generalised to other neuromuscular blockers. An observational study comparing cisatracurium with vecuronium found no difference in mortality but a significant reduction in ventilator days in favour of cisatracurium.16
Corticosteroids
Given the known inflammatory component of ARDS, there would appear to be a potential role for steroids to suppress this phase. There are a number of small studies in this area, many of which were of low quality and were conducted before the advent of lung-protective ventilation. Methylprednisolone 1 mg/kg/day is recommended with moderate certainty for moderate to severe ARDS by the Society of Critical Care Medicine and European Society of Intensive Care Medicine’s corticosteroid guideline.17 Steroids may potentially increase the risk of hospital-acquired infections, neuromyopathy, and delirium but, due to the low quality of the existing evidence, the FICM (Faculty of Intensive Care Medicine)/Intensive Care Society document makes a recommendation for further research only. Suffice to say the role of steroids remains controversial. There is currently limited evidence in this area and further trials are ongoing.
Other Adjunctive Therapies
A number of pharmaceutical therapies have been trialled in various attempts to improve V/Q matching, reduce ventilator-induced lung injury, and modulate the inflammatory process underlying ARDS. The evidence is summarised in Table 4.
SUMMARY
ARDS is a syndrome of hypoxaemic respiratory failure associated with noncardiogenic pulmonary oedema that occurs in 10% of ICU patients. Principles of management aim to avoid exacerbating lung injury by ventilating at low tidal volumes with controlled pressure, appropriate use of PEEP, and a permissive approach to hypercapnoea. Adjuncts include a conservative fluid management strategy and, in moderate to severe cases, prone-position ventilation and neuromuscular blockade; ECMO can be used as a rescue therapy for severe cases.
REFERENCES
- The ARDS Definition Task Force. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307(23):2526- 2533.
- Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315(8):788-800.
- Pierrakos C, Karanikolas M, Scolletta S, et al. Acute respiratory distress syndrome: pathophysiology and therapeutic options. J Clin Med Res. 2012;4(1):7-16.
- Fan E, Del Sorbo L, Goligher EC, et al. An official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine clinical practice guideline: mechanical ventilation in adult patients with acute respiratory distress syndrome. AmJ Respir Crit Care Med. 2017;195(9):1253-1263.
- Guidelines on the management of acute respiratory distress syndrome. The Faculty of Intensive Care Medicine / Intensive Care Society. https://www.ficm.ac.uk/sites/default/files/ficm_ics_ards_guideline_-_july_2018.pdf (accessed on 1/9/19)
- Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. New Engl J Med. 2000;342(18):1301-1308.
- Taccone P, Carlesso E, Marini J. Prone position in acute respiratory distress syndrome. Rationale, indications, and limits. Am J Respir Crit Care Med. 2013;188(11):1286-1293.
- Gue´ rin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome. New Engl J Med. 2013;368(23):2159-2168.
- Zhou Y, Jin X, Lv Y, et al. Early application of airway pressure release ventilation may reduce the duration of mechanical ventilation in acute respiratory distress syndrome. Intensive Care Med. 2017;43(11):1648-1659.
- Young D, Young D, Lamb S, Lamb S, Shah S Shah S et al. High-Frequency Oscillation for Acute Respiratory Distress Syndrome. N Engl J Med 2013; 368:806–813
- Ferguson ND, Ferguson ND, Cook DJ, Cook DJ, Guyatt GH Guyatt GH et al. High-Frequency Oscillation in Early Acute Respiratory Distress Syndrome. N Engl J Med 2013; 368:795–805
- Cavalcanti AB, Suzumura E´ A, Laranjeira LN, et al. Effect of lung recruitment and titrated positive end-expiratory pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2017;318(14):1335-1345.
- Peek GJ, Mugford M, Tiruvoipati R, et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374(9698):1351-1363.
- Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. New Engl J Med. 2018;378(21):1965-1975.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network. Comparison of two fluid-management strategies in acute lung injury. New Engl J Med. 2006;354(24):2564-2575.
- Sottile PD, Kiser TH, Burnham EL, et al. An observational study of the efficacy of cisatracurium compared with vecuronium in patients with or at risk for acute respiratory distress syndrome. Am J Respir Crit Care Med. 2018;197(7):897-904.
- Annane D, Pastores SM, Rochwerg B, et al. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (Part I): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Intensive Care Med. 2017;43(12):1751-1763.
- Gates S, Perkins G, Lamb SE, et al. Beta-agonist lung injury trial-2 (BALTI-2): a multicentre, randomised, double blind, placebo-controlled trial and economic evaluation of intravenous infusion of salbutamol versus placebo in patients with acute respiratory distress syndrome. Health Technol Assess. 2013;17:v-vi,1-87.
- McAuley DF, Laffey JG, O’Kane CM, et al. Simvastatin in the acute respiratory distress syndrome. New Engl J Med. 2014;371(18):1695-1703.



