General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- The following are recognised risk factors for air embolism:
a. Epidural insertion
b. Hypovolaemia
c. Inguinal hernia repair
d. Invasive blood pressure monitoring
e. Exteriorising the uterus during lower segment caesarean section - The following are true regarding venous air embolism:
a. Surgical emphysema is a typical clinical manifestation
b. Arrhythmias are always brady-arrhythmias
c. Sub-sternal chest pain and a dry cough may be reported
d. Fundoscopy findings are typically normal
e. Paradoxical air embolism may occur across the lung vascular bed - Regarding monitoring modalities for venous air embolism
a. Precordial doppler has the highest sensitivity for air embolism
b. Transcranial doppler is easy to use and readily available
c. Mill-wheel murmur auscultated with an oesophageal stethoscope is an early sign
d. End tidal nitrogen can be used to identify venous air embolism
e. Pulmonary artery catheters can be used to aspirate air emboli from the pulmonary vasculature
Key Points
- Air embolism can be both venous and arterial
- The risk factors can be categorised into patient, surgical and anaesthetic factors
- Air embolism may be difficult to detect – a high index of suspicion is required with consideration for potential differential diagnoses
- A number of monitoring modalities exists to aid early detection of air embolism
- Management includes prompt recognition, minimising further air entrainment and supporting affected organ systems
INTRODUCTION
There is significant morbidity and mortality associated with air embolism. It is therefore important for anaesthetists to have an awareness of its risk factors, clinical presentation, means of risk reduction and recognition to prompt timely management of the pathological consequences.
Any gas can result in embolisation if present in the vasculature. Its characteristics, mainly its solubility and volume will determine the clinical sequelae. Air is the commonest cause for this predominantly iatrogenic complication.
The following are basic definitions of different types of embolism. The clinical ramifications are different for each clinical entity.
Venous air embolism: air in the venous circulation occluding or impeding distal flow
Arterial air embolism: air in the arterial circulation, occluding arterioles with resultant distal hypoxaemia
Paradoxical air embolism: air crosses from venous to systemic circulation either via a congenital defect (e.g. a patent foramen ovale, PFO) or via the pulmonary circulation into the left heart.
RISK FACTORS
Any procedure where the operative site is higher than the right atrium and where the vasculature is exposed in a surgical field carries a risk of air embolism. The addition of a pressure gradient between the surgical site and the right heart substantially increases the risk. A pressure difference as small as 5 cm of water will allow 100 ml of air entrainment per second via a 14-gauge cannula.1 This is the rational for head down position during central venous cannulation. Hypovolaemia and negative pressure associated with spontaneous respiration will also increase the pressure difference and hence the risk.
Additional risks include interventions accessing the circulation such as central venous cannulation (causing air embolism) or using carbon dioxide insufflation during laparoscopic surgery (causing CO2 embolism). Air may also be inadvertently injected via a vascular access device.
The risk factors for air embolism can be broadly categorised into patient factors, surgical factors and anaesthetic factors, as detailed in Figure 1.
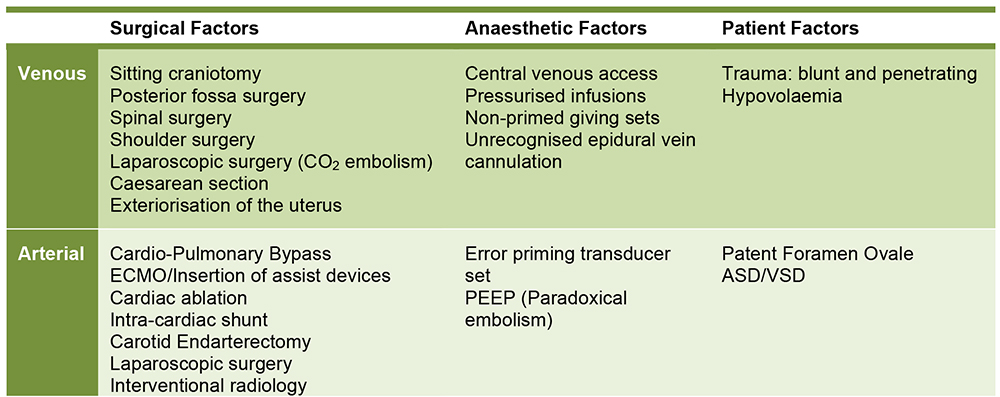
Figure 1: Surgical, anaesthetic and patient related risk factors for air embolism
Surgical procedures with high risk of gas embolism include sitting craniotomy, posterior fossa surgery and laparoscopic surgery.
It is important to note that in some studies, air embolism may have been detected without clinical sequelae such as demonstrated in the caesarean section study by Law et al 2 . The true incidence of air embolism associated with cardiac surgery is unknown, but it is considered an intermediate risk surgery for air embolism, with an estimated incidence of 5- 25%3. The neuropsychological consequences of open heart surgery and their postulated link with air embolism have greatly influenced the development of modern surgical approach and cardiopulmonary bypass circuits with lower flow techniques.
The use of Positive End Expiratory Pressure (PEEP) during patient’s ventilation to minimise the risk of air embolism is controversial. The concept that raising central venous pressure through the use of PEEP (>5 cm H2O) would minimise the risk of air entrainment seems logical. Animal studies also suggest that it can be protective in preventing venous air embolism in supine and upward tilt positions4. However, it has been implicated as a risk factor for paradoxical air embolism in patients with PFO5. Moreover, PEEP may also have an exaggerated effect on reducing preload due to reduced venous return secondary to increased intra-thoracic pressure. Sudden release of PEEP may also increase the rate of air entrainment in open venous beds within the surgical field.
CLINICAL MANIFESTATION
The clinical manifestation depends upon the rate and the volume of air entrained. Venous entrainment generally results in return of air emboli to the right side of the heart. A volume of 5ml/kg is considered large enough to cause an “air-lock” effect in the right ventricular outflow tract, with resultant cardiovascular collapse due to catastrophic reduction in cardiac output. The critical volume of air that is fatal in humans is unknown, but based upon animal models and case reports of fatalities, it is estimated to be approximately 200-300 ml in adults.
The clinical effect of arterial embolisation is highly dependent on the site. Air in small arterioles will usually be compensated for by collateral supply and may be reasonably well tolerated in some organs. However, the heart, lungs and brain are particularly sensitive and small volumes of air will result in significant complications.
The immediate clinical sequelae of air embolism can be considered using an organ system based approach:
Cardiovascular
An awake patient may experience chest pain and palpitations associated with arrhythmias, both brady- and tachyarrhythmias are possible. Ischaemic ECG changes may be found. Small volumes of air accumulating in the right heart and pulmonary vasculatures will lead to a gradual elevation in pulmonary artery pressure, which in turn will put the right heart under increasing strain. Ongoing air entrainment is more likely to be problematic as a large volume of air in the right ventricle (RV) will result in outflow tract obstruction and acute right sided heart failure. The reduced RV outflow will compromise left ventricular preload leading to cardiovascular collapse.
In the left side of the heart, small volumes of air entrained into the coronary circulation, especially the left anterior descending artery, rapidly leads to ischaemia and cardiac arrest. There is also a risk of a paradoxical air embolism occurring in patients with a PFO. PFO is co-incidentally found with an incidence of 35% at post mortem in otherwise healthy people. Paradoxical air embolism commonly results in symptoms of angina and/or an embolic stroke.
Respiratory
In an anaesthetised patient, a sudden drop in end tidal carbon dioxide is observed due to the deadspace ventilation caused by air in the pulmonary circulation. The degree of ventilation-perfusion mismatch will be revealed as hypoxaemia and hypercarbia on arterial blood gas analysis. Air embolism can also trigger an inflammatory cascade, resulting in an acute lung injury and non-cardiogenic pulmonary oedema. In an awake patient, sudden shortness of breath and pleuritic sub-sternal chest pain can occur with a dry cough. Haemoptysis is a relatively late sign.
Central nervous system
Arterial air embolism can lead to ischaemic stroke, which can manifest clinically as failure to wake up following general anaesthesia. Abnormal pupillary response to light may rarely occur if there is a significant paradoxical embolus resulting in a large ischaemic infarct with subsequent mass effect. If blood supply to the brain stem is interrupted, there may be cardiac dysrhythmias and apnoea. In an awake patient, there may be sudden onset of confusion, dysarthria, hemiparesis and seizure. The patient may deteriorate into a coma as cerebral oedema develops during the postoperative period. Gas bubbles may rarely be observed in retinal vessels on fundoscopy.
Gastrointestinal
Arterial air embolism can lead to abdominal pain and bowel ischaemia.
Skin
Crepitus may be palpated over superficial vessels. Surgical emphysema is not typically associated with venous embolism unless as a complication of a laparoscopic procedure.
In all cases of air embolism, a high index of suspicion is required and possible differential diagnoses need to be considered.
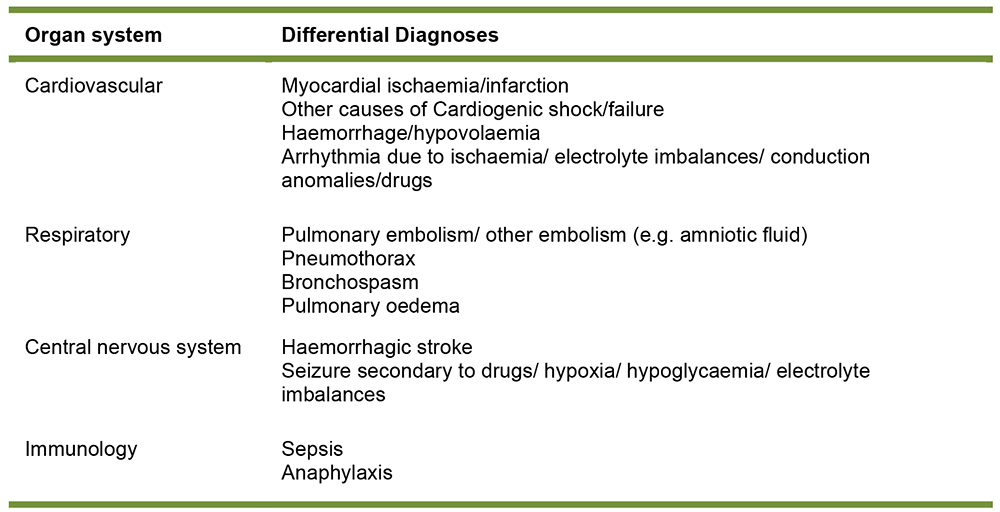
Figure 2: Differential diagnoses of air embolism
PREVENTION
High-risk procedures should be identified early and discussed prior to the operative date as well as during the surgical team brief. Discussion should include patient positioning for the proposed operation since this may alter pre-operative work-up (e.g. arranging a transthoracic or transoesophageal echocardiogram to exclude PFO before craniotomy in the sitting position). The anaesthetist should consider if a central venous catheter or an air aspiration catheter insertion is warranted. Excellent communication between the surgeon and the anaesthetist is vital, especially at high risk period during the procedure as this will allow swift action to be taken if air entrainment is suspected.
Meticulous attention to volume status is important, as maintaining preload will help to minimise the risk of air entrainment. Various novel approaches have been trialled to minimise the risk of air embolism in recognised high-risk procedures by reducing the pressure gradient between the circulation and site of air entrainment. Examples include the use of anti-shock compression garments to raise systemic venous pressure. Recently, the utilisation of intrajugular balloon catheters in a pig model has been demonstrated to be useful in blocking passage of air and partially effective in aspirating air 6.
MONITORING
Close patient monitoring plays a crucial role in early detection of air embolism, allowing early active management and measures to prevent further air entrainment. It is the responsibility of the anaesthetist to stay vigilant and pay careful attention throughout the operation.
The monitoring modalities discussed below are for detecting venous air embolism. There are no specific monitoring modalities used in routine clinical practice to detect arterial air embolism. A high index of suspicion, meticulous monitoring of clinical signs, coupled with timely detection of changes in routine monitoring such as end tidal CO2 and ECG changes are required for detection of arterial air embolism.
Alterations in clinical parameters lack specificity and often occur late after a significant volume of air has been entrained. Therefore, whilst clinical acumen is important, it should not be solely relied upon unless resource limitations do not allow for the monitoring modalities detailed below (Figure 3). Multiple modalities may be used in conjunction to improve specificity (e.g. end tidal CO2 with precordial doppler).
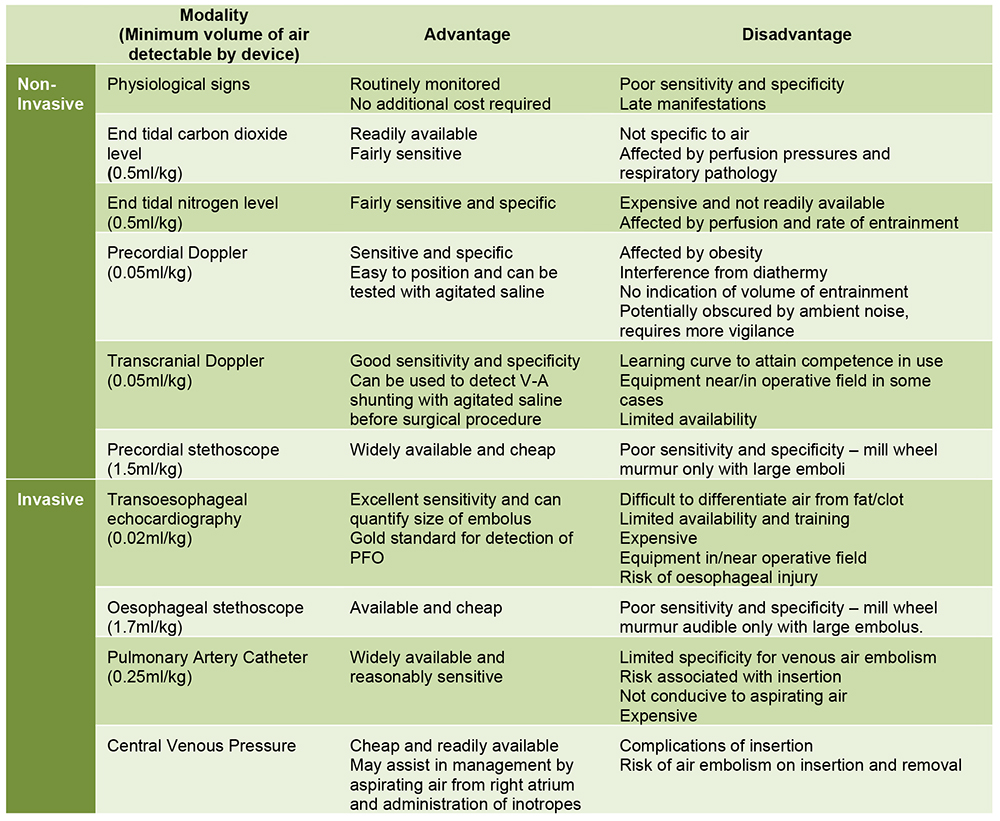
Figure 3: Monitoring modalities for detecting venous air embolism. Use will depend on availability, local expertise and surgical site.
Future developments may include the concurrent use of near infra-red spectroscopy as a means to monitor regional saturations alongside other monitoring modalities such as electroencephalography7. These techniques are still in experimental phase of animal based studies and require validation against current standards of monitoring. In cardiac surgery there are algorithms utilising transoesophageal echocardiography for detecting and managing air embolism intraoperatively8. Transoesophageal echocardiography has the highest sensitivity of all the monitoring devices for air embolism, detecting volumes of 0.02ml / kg – approximately 10 times the sensitivity of precordial Doppler. (Figure 4)
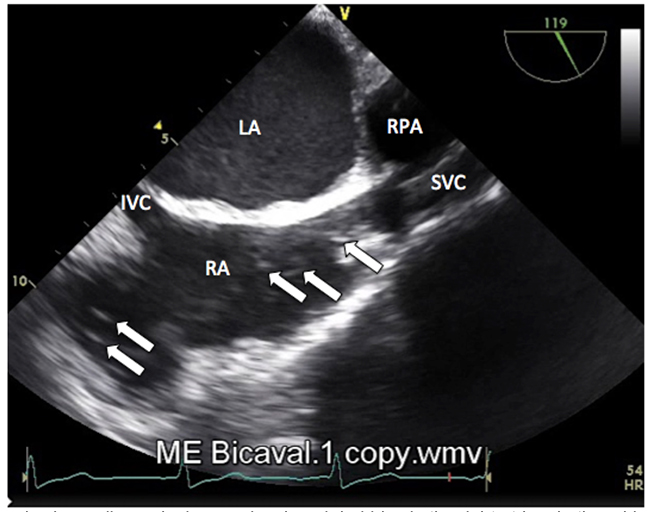
Figure 4: Transoesophageal echocardiography image showing air bubbles in the right atrium in the mid-oesophageal bicaval view. RA- right atrium; LA-left atrium; RPA-right pulmonary artery; SVC- superior vena cava; IVC- inferior vena cava (Photograph supplied by Dr. Chee Yee Eot, Consultant anaesthetist, Queen Mary Hospital, Hong Kong)
CLINICAL MANAGEMENT
Supportive treatment forms the mainstay of clinical management for venous and arterial air emboli diagnosed in the perioperative context. Management can be further subdivided into three elements that are invariably dealt with simultaneously:
- Immediate resuscitation
- Prevention of further air entrainment
- Efforts to remove or halt the progress of the air already entrained
Immediate resuscitation is best achieved by adopting an airway, breathing and circulation approach. In an anaesthetised patient, the airway should be secured with endotracheal intubation if this has not already been done. It is important to ensure that the inspired fraction of oxygen is increased to 1.0 and adequate ventilation is maintained. This can be confirmed by arterial blood gas analysis. Profound cardiovascular collapse and cardiac arrest can fast ensue following large venous or arterial air embolism. Circulatory support should be commenced rapidly to increase venous pressure. These include administering fluids via large bore intravenous cannulae as well as vasopressor or inotropic support as required. If cardiac arrest is imminent or has occurred, the initial rhythm may be pulseless electrical activity or asystole, in which case advanced life support protocol for non-shockable rhythms should be followed accordingly9. Where paradoxical or arterial emboli are suspected, signs of cardiac ischaemia should be sought and a 12 lead ECG should be examined post-operatively.
Attention should be paid to preventing further air entrainment by lowering the operative site to below the level of the heart and by stopping any process through which air could be entrained (e.g. reaming of bones during an orthopaedic surgery). Further air entrainment can also be minimised by directly compressing major blood vessels temporarily, the application of bone wax, flooding the operative sites with irrigation fluid and applying damp swabs over the suspected areas. Any gas pressurised system (e.g. pneumoperitoneum) should be decompressed. Nitrous oxide should be discontinued as it can expand any gas filled intravascular space.
Attempts can be made to aspirate air through an in situ central venous catheter or an air aspiration catheter (16G multiorifice catheter that can be inserted centrally or peripherally if adequate in length). It is preferable to use a multi-orifice tipped catheter to optimise chances of aspirating the air. With a multi-orifice catheter, the tip should be sited approximately 2 cm distal from the junction of the superior vena cava and right atrium. If a single lumen catheter is used, it should be positioned at 3 cm proximal to the superior vena cava-atrial junction. Radiological or intravenous ECG guidance has been recommended but are not always practical or available. To aspirate an air embolism most effectively, the trendelenburg and left lateral decubitus position are advocated because any entrained air within the heart should then theoretically float towards the right atrium and away from the coronary ostia, potentially be at a position allowing easier aspiration via a central line. In practice, it is not straightforward to perform such rapid aspiration unless an aspiration catheter or central venous line is already in situ. The logistics of repositioning the patient may also be difficult to achieve expediently due to concurrent resuscitation and an open surgical field. If a pulmonary artery catheter is in situ, it is unlikely to be effective in aspirating air as the lumens in the catheter are small and the catheters would not be in the optimal position for aspiration by the nature of their intended use.
AIR EMBOLISM SYNDROME
There is evidence from case reports that air embolism may result in a systemic inflammatory response type syndrome, with subsequent multi-organ dysfunction10.
Two pathophysiological theories have been proposed. The first suggests that the air emboli cause microvascular occlusions, leading to tissue ischaemia with resultant inflammation and organ dysfunction. This, however, does not account for why some patients with seemingly low volume air entrainment progress to develop such severe systemic inflammatory response. This is especially true in cases of paradoxical embolisation.
The second theory relates to a gene-environment mismatch11. Infection by certain gas-producing pathogens leads to formation of intravascular gas which can act as a trigger for the body’s innate immune system to combat infection. The same mechanism that can benefit the body during such an infection can have detrimental effects during an air embolism. The presence of air bubble in the circulation promotes platelet aggregation which can lead to systemic inflammation, disseminated intravascular coagulation and resultant multi-organ dysfunction.
Management of air embolism syndrome is based around organ supportive therapies in the intensive care unit.
SUMMARY
Air embolism can occur in the venous, arterial or paradoxically when air crosses from venous to arterial circulation. Risk factors can be considered according to surgical, anaesthetic and patient factors. Monitoring modalities allow early detection of venous air embolism. Management includes resuscitation, prevention of further air entrainment and support of organ dysfunction
ANSWERS TO QUESTIONS
- The following are recognised risk factors for air embolism:
a. True: There have been case reports when using air for loss of resistance or unrecognised vein puncture during epidural insertion.
b. True: Depending on the site of surgery, hypovolaemia can accentuate the pressure difference between the surgical site and right heart, increasing the risk of air entrainment and embolism.
c. False: There have been no reported cases.
d. True: If the pressure transducer set is not completely primed or during sampling, air can be introduced to the arterial system.
e. True: This is a particularly high-risk time for air embolism during caesarean section, as not only are there open venous beds in the surgical field, but the uterus has been raised above the level of the right atrium, creating an additional pressure gradient. - The following are true regarding venous air embolism:
a. False: Surgical emphysema is not associated with air embolism.
b. False: Air embolism can present as both brady- or tachyarrhythmias.
c. True: Both symptoms may be features noted by awake patients
d. True: Fundoscopy is typically normal; rarely may bubbles be seen in retinal vessels during an air embolism.
e. True: This is a recognised rare aetiology of paradoxical air embolism, that may be due to the volume of embolised air exceeding the capacity of the lungs to filter it out 9. - Regarding monitoring modalities for venous air embolism:
a. False: Trans-oesophageal echocardiography has the highest sensitivity for detecting air embolism.
b. False: Use and interpretation of transcranial Doppler requires training. Moreover, the equipment is not readily available.
c. False: Mill-wheel murmur is often only present in cases with large air embolism. It is also a late sign with low sensitivity and specificity.
d. True: This is a method for detecting air embolism as nitrogen diffuses out of the embolised air into the alveoli–see reference 2 and figure 3.
e. False: While air could technically be aspirated from a pulmonary artery catheter, the lumen is so small and the bubbles likely to be of small calibre at this site that it would not be effective.
REFERENCES AND FURTHER READING
- Flanagan JP, Gradisar IA, Gross RJ et al. A lethal complication of subclavian venepuncture. N Engl J Med 1969; 281: 488-9.
- Lew TW, Tay DH, Thomas E. Venous air embolism during caesarean section: more common than previously thought. Anesth Analg 1993; 77; 448-52
- Abu-Omar Y, Balacumaraswami L, Pigott DW, Matthews PM, Taggart DP: Solid and gaseous cerebral microembolization during off-pump, on-pump, and open cardiac surgery procedures. J Thorac Cardiovasc Surg 2004; 127:1759–65
- Pearl RJ, Larson LP. Hemodynamic effects of positive end-expiratory pressure during continuous venous air embolism in the dog. Anesthesiology. 1986; 64(6): 724-9
- Perkins NA, Bedford RF. Hemodynamic consequences of PEEP in seated neurological patients – implications for paradoxical air embolism. Anesth Analg 1984; 63 (4): 429-32
- Eckle VS, Neumann B, Greiner TO et al. Intrajugular balloon catheter reduces air embolism. Br J Anaesth 2015 Jun; 114 (6): 973-8
- Weenink RP, Hollmann MW, Stevens MF et al. Detection of cerebral arterial gas embolism using regional cerebral oxygen saturation, quantitative electroencephalography, and brain oxygen tension in the swing. J Neurosci Methods 2014 May 15; 228: 79-85
- Secretain F, Pollard A, Uddin M et al. A novel software program for detection of potential air emboli during cardiac surgery. Cardiovasc Ultrasound 2015 Jan 12; 13: 3
- Mishra R, Reddy P, Khaja M. Fatal cerebral air embolism: a case series and literature review. Case Rep Crit Care 2016 :3425321
- Kapoor T, Gutierrez G. Air embolism as a cause of the systemic inflammatory response syndrome: a case report. Crit Care 2003; 7: R98-100
- Alcock et al. Gene-environment mismatch in decompression sickness and air embolism. Med Hypotheses 2010; 75 (2): 199-203



