General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- In the diagnosis of anaphylaxis:
a. Suxamethonium and atracurium are the neuromuscular blocking agents most commonly associated with anaphylaxis
b. Mucocutaneous signs are a diagnostic feature in patients with anaphylaxis
c. Hypotension with tachycardia is always present in severe reactions
d. A single sample of elevated plasma total tryptase is diagnostic of anaphylaxis
e. Tryptase levels may be elevated more than 24 hours after the anaphylaxis episode - In the management of anaphylaxis:
a. Adrenaline may be less effective in patients on beta-blockers, so glucagon should be considered as a first line intervention in these patients
b. Intravenous antihistamines do not affect the clinical outcome and may increase the risk of hypotension and tissue necrosis
c. An initial intravenous dose of 100 micrograms of adrenaline should immediately be administered in all adults
presenting with intraoperative anaphylaxis
d. Steroids have been shown to improve clinical outcome when administered early
e. The patient should be admitted to intensive care for close monitoring - Preventative management strategies for a patient with a recent episode of anaphylaxis include:
a. Referral to an immunologist or specialised laboratory for skin testing
b. Skin testing because a positive result will help to verify the cause of anaphylaxis
c. Prompt skin testing as soon as the patient has recovered from the anaphylactic event
d. A desensitisation process if no other alternative medication can be used
e. Education on avoidance of suspected triggers and recognition of signs and symptoms of anaphylaxis
Key Points
- Early recognition and administration of adequate adrenaline is the mainstay of anaphylaxis management.
- Steroids and antihistamines should not be used in the initial management of anaphylaxis.
- Anaphylaxis is a clinical diagnosis, and tests such astotal plasma tryptase are only supportive of the diagnosis.
- The anaesthesiologist plays a key role in coordinating care for the patient during and after a perioperative anaphylaxis event.
INTRODUCTION
Anaphylaxis is a rare and potentially fatal complication with a wide spectrum of clinical presentations. The ability to recognise and treat it in the perioperative setting is imperative for every anaesthesiologist. According to the European Academy of Allergy and Clinical Immunology, anaphylaxis is defined as a severe, potentially life-threatening systemic hypersensitivity reaction.1 The estimated incidence of perioperative anaphylaxis is 1 in 10,000–20,000 anaesthetic procedures,2 with the overall mortality rate for anaphylaxis being less than 0.001%.1
PATHOPHYSIOLOGY
Anaphylaxis can be caused by immunological mechanisms (IgE-mediated or non-IgE-mediated), non-immunological mechanisms, or be idiopathic. A small quantity of allergen is sufficient to stimulate immune cells and cause anaphylaxis. The clinical diagnosis and management of anaphylaxis is the same regardless of the underlying mechanism.
Immunological mechanisms of anaphylaxis involve the production of immunoglobulins. IgE-mediated anaphylaxis is caused by the cross-linking of IgE resulting in degranulation of mast cells and basophils. Upon exposure to an allergen, there is activation of TH2 cells that stimulate the production of IgE antibodies. The IgE binds to FcεRI receptors on the surface of mast cells and basophils causing sensitisation of these cells. This initial sensitisation has no clinical manifestation. When there is re-exposure to the allergen, the receptor-bound IgE aggregates and stimulates sensitised mast cells and basophils to degranulate. This degranulation results in the release of mediators such as histamine, prostaglandins, proteoglycans, and cytokines, leading to the clinical manifestations of anaphylaxis. In non-IgE-mediated anaphylaxis, the hypersensitivity reaction is stimulated through other mechanisms including IgG and immune complex complement-mediated pathways.3
The non-immunological mechanism of anaphylaxis does not involve immunoglobulins and is due to direct stimulation of mast cells by the causative agent e.g. drugs, cold, and exercise. Similarly, there is degranulation of mast cells and release of mediators such as histamine. In the perioperative setting, this may be associated with drugs such as opioids, vancomycin, and neuromuscular blocking agents (e.g. atracurium), and is usually transient and may present with just cutaneous signs.
Idiopathic anaphylaxis is only diagnosed when no specific allergen can be identified from history or skin tests, and there is absence of elevation in serum specific IgE levels.
RISK FACTORS and TRIGGERS
Risk factors for anaphylaxis include a history of atopy, drug or food allergy, multiple operations, systemic mastocytosis, and hereditary angioedema. Common perioperative triggers of anaphylaxis include neuromuscular blocking agents, antibiotics (most commonly beta-lactam antibiotics), and latex. Less commonly, anaphylaxis may be provoked by chlorhexidine, colloids, blue dyes (isosulfan, patent blue V), heparin, protamine, and oxytocin.4 Chlorhexidine can be found in many products such as skin preparations and wipes, lubricant gels, and impregnated central venous lines and hence all the above should be avoided in patients with documented chlorhexidine allergy. It is rare for opioids or hypnotic agents (barbiturates, propofol, etomidate) to be the source of anaphylaxis.
The neuromuscular blocking agents most commonly associated with anaphylaxis are rocuronium and suxamethonium. Anaphylaxis occurs due to IgE sensitisation towards the quarternary ammonium groups in the neuromuscular blocking agents. In a retrospective study conducted by Sadleir et al. over a ten year period in Western Australia, it was noted that amongst the eighty patients diagnosed with life-threatening anaphylaxis to a non-depolarising neuromuscular blocking drug, 56% had a reaction towards rocuronium with an incidence of 8 anaphylaxis episodes per 100,000 administrations (95% CI 5.8–11/100,000). Cross-reactivity with other non-depolarising neuromuscular blocking agents is also highest with rocuronium and suxamethonium. In the study, patients with anaphylaxis to rocuronium had cross-reactivity rates of 44% with suxamethonium, 40% with vecuronium, 20% with atracurium, and 5% with cisatracurium. Cross-reactivity rates in patients with anaphylaxis to suxamethonium were 24% with rocuronium, 12% with vecuronium, and 6% with atracurium.5
DIAGNOSIS
Anaphylaxis is a clinical diagnosis. Laboratory tests are not useful in diagnosing anaphylaxis at the time of presentation because they take time to process and are prone to false negatives and positives.6 Patients may present with acute onset of generalised mucocutaneous signs, respiratory compromise, and cardiovascular instability. These signs may present in isolation or in any combination. Diagnosing anaphylaxis can be difficult because these signs may also arise from other anaesthetic, surgical, or patient-related factors in the perioperative setting. Anaphylaxis should be suspected if there is unexplained hypotension refractory to vasopressors, or unexplained difficulty in ventilation and bronchospasm. The severity of anaphylaxis can be graded from the mildest presentation of generalised mucocutaneous signs to the most severe form with cardiopulmonary arrest. Cutaneous signs may not be evident in all anaphylactic reactions and appear only after hypotension has been treated and cutaneous perfusion restored. Patients also may not have tachycardia with hypotension but instead bradycardia due to the Bezold–Jarisch reflex in severe hypovolaemia.2
It has been suggested that anaphylaxis can be graded clinically, as adapted from the Ring and Messmer four-step grading scale (Figure 1)7
Differential diagnoses of anaphylaxis include exacerbation of asthma, tension pneumothorax, myocardial ischaemia, pulmonary embolism, C1 esterase deficiency, mastocytosis and clonal mast cell disorders.
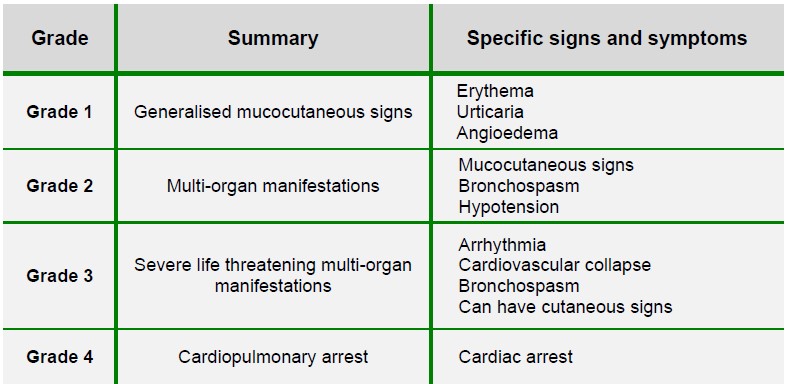
Figure 1: Grading of anaphylaxis, adapted from the Ring and Messmer grading scale 7
IMMEDIATE MANAGEMENT
The immediate management of anaphylaxis is to stop the suspected trigger, secure the airway, give high flow oxygen, and administer adrenaline in titrated doses with close haemodynamic monitoring.
The α1 agonist effect of adrenaline causes vasoconstriction, which decreases mucosal oedema and hypotension. Its β1 agonist effect increases inotropy of the heart while its β2 agonism decreases mediator release and enhances bronchodilation. Adrenaline is effective if anaphylaxis is recognised early and sufficient doses of the drug are administered quickly. The dose should be titrated to clinical response (Figure 2). An intravenous bolus can be given initially, but if the patient requires repeated boluses, a continuous infusion (0.05-0.4 micrograms kg-1 min-1) should be started. If intravenous access is unavailable, adrenaline can be injected intramuscularly at the lateral thigh while large bore intravenous access is established as soon as possible. If the patient is in cardiac arrest, resuscitation should follow the Advanced Life Support guidelines.8
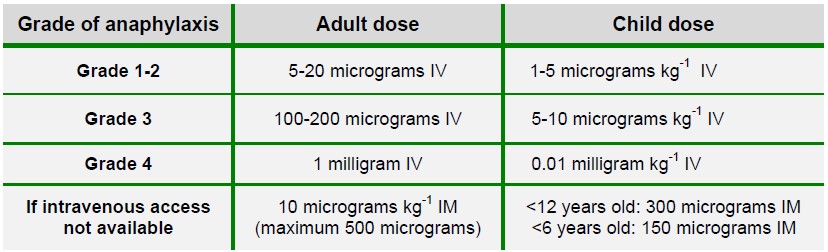
Figure 2: Bolus dose of adrenaline in anaphylaxis management7
Response to adrenaline may be diminished in patients on beta blockers, angiotensin converting enzyme inhibitors, or those who have a spinal blockade. Excessive administration on the other hand, can cause ventricular arrhythmias, pulmonary oedema, and hypertensive crisis. Adrenaline has a narrow therapeutic window and there are serious consequences with under- or overdosing of the drug. Therefore, intravenous adrenaline should be only be used by experienced practitioners (e.g. anaesthesiologists or intensivists) and in the presence of continuous monitoring. For anaphylaxis outside of the perioperative setting, it is recommended that intramuscular adrenaline be used initially.9
Other interventions in the management of patients with anaphylaxis include optimisation of the cardiovascular status by intravenous fluid resuscitation (repeated boluses of 20 ml kg-1 intravenous fluid to effect) and positioning of the patient for improved venous return. In addition to standard monitoring, invasive blood pressure monitoring allows better titration of adrenaline and a central venous catheter facilitates administration of vasopressors and inotropes.
Sugammadex, a reversal agent for neuromuscular blockade caused by rocuronium and vecuronium, may reverse anaphylaxis triggered by rocuronium.9 Sugammadex acts by encapsulating free and IgE-bound rocuronium, facilitating the elimination of the sugammadex-rocuronium complex from the body. The efficacy of sugammadex in reversing anaphylaxis is, however, still under study.
If the patient’s condition does not improve with first line interventions, other differential diagnoses should be considered. Ongoing resistant hypotension can be managed with administration of other vasopressors such as noradrenaline, metaraminol or vasopressin. In patients taking beta-blockers, glucagon can be given to reverse the beta blocking effects to improve hypotension. Resistant bronchospasm can be treated with intravenous salbutamol.
POST-CRISIS MANAGEMENT
After the initial resuscitation of the patient, steroids and antihistamines can be considered. However, both drugs have slow onset of action and have not been proven to alter the clinical outcome.7 Steroids (dexamethasone 0.1-0.4mg kg-1 or hydrocortisone 2-4mg kg-1) act by inhibiting the transcription of genes that encode pro-inflammatory proteins and may be effective in biphasic reactions. Intravenous antihistamine may carry more risks than benefits, including the risk of hypotension with rapid injection and tissue injury if extravasated.8 Oral antihistamines have a better side effect profile and may be effective in grade 1 anaphylactic reactions. Combined H1 and H2 receptor antagonists (e.g. use of diphenhydramine with ranitidine) may be more effective than H1 antagonists alone in treating cutaneous manifestations of anaphylaxis.10, 11
When intraoperative anaphylaxis occurs, it is prudent to conclude the surgery as soon as possible. Postoperative intensive care admission should be organised for monitoring because the condition can be prolonged up to 32 hours and biphasic reactions occur in up to 20% of cases. Even with adequate response to treatment, patients should be meticulously monitored for at least 6-8 hours if they had respiratory symptoms, and for no less than 12-24 hours if they had cardiovascular collapse. The pathophysiology and risk factors of biphasic reactions are unknown as there have not been consistent findings between studies. Several hypotheses of biphasic reaction include inadequate treatment of the initial reaction, release of late-phase mediators from immune cells, delayed absorption of the antigen from the gastrointestinal tract into the systemic circulation, and activation of secondary mediator pathways.12
Appropriate investigations such as arterial blood gas, renal and liver function tests should also be considered to detect hypoperfusion injuries in other organs. Plasma total tryptase levels may help to support the diagnosis. Tryptase is found in mast cells in two forms, α-protryptase and β-tryptase. While plasma α-protryptase can be found in normal conditions, β-tryptase is only released from mast cell granules during an allergic reaction. Plasma total tryptase levels measure both α-protryptase and β-tryptase and may be elevated in anaphylaxis. Plasma β-tryptase level is more specific for anaphylaxis and may be done if available. Tryptase level peaks within 15-120 minutes after the onset of anaphylaxis before declining slowly back to baseline levels by 24 hours. As a definite cut-off value for the diagnosis has not been determined, it is important that at least two blood samples for total tryptase be taken – the first within 60 minutes of the reaction and the second at 24 hours. Anaphylaxis is likely if the first sample is significantly higher than the second. If tryptase levels remain increased beyond 24 hours, it may indicate late-onset anaphylaxis, biphasic reaction, or underlying mastocytosis or clonal mast cell disorders.
Elevated total tryptase level is not specific to anaphylaxis and can be raised in other unrelated conditions such as myocardial infarction, amniotic fluid embolism, or trauma. Elevated tryptase may also be absent in anaphylaxis if the patient had basophil-mediated or non-IgE-mediated anaphylaxis. In a study where the tryptase levels in patients with non-anaphylaxis related shock were compared to the levels in patients with grade 3 or 4 anaphylaxis, the positive predictive value of total tryptase levels for the diagnosis of anaphylaxis was 99.7% and the negative predictive value 27.9%.13 Tryptase levels can only aid the diagnosis of anaphylaxis in the context of the clinical manifestations.
Another test to support the diagnosis of anaphylaxis is elevated plasma histamine levels. However, this is not specific for anaphylaxis and due to histamine’s short plasma half-life of 15-20 minutes, it is also difficult in practice to capture an elevated level during resuscitation. Twenty-four hour urine histamine levels are more specific for anaphylaxis and may be done if available.
PREVENTION OF ANAPHYLAXIS
One important aspect in the prevention of future anaphylaxis is the identification of the possible allergen(s). The patient should be referred to an immunologist or a centre specialising in allergy testing. Either skin tests (skin prick test, intradermal tests), measurement of serum allergen-specific IgE levels, or challenge tests can be performed to identify possible allergens. Skin tests can be done for many common allergens, including neuromuscular blocking agents, intravenous and local anaesthetics, antibiotics, latex, chlorhexidine, colloids, and blue dyes. They are less helpful in diagnosing allergy to opioids, non-steroidal anti-inflammatory drugs, and paracetamol, in which cases oral challenge tests may be needed instead.14
Skin tests should be deferred to 3-4 weeks after the initial anaphylactic episode to minimise any false negative response due to histamine depletion. If skin prick tests are negative but the patient is strongly suspected to be allergic to a particular drug, an intradermal test or serum agent-specific IgE can be measured. However, asymptomatic sensitisation is common. A positive skin test or elevated agent-specific IgE level only indicates that the patient is sensitised towards that agent but does not verify that it is the cause of anaphylaxis. Interpretation of allergy testing should be done in collaboration with an immunologist. Important considerations include the temporal relationship between drug administration and anaphylactic presentation, severity and clinical manifestation of the anaphylactic episode, and patient’s response to treatment.
Allergy testing should be carried out in all high-risk patients before any future anaesthesia. High-risk patients include those with history of intraoperative anaphylaxis, known allergy to drugs likely to be used in anaesthesia, children who have undergone multiple interventions (in particular surgery for spina bifida or myelomeningocele), and patients with latex allergy or food allergy to exotic fruits.15 Routine testing of patients not at risk has no role.
After an anaphylactic episode, the anaesthesiologist should ensure clear documentation of the incident, suspected allergen, treatment administered, and the effects of the treatment. A letter containing this information should be sent to both the primary and secondary care team of the patient. The allergy alerts should be updated on all patient records. The anaesthesiologist is also responsible for referring the patient to the immunologist for further investigation and conducting a follow-up appointment to advise on future anaesthesia. Patient education on the causes, symptoms, and immediate management of anaphylaxis, including the use of adrenaline auto-injectors, is also important to prevent recurrence.
Besides avoidance of drugs that tested positive for hypersensitivity reaction, other ways to further optimise high-risk patients in preventing perioperative anaphylaxis have been studied. These include pre- emptive administration of glucocorticoids or antihistamines for procedures that are deemed high-risk for anaphylaxis e.g. diagnostic procedures involving iodinated contrast media, anti-venom therapy, immunotherapy, and anti-IgE antibody therapy. However, there have been few randomised controlled trials, and thus far no strong evidence exists indicating that premedication can reduce the risk of life-threatening anaphylaxis.16 There is mixed evidence to support the use of antihistamines or steroids as premedication in patients with mastocytosis. Anxiety-triggered mast cell degranulation in mastocytosis may be ameliorated by premedication with a benzodiazepine.17
The choice of drugs in subsequent anaesthesia is guided by the result of allergy tests and the clinical indication for the drugs. If no safe alternative is available, the patient may need to undergo desensitisation towards the triggering medication before an elective procedure. Desensitisation needs to be done by the immunologist in the intensive care setting with close monitoring of the patient. In patients with a history of anaphylaxis to neuromuscular blocking agents, regional or local anaesthetic techniques should be used where possible. If general anaesthesia with endotracheal intubation is required, neuromuscular blocking agents should be avoided and alternatives such as opioids be used to facilitate the procedure.
SUMMARY
Knowledge of the recognition, management, and prevention of perioperative anaphylaxis is necessary for all anaesthesiologists. Due to the large number of drugs and substances that patients are exposed to during anaesthesia, identifying the triggering agents can be difficult. Immunological tests play an important role but the findings must be interpreted with caution as there are high rates of false positives and negatives. Adrenaline remains the most effective first-line intervention in the management of anaphylaxis. As there is growing understanding of the pathophysiology of anaphylaxis, studies on the effectiveness of other drugs for initial treatment and alternative biomarkers for diagnosis are underway.
ANSWERS TO QUESTIONS
- ..
a. False. The most common neuromuscular blocking agents associated with anaphylaxis are rocuronium and suxamethonium. There is an incidence of 8 anaphylaxis episodes per 100,000 administrations of rocuronium.
b. False. Anaphylaxis can present with different severities and does not always have mucocutaneous signs. Cutaneous signs may not be present if there is severe hypotension.
c. False. There may be bradycardia with hypotension due to the Bezold–Jarisch reflex in severe hypovolaemia.
d. False. A comparison of at least two plasma total tryptase levels should be used because there are many other causes of a single elevated plasma tryptase level.
e. True. Elevated tryptase levels beyond 24 hours may be due to late-onset anaphylaxis or a biphasic reaction. Other differential diagnoses should also be considered including mastocytosis or clonal mast cell disorders. - ..
a. False. Adrenaline is always the first line intervention for anaphylaxis; glucagon can be given if the patient on beta-blockers is refractory to adrenaline.
b. True. Antihistamines should not be used in the immediate management of anaphylaxis. If they are to be used, oral antihistamines have a safer side effect profile than intravenous antihistamines.
c. False. The dose of adrenaline depends on the grading of anaphylaxis and should be titrated to clinical response.
d. False. Steroids are not used in the immediate management because their onset is slow and have not been shown to improve clinical outcome.
e. True. Anaphylaxis can be protracted up to 32 hours or the patient may have a biphasic reaction. - ..
a. True. Appropriate skin tests should be selected and interpreted by experienced immunologists.
b. False. A positive skin test only indicates sensitisation to the allergen, but does not verify that the allergen is the cause of anaphylaxis.
c. False. Skin tests should be done at least 3-4 weeks after anaphylaxis because histamine may be depleted during anaphylaxis, resulting in a risk of false negative results.
d. True. If no safe alternative is available, the patient may need to undergo desensitization to the medication under close monitoring in the intensive care unit.
e. True. Since certain triggers can be found in the community setting, it is important for the patient to avoid the triggers and seek medical attention when needed.
REFERENCES and FURTHER READING
- Muraro A, et al. on behalf of the EAACI Food Allergy and Anaphylaxis Guidelines Group. Anaphylaxis: guidelines from the European Academy of Allergy and Clinical Immunology. Allergy 2014;69:1026-1045.
- Dewachter P, Mouton-Faivre C, Emala CW. Anaphylaxis and Anesthesia. Anesthesiology 2009;111:1141-1150.
- FE Simons. Anaphylaxis. J Allergy Clin Immunol 2010;125:161-181.
- Kannan JA, Bernstein JA. Perioperative Anaphylaxis. Immunol Allergy Clin North Am 2015;35:321-334.
- Sadleir PHM, Clarke RC, Bunning DL, Platt PR. Anaphylaxis to neuromuscular blocking drugs: incidence and cross-reactivity in Western Australia from 2002 to 2011. Br J Anaesth 2013;110:981-987.
- Simons FER, Ardusso LRF, Bilo MB, Cardona V, Ebisawa M, El-Gamal YM. International consensus on anaphylaxis. World Allergy Organ J 2014;7:9.
- Australian and New Zealand College of Anaesthetists. ANZAAG-ANZCA Anaphylaxis Management Guidelines Version 1.1; 2013
- Link MS, Berkow LC, Kudenchuk PJ, Halperin HR, Hess EP, Moitra VK, Neumar RW, O’Neil BJ, Paxton JH, Silvers SM, White RD, Yannopoulos D, Donnino MW. Part 7: adult advanced cardiovascular life support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2015; 132(suppl 2):S444–S464.
- Simons FER, Ardusso LRF, Dimov V, Ebisawa M, El-Gamal YM, Lockey RF, et al. World Allergy Organization Anaphylaxis Guidelines: 2013 Update of the Evidence Base. Int Arch Allergy Immunol. 2013;162:193-204.
- Wechsler JB, Schroeder HA, Byrne AJ, Chien KB, Bryce PJ. Anaphylactic responses to histamine in mice utilize both histamine receptors 1 and 2. Allergy 2013;68:1338-1340.
- Lin RY, Curry A, Pesola GR, Knight RJ, Lee HS, Bakalchuk L, et al. Improved outcomes in patients with acute allergic syndromes who are treated with combined H1 and H2 antagonists. Ann Emerg Med 2000; 36:462-8.
- Tole JW, Lieberman P. Biphasic anaphylaxis: review of incidence, clinical predictors, and observation recommendations. Immunol Allergy Clin N Am. 2007; 27: 309-326.
- Laroche D, Gomis P, Gallimidi E, Malinovsky JM, Mertes PM. Diagnostic value of histamine and tryptase concentrations in severe anaphylaxis with shock or cardiac arrest during anesthesia. Anesthesiology 2014;121:272-279.
- Ewan PW, Dugue P, Mirakian R, et al. BSACI guidelines for the investigation of suspected anaphylaxis during general anaesthesia. Clin Exp Allergy 2010;40:15-31.
- Mertes PM, Malinovsky JM, Jouffroy L, Aberer W, Terreehorst I, Brockow K. Reducing the Risk of Anaphylaxis During Anesthesia: 2011 Updated Guidelines for Clinical Practice. J Investig Allergol Clin Immunol 2011;21:442-453.
- Sheikh A. Glucocorticoids for the treatment and prevention of anaphylaxis. Curr Opin Allergy Clin Immunol 2013;13:263-267.
- Bonadonna P, Lombardo C. Drug allergy in mastocytosis. Immunol Allergy Clin North Am 2014;34:397-405.



