Basic Sciences
KEY POINTS
- Regional anaesthesia has shown many benefits, including improved patient satisfaction, reduced incidence of cardiac and pulmonarycomplications, shorter hospital stay, better pain management, lower healthcare costs, reduced opioid consumption, and reduced perioperative morbidity and mortality.
- Adjuvant medications can be combined with local anaesthetics given perineurally to prolong their clinical effect and improve postoperative analgesia.
- Perineural administration of agents such as epinephrine, buprenorphine, clonidine, magnesium, and, more recently, dexamethasone and dexmedetomidine, have shown some ability to improve or prolong analgesia.
- Unfortunately, perineural administration of adjuvant medications can result in undesirable side effects.
- Current meta-analyses on the effectiveness and safety of adjuvant agents are based on highly heterogeneous studies with the possibility of publication bias. High-quality randomized controlled trials are needed.
INTRODUCTION
Perineural injection of local anaesthetics (LAs) provides analgesia or anaesthesia through the reversible blockade of sodium channels resulting in inhibition of nociceptive conduction.1 There are many benefits to regional anaesthesia, including improved patient satisfaction, reduced incidence of cardiac and pulmonary complications, shorter hospital stay, better pain management, lower healthcare costs, reduced opioid consumption, and reduced perioperative morbidity and mortality.1 In some patients, postoperative pain can persist beyond the time required for the injured tissue to heal, resulting in the development of chronic postsurgical pain. This is estimated to affect ~10% of all surgical patients resulting in a significant cost burden. Chronic postsurgical pain is typically defined as postsurgical pain lasting greater than 3 to 6 months following surgery.2 One strategy to reduce this risk is effective analgesia to treat acute perioperative pain, including using regional anaesthesia blocks.2
The maximum duration from a single-shot peripheral nerve block depends mainly on the location or site of injection, and the type, volume, and concentration of LA used. Analgesia can be prolonged with (1) insertion of perineural catheters for continuous or intermittent infusion of LA, (2) use of systemic opioids, and/or (3) adjuvant medications to prolong the LA’s clinical effect. All such methods, however, present limitations. For instance, problems associated with perineural catheters include early and/or inadvertent dislodgement, need for skilled personnel for adequate and safe insertion, need for additional equipment (eg, ultrasound, infusion pump), and increased cost. Common side effects associated with opioid therapy include nausea, vomiting, constipation, urinary retention, respiratory depression, pruritus, and, potentially, hyperalgesia.1,2 This tutorial evaluates the current evidence on perineural (compared with systemic) adjuvant medications in regional anaesthesia to improve and prolong analgesia. This tutorial will not discuss the use of adjuvant medications in intrathecal or epidural blocks.
ADJUVANT MEDICATIONS
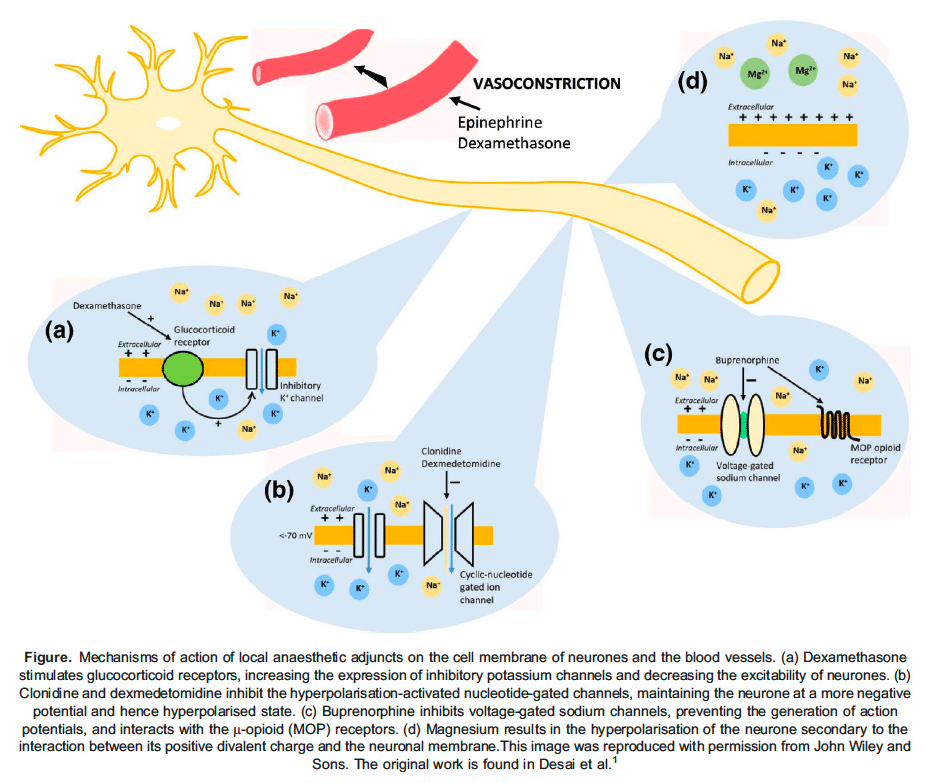
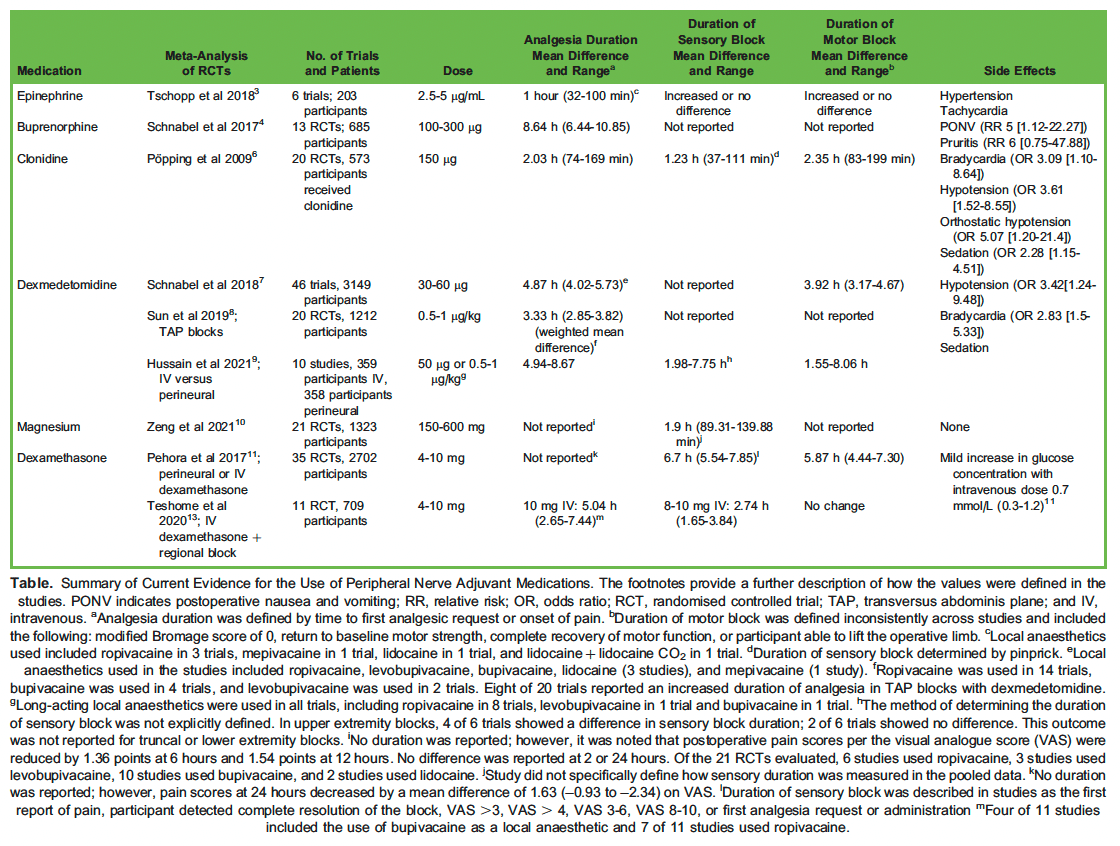
The following section will describe the role of adjuvant medications used and studied in peripheral nerve blocks. Each section will refer to the Figure to illustrate the proposed mechanism of action for the described adjuvant medications in this tutorial. As well, the dose, side effects, and effect on sensory and motor block for each of the adjuvant medications are summarized in the Table.
Epinephrine
Epinephrine was one of the first adjuvant medications used in peripheral nerve blockade. Around the nerve, epinephrine causes localized vasoconstriction through the activation of α 1 receptors (Figure).1 As a result, the LA remains close to the nerve for a longer period, extending its clinical effect. Vasoconstriction of afferent and efferent blood flow to the nerve decreases the risk of local anaesthetic systemic toxicity.3 Most commonly, a dose of 2.5 to 5 μg of epinephrine is added for each millilitre of LA to achieve the desired vasoconstriction. Epinephrine has been shown to increase analgesic duration when combined with lidocaine and mepivacaine (Table).3 Notably, epinephrine does not increase block duration when added to ropivacaine.3 This is likely due to ropivacaine’s intrinsic vasoconstricting properties.3 Tschopp et al3 did not specifically evaluate the combination of epinephrine with bupivacaine or other long-acting LAs; however, with only modest increases in analgesic duration with short- acting agents and no increase with (long-acting) ropivacaine, one may speculate that the effects would likely be minimal.
Epinephrine is also a useful adjuvant medication to detect intravascular injection of LA. Indeed, epinephrine has often been advocated as anadditional safety measure for regional anaesthesia practitioners resulting in tachycardia and hypertension in intravascular injection, potentially allowing clinicians to detect intravascular injection before the onset of local anaesthetic systemic toxicity symptoms.1 Notably, the advent of ultrasound-guided blocks has enhanced our ability to deposit LAs around the nerve safely; however, it does not completely eliminate the risk of intravascular injection. Consequently, using epinephrine to detect inadvertent intravascular injection remains clinically relevant.
Perineural epinephrine may be associated with local neurotoxic and myotoxic effects due to vasoconstriction.3 This should be considered when administering to patients at a higher risk of neurovascular injuries, such as those with pre-existing (especially long-standing or uncontrolled) diabetes and peripheral vascular disease.1 In addition, should an inadvertent intravascular injection occur, the resulting hypertension and tachycardia may lead to increased myocardial oxygen demand.
Buprenorphine
Buprenorphine is a synthetic partial opioid agonist at the μ and μ opioid receptors and a full opioid-receptor-ligand-1 agonist.4 Buprenorphineacts in a similar way to LAs because it is a potent inhibitor of the α subunits of voltage-gated sodium channels and likely contributes to analgesia by interacting with μ-opioid receptors on peripheral sensory axons (Figure).4 Other medications, including ketamine, clonidine, and other opioids, inhibit α subunits of sodium channels to varying degrees; however, current literature suggests that buprenorphine has the most significant effect.4 A meta-analysis of 13 randomized controlled trials (RCTs) evaluating the efficacy and safety of buprenorphine (2-3 μg/kg or 100-300 μg) in upper and lower extremity blocks showed a significant increase in the duration of analgesia measured by time to first analgesic request or visual analogue score (Table).4 The most common LAs (combined with buprenorphine) included in this review were bupivacaine and ropivacaine. Furthermore, the duration of analgesia was similarly prolonged when comparing perineural versus intramuscular buprenorphine.4
Side effects include postoperative nausea and vomiting (PONV) and pruritis. The incidence of PONV was 5-fold higher in patients receiving buprenorphine without antiemetic prophylaxis.4 The most common adverse reaction was pruritis, although these results were deemed very low to low in quality due to possible publication bias.4 In vitro experiments have shown that buprenorphine did not influence ropivacaine-induced neurotoxicity at clinical concentrations.5
Clonidine
Clonidine is a partial agonist of the α2 receptors found in abundance in the dorsal horn of the spinal cord and locus coeruleus.1 Injection of clonidine around sensory nerves most likely causes hyperpolarization of A and C fibres.1 Clonidine binds with activated nucleotide-gated channels, inhibiting the neurone from returning to a resting state and preventing further action potential generation (Figure).1 The use of clonidine has been studied extensively in neuraxial and peripheral nerve blockades. A 2009 meta-analysis of 20 RCTs evaluating the effects of perineural clonidine (30-300 μg, most commonly 150 μg) in upper and lower extremity peripheral nerve blocks or plexus blocks showed that sensory and motor blocks were prolonged by approximately 1.5 to 2 hours.6 The analgesic benefits of perineural clonidine largely depended on the nature of the LA used. For example, when combined with an intermediate-acting LA (mepivacaine, prilocaine, lidocaine), the duration of analgesia increased by an average of 200 minutes, whereas, when combined with a long-acting LA (ropivacaine, bupivacaine, levobupivacaine) the duration increased by 152 minutes.6 Two studies compared the perineural administration of clonidine with LA versus either subcutaneous clonidine or intramuscular clonidine.6 The first study determined the duration of analgesia was prolonged with perineural compared with subcutaneous clonidine in brachial plexus blocks. In contrast, the second found no analgesic difference when clonidine was administered perineurally versus intramuscularly in sciatic or femoral nerve blocks.6 Based on the limited number of studies and the apparent lack of analgesic difference in perineural versus intramuscular injection of clonidine, it is difficult to determine whether the analgesic benefit of clonidine is the result of systemic reabsorption versus perineural interaction. More studies are warranted.
Clonidine was also associated with a higher risk of hypotension, bradycardia, sedation, and orthostatic hypotension or fainting (Table).6 The authors of this meta-analysis could not find dose responsiveness for harm or benefit because most patients included in the RCTs received 150 μg.6 This marginal block improvement and high incidence of side effects have limited clonidine’s use in peripheral nerve blocks.
In terms of side effects in vitro, prolonged (24-hour) exposure of sensory neurons to higher than clinically used clonidine concentrations did not significantly affect cell death, and clonidine was significantly less neurotoxic than ropivacaine alone.5 When used at clinical concentrations and in combination with buprenorphine, there was no influence on ropivacaine-induced neurotoxicity.5
Dexmedetomidine
Dexmedetomidine is a selective α2-adrenoreceptor agonist that has recently gained significant popularity in anaesthesia due to its sedative, analgesic, and anxiolytic properties and is gaining attraction as a perineural adjunct to regional anaesthesia.1 Dexmedetomidine has a selectivity of 1620:1 for the α2 receptor compared with the α1 receptor.1 Similar to clonidine, dexmedetomidine binds with activated nucleotide-gated channels, inhibits the neurone from returning to a resting state, and prevents further action potential generation (Figure).1 Multiple meta-analyses evaluating the use of perineural dexmedetomidine have concluded that doses ranging from 25 to 100μg in combination with LA result in prolonged analgesia or sensory block and motor block by approximately 4.5 hours and 4 hours, respectively (Table).7 The analgesic effect of perineural dexmedetomidine appeared to be greatest in supraclavicular, interscalene, and femoral nerve blocks; however, it also significantly lengthened analgesic duration in axillary, infraclavicular, transversus abdominis plane (TAP), and eye blocks compared with LA alone.7 With moderate quality of evidence, the mean duration of analgesia of perineural dexmedetomidine combined with LA in peripheral nerve blocks is 4.87 hours.7 In truncal blocks, including TAP blocks, the addition of dexmedetomidine (0.5-1 μg/kg) appears to improve postoperative pain, extend block duration, and reduce opioid consumption in the first 24 hours compared with controls.8 Hussain et al9performed a systematic review evaluating the duration of sensory and motor block characteristics in perineural versus systemic dexmedetomidine (combined with LA) in peripheral nerve blocks. The duration of sensory blockade increased with perineural compared with systemic administration in 4 out of 6 studies in upper extremity blocks with a duration ranging from 1.98 hours (50 μg dose) to 7.75 hours (1 μg/kgdose).9 Similarly, 5 out of 7 studies reported prolonged motor block with perineural compared with systemic dexmedetomidine ranging from 1.55hours (0.75 μg/kg dose) to 8.06 hours (1 μg/kg dose).9 This systemic review suggested the inferiority of intravenous compared with perineural administration when dexmedetomidine is used as a peripheral nerve adjunct.
The perineural administration of dexmedetomidine is associated with an approximately 3-fold increase in the risk of hypotension and bradycardia.7 In the systematic review by Hussain et al9 comparing perineural with intravenous dexmedetomidine in regional blocks, there was no difference in the incidence of intraoperative bradycardia. Interestingly, in 1 out of 6 studies reporting adverse events, Hussain et al9found the incidence of intraoperative hypotension was higher in patients receiving intravenous compared with perineural dexmedetomidine. Despite evidence favouring the perineural administration of dexmedetomidine for prolonged analgesic benefit, there should be consideration of its motor block–prolonging effects because this has the potential to delay ambulation and discharge following outpatient surgery.
Multiple studies have examined the risk of neurotoxicity in perineurally administered dexmedetomidine. Schnabel et al,7 for example, summarized that the injection of rat sciatic nerves with dexmedetomidine in combination with either ropivacaine or bupivacaine did not show significant changes within the nerve compared with local LAs alone. Furthermore, adding dexmedetomidine to LAs might have a protective effect on reducing the inflammatory effects of LA.7
Magnesium
Magnesium is an N-methyl-D-aspartate receptor antagonist evaluated extensively in peripheral nerve blocks. There are several theories as to how it exerts its effects. Magnesium’s cationic charge may neutralize the negative charge on nerve membranes.1,10 This neutralization affects sodium channel gating, thus causing hyperpolarization of the cell surface and raising the cell’s threshold for generating an action potential (Figure).1,10 Magnesium may also act via the nitric oxide pathway, which may be involved in its anti-hyperalgesia effect.1,10
In a recent meta-analysis of 21 RCTs including upper extremity, lower extremity, and truncal blocks, the addition of magnesium resulted in a prolonged sensory block and reduced pain scores at 6 and 12 hours postoperatively in brachial plexus, TAP, sciatic, and femoral nerve blocks (Table).10 The doses used in the RCTs ranged from 150 to 600 mg of magnesium combined with either lidocaine, bupivacaine, ropivacaine, or levobupivacaine and administered perineurally.10
Interestingly, the addition of magnesium in TAP blocks was associated with a reduction in PONV compared with LA alone.10 There were no side effects or adverse reactions reported in the meta-analysis when magnesium was added to LAs for peripheral nerve blocks. Trials are lacking in evaluating magnesium neurotoxicity, and its perineural safety is not well established.1
Dexamethasone
Dexamethasone is a potent glucocorticoid medication commonly used as an adjuvant to reduce postoperative pain.11 The mechanisms of perineurally administered dexamethasone are likely attributed to complex interactions, including direct inhibition of signal transmission in nociceptive C fibres, local vasoconstriction, and reduced local inflammation.1 Dexamethasone may bind with glucocorticoid receptors on sensory axons, resulting in an increased expression of inhibitory potassium channels and reduced neuronal excitability (Figure).11
There is an ongoing debate on the superiority of intravenous versus perineural administration of dexamethasone for pain management. De Oliveira et al12 showed that intravenous administration of 0.1 to 0.2 mg/kg dexamethasone reduced postoperative pain and opioid consumption without significant side effects. Similarly, a meta-analysis evaluating pain control with intravenous dexamethasone in combination with peripheral nerve block found that the analgesic duration improved in patients receiving 8 to 10 mg of dexamethasone.13 In a Cochrane review, Pehora et al11 suggested that either intravenous or perineural dexamethasone can improve pain scores postoperatively, with perineural administration resulting in a significantly prolonged sensory block compared with placebo (Table). Furthermore, when comparing intravenous versus perineural administration, the latter showed a more extended sensory block without a significant difference in pain scores or opioid consumption within the first 24 hours.11 Many authors advocate for the use of intravenous dexamethasone at doses of 0.1 to 0.2 mg/kg in patients undergoing moderately to severely painful surgical procedures because this mode of administration has consistently provided evidence for better pain control and lower rates of PONV.1,11-13
In vitro studies examining the effects of perineural dexamethasone at doses of 0.15 mg/kg on rat sciatic nerve blocks have shown no significant effect on perineural inflammation or neuronal toxicity after a single dose of perineural dexamethasone.14 Single doses of intravenous dexamethasone in surgical patients have not been associated with increased surgical site infections; however, it may result in a mild increase in serum glucose concentrations.15 Consideration for its use in patients with poorly controlled diabetes is warranted because it has the potential to increase maximal perioperative serum glucose concentrations, mainly when using higher doses of dexamethasone.15 Desai et al1 have summarized multiple studies evaluating the risk of neurotoxicity with perineural dexamethasone. Most data suggest that dexamethasone is not associated with neurotoxicity when used in regional anaesthesia. Preservative-free dexamethasone is necessary when used perineurally because additives to dexamethasone have been known to cause neurotoxic effects.1 Desai et al1 concluded that the lack of evidence for harm in adult and animal studies does not mean that risk is absent; thus, clinicians should consider this when using adjuvant medications for perineural blockade.
Other Adjunct Medications
Many other adjunct medications have been tested in peripheral regional anaesthesia for their potential to prolong analgesia. The perineural use of fentanyl and morphine has shown conflicting results for effectiveness without any practical benefit over intravenous administration.1,4 Furthermore, it is difficult to ascertain if the effects of perineural opioids, other than buprenorphine, result from activation of peripheral opioid receptors or are simply due to redistribution.1,4 Other medications including tramadol, nonsteroidal anti-inflammatory drugs, ketamine, and neostigmine have also been used as adjuvants in peripheral nerve blockade; however, evidence for their use is conflicting and has not consistently shown benefit for block improvement.1 Furthermore, ketamine and neostigmine are associated with adverse reactions such as nausea, vomiting, and possibly neurotoxicity.1 Finally, midazolam is a benzodiazepine that may peripherally interact with translocator proteins.1 Despite studies evaluating its intrathecal use, the effectiveness of perineural administration is limited, and it may be associated with neurotoxicity.1
The Table outlines the studies evaluated, the doses of perineural adjuvants, the duration of analgesia, the effect and duration of sensory and motor blocks, and the adverse reactions of perineural adjuvants as reported in the listed studies.
SUMMARY
The advent of peripheral nerve blocks has significantly enhanced anaesthesiologists’ ability to manage acute postsurgical pain. Unfortunately, the benefits of peripheral nerve blockade are limited due to the duration of action of the LA. As a result, adjunct agents have been studied extensively to prolong the duration of regional nerve block analgesia. Many adjuvant medications have shown promising results in extending both sensory and motor blocks and improving postoperative analgesia. Despite these promising results, many studies have not demonstrated improved analgesia 24 hours following a surgical procedure. In addition, clinicians must consider other unwanted side effects of perineural block adjuvants including prolonged motor blockade, which may delay ambulation in day-surgery patients. Perineural use of these medications is not an approved or listed indication for clinical use according to the drug monographs; thus, this route of administration is considered off-label. There are also associated side effects and theoretical risks of neurotoxicity, which should be considered when choosing which adjuvant medication to administer perineurally. Despite the lack of neurotoxic effects observed in studies examining the medications described, this does not necessarily mean harm is absent, thus clinicians should consider if these adjuvant medications are suited to their patient’s needs. The best evidence for improved analgesia when using a peripheral nerve block appears to be the use of intravenous dexamethasone at doses of 0.1 to 0.2 mg/kg in moderately to severely painful surgical procedures.
REFERENCES
- Desai N, Kirkham KR, Albrecht Local anaesthetic adjuncts for peripheral regional anaesthesia: a narrative review. Anaesthesia. 2021;76(S1):100-109. doi:10.1111/anae.15245
- Glare P, Aubrey KR, Myles Transition from acute to chronic pain after surgery. Lancet. 2019;393(10180):1537-1546. doi:10.1016/S0140-6736(19)30352-6
- Tschopp C, Tramer MR, Schneider A, Zaarour M, Elia Benefit and harm of adding epinephrine to a local anesthetic for neuraxial and locoregional anesthesia: a meta-analysis of randomized controlled trials with trial sequential analyses. Anesth Analg. 2018;127(1):228-239. doi:10.1213/ANE.0000000000003417
- Schnabel A, Reichl SU, Zahn PK, Pogatzki-Zahn EM, Meyer-Frießem CH. Efficacy and safety of buprenorphine in peripheral nerve blocks:a meta-analysis of randomised controlled Eur J Anaesthesiol. 2017;34(9):576-586. doi:10. 1097/EJA.0000000000000628
- Williams B, Hough K, Tsui B, Ibinson J, Gold M, Gebhart Neurotoxicity of adjuvants used in perineural anesthesia and analgesia in comparison with ropivicaine. Reg Anesth Pain Med. 2011;36(3):1-7. doi:10.1097/AAP.0b013e3182176f70. Neurotoxicity
- Popping DM, Elia N, Marret E, et Clonidine as an adjuvant to local anesthetics for peripheral nerve and plexus blocks. Anesthesiology. 2009;111(2):406-415. doi:10.1097/aln.0b013e3181aae897
- Schnabel A, Reichl SU, Weibel S, et Efficacy and safety of dexmedetomidine in peripheral nerve blocks: a meta- analysis and trial sequential analysis. Eur J Anaesthesiol. 2018;35(10):745-758. doi:10.1097/EJA.0000000000000870
- Sun Q, Liu S, Wu H, et Dexmedetomidine as an adjuvant to local anesthetics in transversus abdominis plane block. Clin J Pain. 2019;35(4):375-384. doi:10.1097/AJP.0000000000000671
- Hussain N, Brummett CM, Brull R, et Efficacy of perineural versus intravenous dexmedetomidine as a peripheral nerve block adjunct: a systematic review. Reg Anesth Pain Med. 2021;46(8):704-712. doi:10.1136/rapm-2020-102353
- Zeng J, Chen Q, Yu C, Zhou J, Yang B. The use of magnesium sulfate and peripheral nerve blocks: an updated meta- analysis and systematic review. Clin J Pain. 2021;37(8):629-637. doi:10.1097/AJP.0000000000000944
- Pehora C, Pearson AME, Kaushal A, Crawford MW, Johnston Dexamethasone as an adjuvant to peripheral nerve block. Cochrane Database Syst Rev. 2017;2017(11). doi:10.1002/14651858.CD011770.pub2
- De Oliveira GS, Almeida MD, Benzon HT, McCarthy perioperative single dose systemic dexamethasone for postoperative pain. Anesthesiology. 2011;115(3):575-588. doi:10.1097/aln.0b013e31822a24c2
- Teshome D, Fenta E, Hunie M. Intravenous dexamethasone and peripheral nerve blocks: a systemic review and meta- analysis of randomized controlled trials. Int J Surg Open. 2020;26:86-96. doi:10.1016/j.ijso.2020.08.014
- Marty P, Bennis M, Legaillard B, et A new step toward evidence of in vivo perineural dexamethasone safety: an animal study. Reg Anesth Pain Med. 2018;43(2):180-185. doi:10.1097/AAP.0000000000000392
- Polderman JAW, Farhang-Razi V, Van Dieren S, et Adverse side effects of dexamethasone in surgical patients. Cochrane Database Syst Rev. 2018. doi:10.1002/14651858.CD011940.pub3



