Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding sugammadex dosing:
a. Neuromuscular blockade of train-of-four count of two requires 4mg/kg sugammadex to adequately reverse the block
b. Reduced dosing is recommended in liver dysfunction
c. Reduced dosing is recommended in adults >75 years
d. Doses of 96mg/kg have been administered safely to adults
e. Reduced dosing is recommended in myasthenia gravis - Sugammadex side-effects include:
a. Dysguesia
b. Anaphylaxis
c. Post-operative recurisation with sugammadex doses >4mg/kg
d. Reduced efficacy of progesterone contraceptives
e. Has no effect on coagulation screen - Sugammadex in clinical use:
a. Is safe and effective in the pregnant population
b. Can reverse all depths of rocuronium induced neuromuscular blockade
c. Is an effective treatment for rocuronium-induced anaphylaxis
d. Recurisation may occur with co-administration with flucloxacillin
e. Can reverse atracurium induced neuromuscular blockade
Key Points
- Sugammadex is the first reversal agent to encapsulate aminosteroid neuromuscular blocking agents, reversing all depths of rocuronium neuromuscular blockade.
- The reversal dose of sugammadex required depends on the depth of neuromuscular blockade and the timing of when the neuromuscular blocking agent was given.
- Sugammadex alters the pharmacokinetics of rocuronium when bound as the rocuronium:sugammadex complex.
- Hypersensitivity concerns still exist with sugammadex, although globally sugammadex use appears to be safe and effective in most patient populations.
INTRODUCTION
Sugammadex is a unique reversal agent of aminosteroid-induced neuromuscular blockade. It is the first agent in its class. Initial human trials in 20051 found sugammadex to be safe and effective. Further studies followed, resulting in sugammadex being approved for clinical use in Europe and Australia in 2008.2,3 In 2015, however, the Food and Drug Administration (FDA) agency rejected licensing approval for a third time, citing hypersensitivity concerns. These issues have yet to be resolved with sugammadex still being unavailable in the United States of America.2 Sugammadex is a credible alternative to anticholinesterases in anaesthesia, it allows the use of rocuronium as a substitute to suxamethonium for rapid sequence induction (RSI) and has many potential uses in clinical practice, some of which we will discuss in this review.3
MECHANISM OF ACTION
Sugammadex is a direct reversal agent for aminosteroid neuromuscular blocking drugs (NMBD). It is a modified gamma cyclodextrin with a hydrophilic exterior and lipophilic centre, allowing it to encapsulate all aminosteroid NMBDs, with greatest affinity for rocuronium followed by vecuronium and then pancuronium.2 Each molecule of sugammadex encapsulates one molecule of rocuronium2 (Figure 1).
Sugammadex works by two mechanisms, firstly on entering the plasma it encapsulates the circulating aminosteroid rendering it inactive. Secondly, it promotes dissociation of the aminosteroid from the neuromuscular junction (NMJ) by creating a concentration gradient from the NMJ to the plasma, where it too is also encapsulated.2 This encapsulation of the NMBD allows return of neuromuscular function (NMF).
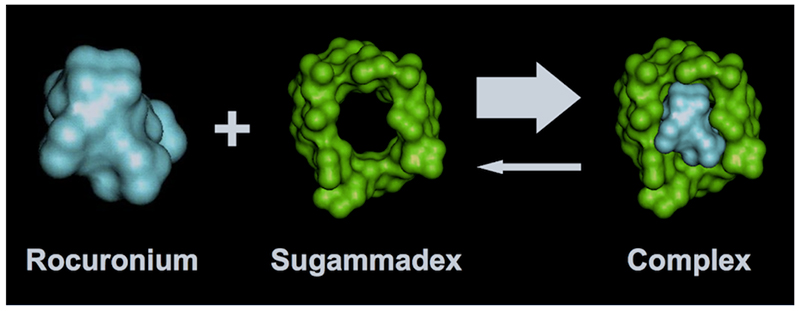
Figure 1: Rocuronium encapsulated by sugammadex. Image contributed by Dr Ronnie Palin and John MaClean at Merck
PHARMACOKINETICS
The pharmacokinetics of sugammadex is linear in the dose range 0.1mg/kg – 32mg/kg.2,4 However, one safety and efficacy study has suggested this linear relationship extends up to 96mg/kg.5 In the healthy adult population sugammadex has an estimated volume of distribution of 11-14 litres, an elimination half-life of around two hours and clearance of 88ml/min.4 Sugammadex is not metabolized and is excreted almost exclusively unchanged by the kidneys with more than 90% being renally excreted within 24 hours.4,5 Interestingly, administration of sugammadex alters the pharmacokinetics of rocuronium with the rocuronium-sugammadex complex behaving in a similar manner to sugammadex, with elimination of rocuronium being shifted away from the biliary system to predominant renal excretion.1,4,5
DOSING
Due to one-to-one binding capacity with rocuronium, sugammadex has the ability to reverse any depth of rocuronium induced neuromuscular blockade. The dose of sugammadex required to ensure complete reversal is dependent upon the depth of block, the timing and dose of rocuronium.4,6 The terminology used for depth of block: routine, moderate and profound, is defined by response to neuromuscular monitoring, train-of-four (TOF) and post-tetanic count (PTC). Routine block is represented by a TOF of 2 or more. Moderate block represents a deeper level of block and corresponds to a TOF of 0 and PTC 1-2. Profound block is the deepest level of neuromuscular block and occurs 3-5 minutes post NMBDs when there is no response on neuromuscular monitoring to TOF or PTC. Adequate reversal is determined by a TOF of >0.9, were the height of the forth twitch is 90% of the height of the first twitch.
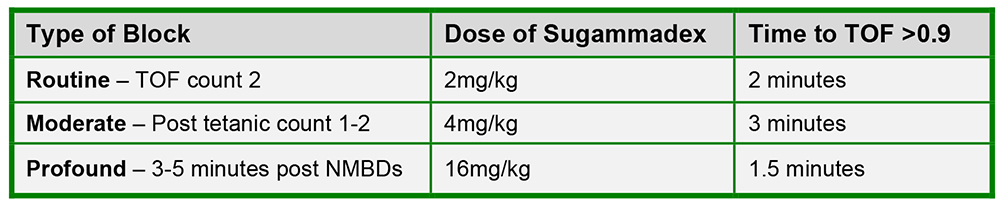
Figure 2: Table showing dose of sugammadex required to reverse different levels of rocuronium neuromuscular blockade.4
For reversal of routine and moderate rocuronium and also vecuronium-induced blockade, similar doses of sugammadex are required.4,6 However, the dose of sugammadex for reversal of profound vecuronium induced block has not been studied and therefore has not been formally determined.4,6
It is important to note that there is great variability in recovery times after administration of sugammadex at all depths of block and therefore neuromuscular monitoring is recommended until complete reversal is clinically achieved.3,4,6 Reduced doses of sugammadex (<2mg/kg) is associated with prolonged recovery times and incomplete reversal and is therefore not recommended.1,3,4,6
Should the situation arise that the patient requires repeat neuromuscular blockade after having received a dose of sugammadex, the recommendation is to utilise an alternative NMBD, for example, suxamethonium or a benzylisoquinolinium in the first instance. However, it is sometimes possible to re-paralyse the patient with rocuronium, but the dose required depends on the sugammadex dose given and the time since administration (Table Two).4 Paralysis onset with rocuronium in this situation is often delayed, with duration of action being shorter than normal.4
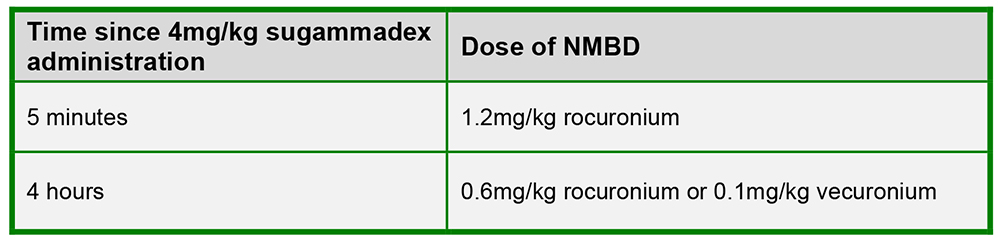
Figure 3: Table showing the dose of rocuronium required after administration of sugammadex.4
IMPLICATIONS FOR PRACTICE
Sugammadex versus Neostigmine
Following the administration of a non-depolarising NMBD, reversal often occurs spontaneously over a set duration of time or can in most cases be expedited through the administration of an anticholinesterase such as neostigmine in combination with glycopyrrolate. Neostigmine has well recognised undesirable effects and can result in variable reversal of neuromuscular blockade.2 It also has the disadvantage of not being able to reverse profound depth of blockade. If given during profound or moderate level of neuromuscular blockade, neostigmine can potentially exacerbate neuromuscular block by direct action on open nicotinic receptors. Early studies established that sugammadex compared with neostigmine was superior in producing complete reversal of neuromuscular blockade from both routine and moderate levels of block, occurring up to 17 times faster with sugammadex.6 These results have been consistent in subsequent studies, along with recognition of a reduced risk of post-operative residual blockade with sugammadex compared to neostigmine.1-3 A recent systematic review looked at the clinical benefit of sugammadex versus neostigmine in reducing events associated with post-operative residual blockade.7 This review found no difference in the occurrence of nausea and vomiting or any major life-threatening post-operative event when comparing sugammadex and neostigmine use.7
Another postulated benefit of sugammadex is facilitation of a moderate to profound neuromuscular blockade to the end of surgery, to improve surgical operating conditions and in theory outcomes.8 Determining clinical benefit of this technique is difficult as discussed in a recent review of laparoscopic surgery.8 Optimisation of operating conditions is not solely dependent on depth of block but also factors such as anaesthetic technique and laparoscopic inflation pressures.8 Most operating surgeons are often unable to ascertain differences between depths of blockade clinically.8
Sugammadex / rocuronium versus Suxamethonium
Suxamethonium has been the muscle relaxant of choice in rapid sequence induction (RSI) due to its rapid onset and offset of action.2 However, it is associated with numerous and potentially life-threatening adverse effects.2 Rocuronium 1.2mg/kg can emulate the intubating conditions of suxamethonium for RSI, but with the consequence of a prolonged duration of action, 35-75 minutes compared with less than 10 minutes.2 Administration of sugammadex after high dose rocuronium 1.2mg/kg, profound blockade, can result in a quicker return of NMF when compared to spontaneous resolution of suxamethonium induced blockade.1,6 Sugammadex at a dose of 16mg/kg will reverse profound neuromuscular blockade within 1.3 to1.9 minutes post administration.6 However, time taken to locate, calculate the dose and administer sugammadex may significantly delay time to reversal. It must be recognised that return of NMF does not necessarily equate to return of spontaneous ventilation in a ‘can’t intubate, can’t ventilate’ (CICV) situation.3 However, there are some case reports of successful use of sugammadex in CICV situations, warranting its inclusion on a departmental difficult airway trolley.1,3
Compared with suxamethonium, the use of rocuronium and sugammadex to facilitate electroconvulsive therapy (ECT) has been associated with shorter recovery times and reduced post-operative complaints such as myalgia and headache.3
Sugammadex for the treatment of rocuronium-induced anaphylaxis
It has been postulated that sugammadex has a role to play in the treatment of rocuronium-induced anaphylaxis, leading to a number of case reports.1,9 Such findings have not been supported by cutaneous testing or in-vitro experiments.1,9 A recent case-controlled study analysing sugammadex in the management of presumed rocuronium-induced anaphylaxis also could not endorse sugammadex use in this setting.9
SPECIFIC POPULATIONS
Elderly
The recommended dose of sugammadex for the elderly (>75yrs) is the same as younger adult patients.4 However, onset of action may be slower, which is possibly attributed to a reduced cardiac output in the elderly population.1
Paediatrics
Routine reversal of TOF count 2 with sugammadex 2mg/kg in children age two to 17 is recommended. However, despite safe use in a small number of case reports, sugammadex use is not endorsed in neonates or infants.1,4
Pregnancy and breastfeeding
There are no documented cases of sugammadex being used during pregnancy and is not recommended4 despite minimal placental transfer.2 There is however, multiple case reports of administration to women post caesarean section, which may allow rocuronium to become an acceptable alternative NMBD to suxamethonium for emergency obstetric intubation.1,3
Obesity
There is an ongoing debate surrounding the dosing of sugammadex in obese patients, as currently dosing is based on actual body weight.4 However, dose based on ideal body weight (IBW) plus 40% appears to be safe. Dosing based on IBW alone has been associated with slower recovery times and may lead to an increased risk of inadequate reversal.3
SPECIFIC CONSIDERATIONS
Neurological disease
Patients with myasthenia gravis pose particular challenges regarding NMBDs, as they require reduced doses, which have a variable duration of action, increasing the risk of post-operative residual blockade.10 Cholinesterase inhibitors such as neostigmine can result in inadequate reversal of NMBDs especially in patients who are taking pyridostigmine.10 A case series of 21 patients demonstrated that sugammadex was a safe and an effective alternative to neostigmine in patients with myasthenia gravis.10 De Boer et al also showed recovery times were similar to patients without neuromuscular diseases for reversal of both routine and moderate block to a TOF >0.9.10 There are case reports of safe sugammadex use in other neurological conditions including myotonic dystrophy, amyotrophic lateral sclerosis, Duchenne Muscular Dystrophy and Huntington’s disease.1,3
Cardiorespiratory disease
Sugammadex is safe for use in patients with pulmonary diseases such as bronchiectasis and cystic fibrosis. Bronchospasm has been documented following administration in a small number of patients.1,4 Sugammadex has also been used effectively in patients with cardiac failure and those who have received a heart transplant, but with a prolonged recovery time compared to the adult population.1,3 It is postulated again that this delayed recovery is due to reduced cardiac output as with the elderly population.1,4
Renal disease
Sugammadex use in patients with a creatinine clearance <30ml/min is not recommended.4 However a recent safety and efficacy study comparing reversal of moderate block with sugammadex 4mg/kg in patients with creatinine clearance <30ml/min to those with >80ml/min demonstrated complete reversal in those with severe renal impairment.11 Sugammadex was significantly slower at achieving a TOF>0.9 in the renally impaired compared to the normal population group, 3.1 versus 1.9 minutes.11 Sugammadex-rocuronium clearance was significantly reduced and remained detectable in a number of subjects seven days post administration.11 Using a high flux filter, haemodialysis can be used to remove sugammadex.1,4,11
Liver disease
Evidence for the use of sugammadex in liver dysfunction is limited. However, as sugammadex is almost solely excreted renally, no dose reduction is necessary in patients with mild to moderate hepatic dysfunction.4 More studies are needed in this area.
DRUG INTERACTIONS
Co-administration of sugammadex is not recommended with ondansetron, verapamil and ranitidine due to physical incompatibility.1 The effect of sugammadex is not altered by administration of magnesium and appears to be equally effective with both volatile and intravenous anaesthesia.3 The main drug interactions associated with sugammadex are displacement and capture reactions. Displacement reaction occurs when a drug displaces rocuronium from its sugammadex-rocuronium complex causing a theoretical risk of recurisation. Potential drugs causing displacement include toremifene, fusidic acid, flucloxacillin and diclofenac.1,2,4 However, such reactions have not been correlated into clinical practice.1 It should also be noted that rocuronium-sugammadex has an extremely high association constant and very low dissociation constant.2 Capturing reactions reduce the efficacy of other drugs when sugammadex encapsulates them. The most significant risk is with hormonal contraceptives, which may be less effective. Any woman who has received sugammadex should be counselled accordingly. They should obey the seven-day contraceptive rule and use extra contraceptive precautions for seven days following administration of sugammadex.1,2,4
ADVERSE EFFECTS
Hypersensitivity although rare, is by far the most concerning adverse effect of sugammadex, with a literature review highlighting that this can be severe and rapid in nature (anaphylaxis), with most cases presenting within five minutes of sugammadex exposure.12 It is important to note that none of the patients in the literature review died.12 It is not clear why sugammadex exposure may result in hypersensitivity, but previous food and pharmaceutical cyclodextrin exposure may be to blame.1,5,12 The incidence of hypersensitivity reactions is estimated at <1% after a single dose and is likely to increase with higher and repeated dosing of sugammadex.2,3 Diagnosis is confirmed via intradermal skin prick testing.12
Despite this, sugammadex is generally well tolerated.5,11 Common side effects include dysgeusia, headache, fatigue, nausea, vomiting, dizziness, urticaria and abdominal pain.4,5 An issue noted in initial studies was symptoms of inadequate depth of anaesthesia post administration of sugammadex such as grimacing, sucking and coughing.6 Elevated bispectral index levels were also noted post sugammadex administration.1 The FDA raised concerns regarding QTc prolongation, however subsequent studies using doses of sugammadex up to 32mg/kg have demonstrated there is no difference in QTc compared with placebo.1,2 A further issue highlighted by the FDA was the effect of sugammadex on coagulation due to early studies suggesting an increase in activated partial thromboplastin time (aPTT) possibly by the transient inhibition of factor Xa.2,13 A transient increase in aPTT and prothrombin time (PT) at 10 minutes that normalises by one hour in doses of 4mg/kg have not been associated with adverse clinical consequences.13 These results have been mirrored in other studies with sugammadex 4mg/kg, however further studies are needed with higher doses of sugammadex.2
Animal studies have suggested that sugammadex may be neurotoxic, but also highlighted that penetration of sugammadex across the blood-brain barrier is minimal, reducing this potential risk in humans.3
COST IMPLICATIONS
In the UK, sugammadex costs £29.82 per millilitre (100mg/ml)1. This compares with £1.15 per millilitre of neostigmine (2.5mg combined with 500 micrograms of glycopyrronium bromide). Unrestricted access to sugammadex has been associated with increased financial cost in a number of studies, without necessarily increasing patient turnover.1,2 Failure to use sugammadex in an emergency has also been cited as a potential medicolegal issue.3 Increased use of rocuronium may be associated with its own consequences such as increased rates of anaphylaxis.2
SUMMARY
Sugammadex is an effective and safe agent for reversing aminosteroid neuromuscular blockade, in particular all depths of rocuronium-induced blockade. Sugammadex provides a real alternative to neostigmine and has been used successfully in many clinical areas and patient populations that would have previously posed great angst to the anaesthetist, including patients with neuromuscular disorders. Sugammadex use has allowed rocuronium to become a comparable alternative to suxamethonium for RSI. Despite these virtues and being generally well tolerated globally, there are still some concerns regarding hypersensitivity, particularly by the FDA in the United States.
Experience with sugammadex worldwide is increasing, with many countries approving and using sugammadex safely. However, at present, routine first-line use of sugammadex has not been shown to be cost-effective. Further research is needed to improve how sugammadex use could be tailored to improve specific situations and become more financially viable. The safety of sugammadex in terms of hypersensitivity reactions and safety in certain populations such as paediatrics and pregnancy needs to be fully determined.
ANSWERS TO QUESTIONS
- Regarding sugammadex dosing
a) False: Neuromuscular blockade of train-of-four count two requires 2mg/kg sugammadex to adequately reverse the block
b) False: The same dosing is recommended in liver dysfunction as with non liver dysfunction
c) False: The same dosing is recommended in adults >75 years however the onset will be slower
d) True: Doses of 96mg/kg have been administered safely to adults
e) False: The same dosing is recommended in myasthenia gravis as with the normal adult population. - Sugammadex side-effects include:
a) True: Dysguesia can occur
b) True: Anaphylaxis is a rare side-effect. Hypersensitivity occurs in <1% of patients.
c) False: Post-operative residual curisation or residual blockade is rare and associated with doses <2mg/kg.
d) True: Risk of encapturing reaction reducing contraceptive efficacy
e) False: Sugammadex is associated with a transiently increased aPTT and PT - Sugammadex in clinical use
a) False: Sugammadex has not been studied in the pregnant population to deem whether it is safe and effective. However, there are many case reports clinically of its safe use in obstetrics post-caesarian section
b) True: It can reverse all levels of rocuronium induced neuromuscular blockade
c) False: Sugammadex has not been proven to be effective at treating rocuronium induced anaphylaxis
d) True: Recurisation may occur with co-administration with flucloxacillin due to a displacement reaction.
e) False: Sugammadex can only reverse aminosteroid induced neuromuscular blockade (rocuronium, vecuronium, pancuronium).
References and Further Reading
- Lobaz S, Clymer M, Sammut M. Safety and Efficacy of Sugammadex for Neuromuscular Blockade Reversal. Clinical Medicine Insights: Therapeutics. 2014;6:1-14
- Jahr JS, Miller JE, Hiruma J, Emaus K, You M, Meistelman C. Sugammadex: A Scientific Review Including Safety and Efficacy, Update on Regulatory Issues, and Clinical Use in Europe. Am J Ther. 2015;22(4):288-297
- Ledowski T. Sugammadex: what do we know and what do we still need to know? A review of the recent (2013 to 2014) literature. Anaesth Intensive Care 2015;43:14-22
- eMC. Bridion 100mg/ml solution for injection. https://medicines.org.uk/EMC/medicine/21299/SPC 2015. Updated May 28, 2015. Accessed on 15/10/2015.
- Peeters PAM, van den Heuvel MW, van Heumen E, Passier PCCM, Smeets JMW, van Iersel T, Zwiers A. Safety, Tolerability and Pharmacokinetics of Sugammadex Using Single High Doses (Up to 96mg/kg) in Healthy Adult Subjects. Clin Drug Investig 2010;30(12):867-874
- Abrishami A, Ho J, Wong J, Yin L, Chung F. Sugammadex, a selective reversal medication for preventing postoperative residual neuromuscular blockade. Cochrane Database Syst Rev. 2009;4:CD007362
- Abad-Gurumeta A, Ripollés-Melchor J, Casans-Francés R, Espinosa A, Martínez-Hurtado E, Fernández-Pérez C, Ramírez JM, López-Timoneda F, Calvo-Vecino JM, Evidence Anaesthesia Review Group. A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade. Anaesthesia 2015;70:1441-52
- Kopman AF, Naguib M. Laparoscopic Surgery and Muscle Relaxants: Is Deep Block Helpful? Anesthesia & Analgesia. 2015;120:51-8
- Platt PR, Clarke RC, Johnson GH, Sadleir PHM. Efficacy of sugammadex in rocuronium-induced or antibiotic induced anaphylaxis. A case-control study. Anaesthesia 2015;70:1264-7
- de Boer HD, Shields MO, Booij LHDJ. Reversal of neuromuscular blockade with sugammadex in patients with myasthenia gravis. A case series of 21 patients and review of the literature. Eur J Anaesthesiol 2014;31:708-721
- Panhuizen IF, Gold SJA, Buerkle C, Snoeck MMJ, Harper NJN, Kaspers MJGH, van den Heuvel MW, Hollmann MW. Efficacy, safety and pharmacokinetics of sugammadex 4mgkg-1 for reversal of deep neuromuscular blockade in patients with severe renal impairment. BJA 2015;114(5):777-784
- Tsur A, Kalansky A. Hypersensitivity associated with sugammadex administration: a systematic review. Anaesthesia 2014;69:1251-1257
- Rahe-Meyer N, Fennema H, Schulman S, Klimscha W, Przemeck M, Blobner M, Wulf H, Speek M, McCrary C, Williams-Herman D, Woo T, Szegedi A. Effect of Reversal of Neuromuscular Blockade with Sugammadex versus Usual Care on Bleeding Risk in a Randomized Study of Surgical Patients. Anesthesiology 2014;1212:969-77



