Obstetrics Anaesthesia
KEY POINTS
- General anaesthesia in pregnant women is associated with a higher risk of difficult airway and awareness when compared with the nonobstetric population.
- Neuraxial anaesthesia is preferable to general anaesthesia for caesarean delivery as it is associated with less maternal and neonatal morbidity and mortality.
- Small-gauge pencil-point spinal needles (25 gauge or smaller) are less likely to cause postdural puncture headache.
- Prophylactic measures to avoid spinal-induced hypotension are important to maintain utero-placental perfusion—a phenylephrine infusion is recommended (initiated at 25-50µg/min) immediately after administration of intrathecal medication(s).
- Oxytocin is recommended with an initial bolus dose of 1 to 3 IU intravenously, followed by a titrated infusion.
- The addition of neuraxial opioids improves the quality of the intraoperative block and postoperative analgesia.
INTRODUCTION
Caesarean delivery (CD) is one of the commonest surgeries performed globally.1 According to the 2015 World Health Organization (WHO) systematic review, CD rates of 10% to 15% or less are associated with a decrease in maternal and perinatal mortality when performed for medical indications.2 The WHO recommends that CD should be available to any woman who requires it, rather than aiming for a specific rate.3 CD rates are increasing globally; it is imperative to optimise surgical and anaesthetic practices to maximise maternal and neonatal outcomes.
CHOICE OF ANAESTHESIA FOR CD
The change in practice from general anaesthesia (GA) to neuraxial anaesthesia (NA) halved anaesthesia-related maternal mortality in the United Kingdom from 30 to 50 per 100 000 maternities per triennial in the 1970s, to 19 per 100 000 maternities in the early 1980s.4 Recent evidence suggests that NA is associated with better foetal outcomes (Apgar scores and cord pH) when compared to GA.5
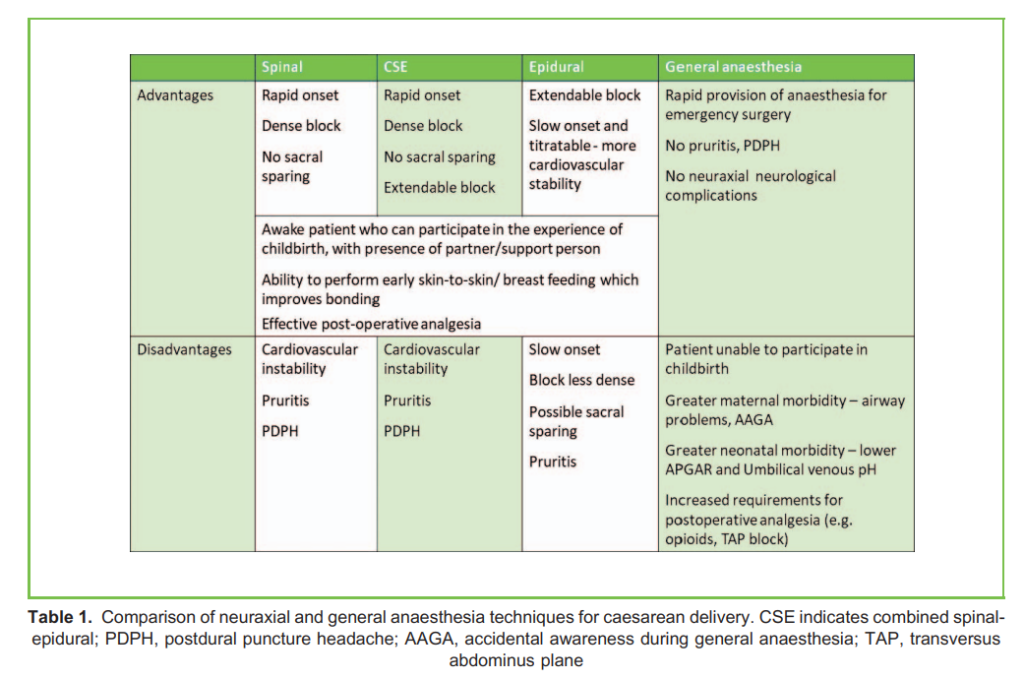
Although NA is favourable for CD, there are times when GA is indicated, for example in emergent situations when there is not enough time to establish NA, or if there is a contraindication to NA. Table 1 lists advantages and disadvantages of anaesthesia
techniques for CD.
DEFINING URGENCY OF CD
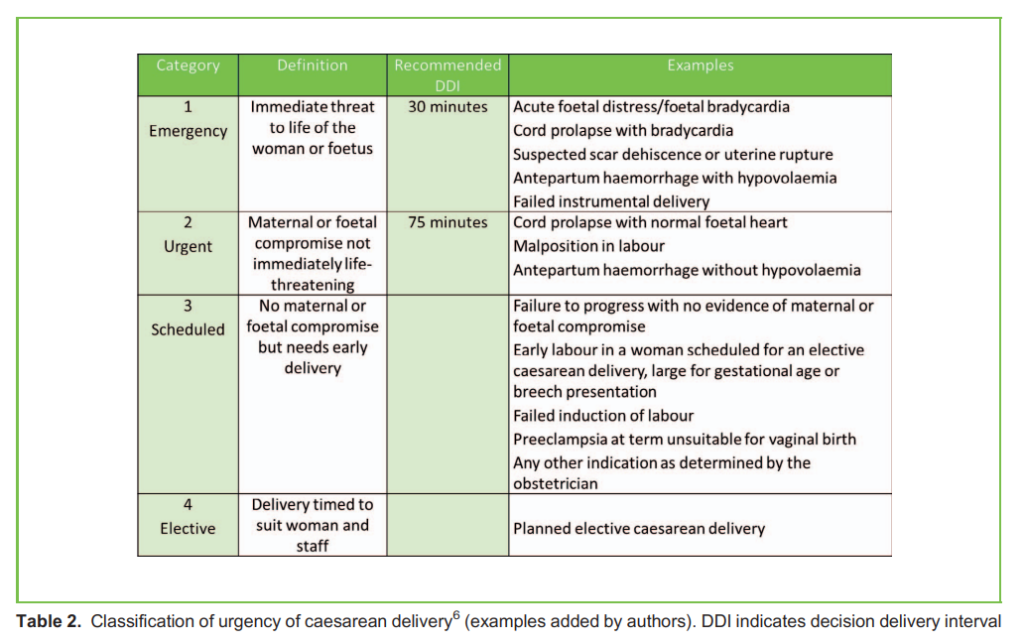
To better communicate the urgency of CD, the Royal College of Obstetricians and Gynaecologists developed a 4-category classification (Table 2).6
The optimal timing for elective CD is approximately 39 weeks gestation in order to allow foetal lung maturation and minimise neonatal respiratory distress.7 However, about 10% of women may experience spontaneous labour prior to this time, and potentially necessitate an emergent CD.8
NEURAXIAL ANAESTHESIA FOR CD
There are various NA techniques including single-shot spinal, epidural, and combined spinal-epidural (CSE) that share common technical considerations:
- The sitting position (versus the lateral) is more commonly practiced because it is easier to approximate the midline and anticipate the needle trajectory in relation to anatomical landmarks.
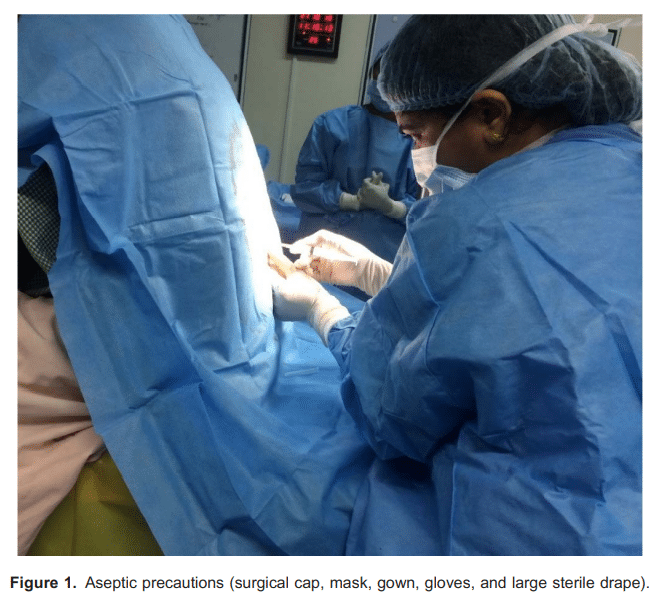
- Meticulous aseptic precautions should be taken, which include surgical cap and mask; hands and forearms washed with an antiseptic solution, ideally alcohol-based9 ; sterile gloves (sterile gown optional); and use of a large sterile drape (Figure 1). Chlorhexidine in alcohol is recommended over iodine for skin disinfection because it has a greater
bactericidal effect that lasts several hours. There is geographical variation in expert consensus as to the relative risks and benefits of 0.5% (commonly used in Europe) versus 2% chlorhexidine (commonly used in North America) with regard to neurotoxicity and antiseptic efficacy. Reliable procedural barriers to prevent potential contamination are essential: prepping skin prior to opening sterile neuraxial kit; using coloured antiseptic solution so any contamination of drugs, equipment (needle or syringe), or gloves is obvious; and use of a stick rather than a spray or swabs to reduce splashing potential. The solution should not pool and should be allowed to dry fully (at least 3 minutes) prior to the NA procedure.10 - The skin puncture should be caudal to where the spinal cord terminates; insertion at or below the L3-4 interspace avoids trauma to the conus. Estimation of the spinal level using anatomical landmarks can be inaccurate, with one study showing correct identification in only 29% of cases with the majority of cases being at a higher level than expected (as many as 4 vertebral levels higher), which places the patient at risk of direct spinal cord injury from a spinal needle.11 Preprocedural ultrasound can assist with appropriate identification of the vertebral level.12
- Prior to the skin incision, the adequacy of the block should be assessed, ideally by more than one sensory modality. A lower limb motor block is reassuring and bilateral loss of sensation to pinprick to the T4 dermatomes is a reliable indicator that the patient has adequate surgical anaesthesia for CD. Sensation should be checked in the midclavicular line and not in the midaxillary line as dermatomes slope forward and downward toward midline, which could give false verification of an adequate block height. Patients will still experience a pressure sensation during the CD, most notably with stretching of the rectus muscles and fundal pressure at delivery.
- Supplementary analgesia (or anxiolysis) may be required despite a documented ‘adequate’ NA block for CD, so the anaesthetist needs to respond to any patient discomfort. In a minority of cases, NA will need to be converted to GA (eg if the patient experiences pain unrelieved by adjuncts, or during prolonged surgery).
Single-Shot Spinal Technique for CD
Single-shot spinal anaesthesia is commonly used as the NA technique for scheduled CD because of its reliability and rapid onset of action.
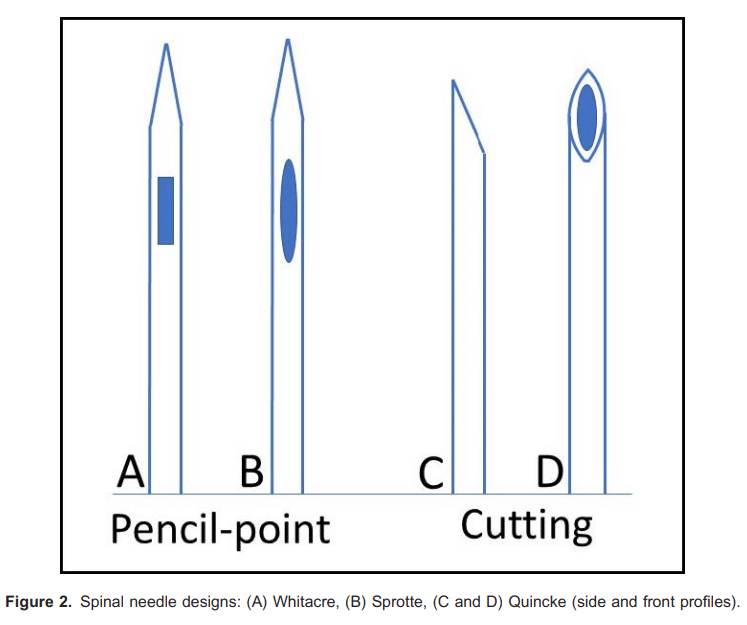
Small-gauged (ideally 25 gauge or smaller) pencil-point spinal needles (eg Whitacre or Sprotte) reduce the incidence of postdural puncture headache compared to cutting needles (eg Quincke) (Figure 2). Care should be taken in thin patients to avoid inserting the introducer needle too deep, which could inadvertently cause a dural puncture (Figure 3).
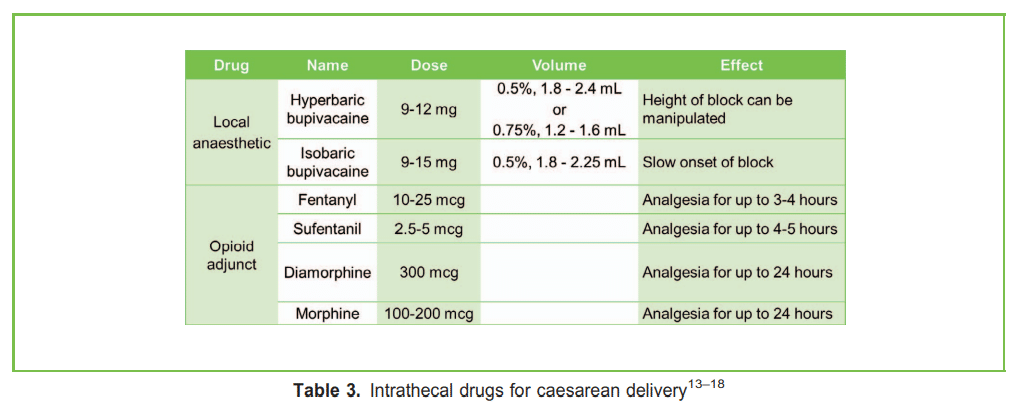
Drugs for Spinal Anaesthesia (Table 3)
- The dose required to achieve the desired effect in 95% of the population (ED95) of hyperbaric (contains 80 mg/mL glucose) bupivacaine is 11.2 mg when administered with opioid for CD.13 Commonly, 10 to 15 mg of intrathecal bupivacaine is administered for CD using 2.0 to 3.0 mL of 0.5% bupivacaine, or 1.3 to 2.0 mL of 0.75% bupivacaine.14 Prior to the hyperbaric solution ‘setting’, the height of the block can be manipulated by slightly tilting the table into a Trendelenburg position.
- Isobaric bupivacaine can be administered for spinal anaesthesia, and the ED95 is 13 mg when administered with opioid for CD.13 A systematic review comparing isobaric with hyperbaric bupivacaine did not find compelling evidence favouring one over the other.14
 Intrathecal opioid adjuncts such as fentanyl (10-25 µg) or sufentanil (2.5-5 µµg) improve the quality of spinal anaesthesia by augmenting the effect of the local anaesthetic, which reduces intraoperative discomfort in particular if the uterus is exteriorised. Higher doses (eg fentanyl . 20µg and sufentanil . 2.5 µg) increase the incidence of pruritus.15,16
Intrathecal opioid adjuncts such as fentanyl (10-25 µg) or sufentanil (2.5-5 µµg) improve the quality of spinal anaesthesia by augmenting the effect of the local anaesthetic, which reduces intraoperative discomfort in particular if the uterus is exteriorised. Higher doses (eg fentanyl . 20µg and sufentanil . 2.5 µg) increase the incidence of pruritus.15,16 - Adding preservative-free diamorphine (300 µg) or preservative-free morphine (100-200 µg) to the spinal anaesthesia contributes to postoperative analgesia for up to 24 hours.17,18
Failed or Inadequate Spinal Anaesthesia
The potential aetiology of failure can be classified as follows:
- Anatomical:
* Failure of dural puncture with the spinal needle due to spinal abnormalities such as kyphosis, scoliosis, limitation of spinal flexion, or calcified ligaments
* Failure of local anaesthetic spread caused by adhesions or septae in the epidural space (eg secondary to previous surgery)
- Technique:
* Loss of drug between the needle hub and syringe, or partial deposition of the anaesthetic solution in the subdural or epidural space resulting in an inadequate effect despite obtaining flow of cerebrospinal fluid in the spinal needle19
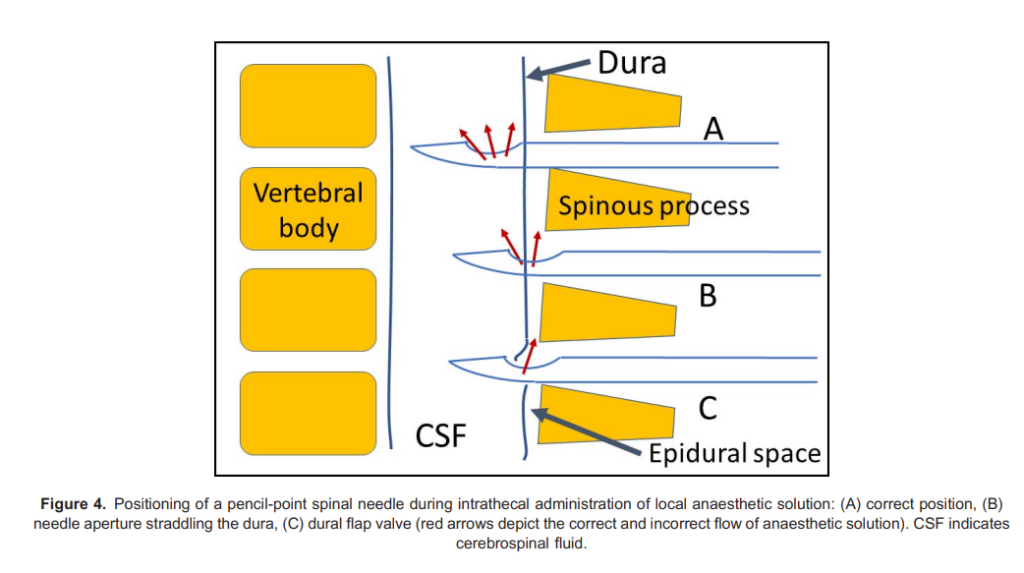
* Flap valve caused by the aperture in the pencil-point needle straddling the dura, or a dural tag; if there is no (or very slow) flow of cerebrospinal fluid in the spinal needle, gentle and slight rotation of the spinal needle may move the dural tag and allow good flow of cerebrospinal fluid (Figure 4) - Drug error:
* Incorrect intrathecal drug administration, which can be disastrous; for example Patel et al20 identified 21 reported cases of intrathecal tranexamic acid injection, of which 20 resulted in life-threatening conditions and 10 were fatal - Equipment:
* Blocked spinal needle, resulting in a dry tap despite the needle tip entering the subarachnoid space
Epidural or CSE Techniques for CD
A CSE technique is recommended when there are indications that the duration of surgery may be extended (eg multiple previous CDs, prior abdominal surgery with extensive adhesions, or morbid obesity), and it can be performed with a needle-through-needle technique, or with 2 separate needle punctures.
In patients with significant cardiovascular (eg severe aortic or mitral stenosis) or respiratory insufficiency, an epidural or CSE technique with a reduced spinal dose of bupivacaine (but with standard doses of intrathecal opioids, eg fentanyl and preservative-free morphine) can be beneficial as the decrease in systemic vascular resistance can be less compared with
standard spinal doses. Titration with incremental dosing of local anaesthetic solution via the epidural catheter allows the desired block level to be established gradually and can avoid both a low block and high block.
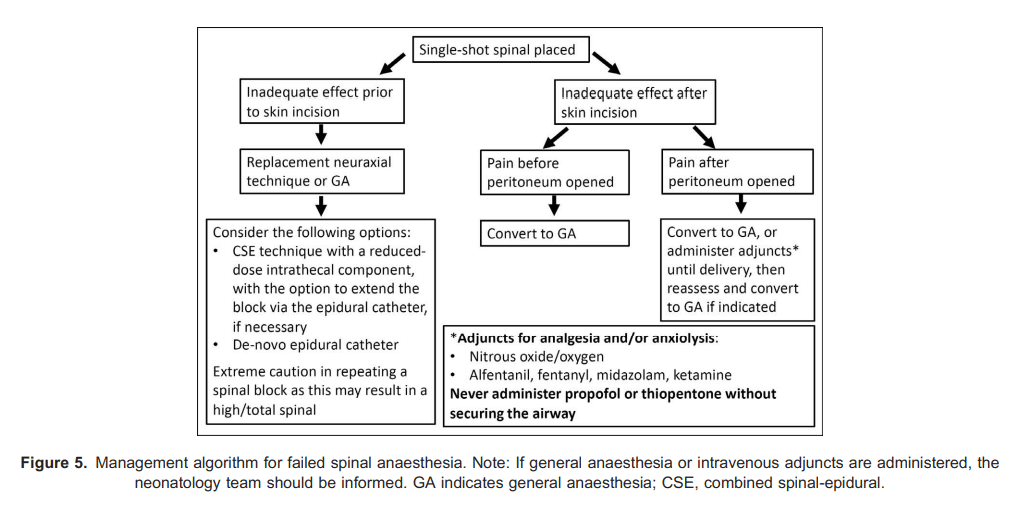
When CD is required in a patient with an epidural catheter in situ (eg intrapartum CD), the epidural catheter can be dosed with a fast-acting local anaesthetic solution such as 2% lidocaine with 1:200 000 adrenaline, with or without fentanyl and/or sodium bicarbonate (with epidural preservative-free morphine (eg 2-3 mg) added postdelivery for postoperative analgesia).21,22 The majority of epidural catheters placed for labour can be successfully ‘topped up’ for CD; however, a poorly functioning labour epidural catheter with increased requirement for supplemental boluses during labour suggests a high probability of failure to obtain adequate surgical anaesthesia for CD.23 This underlines the importance of monitoring the efficacy of a labour epidural catheter and replacing it early, if indicated. In a failed epidural catheter situation, options include replacement with a CSE technique or insertion of a replacement epidural catheter. A standard spinal dose should be avoided as it can result in a high or total spinal if it is administered when the epidural space already contains fluid (eg local anaesthetic solution) due to pressure exerted on the intrathecal sac from the epidural space. Alternatively, depending on the maternal/foetal status, GA may be indicated in the event of a failed NA technique (Figure 5).
GENERAL ANAESTHESIA FOR CD
Indications for GA include emergent delivery, contraindications to NA including maternal refusal, coagulopathy, sepsis, severe hypovolaemia, or intraoperative conversion if the patient is experiencing pain under NA.
The Maternal Airway
The maternal airway may be unexpectedly difficult; factors contributing to this include the following:
- Oedema and increased vascularity of airway mucosa, exacerbated by labour, oxytocin, intravenous (IV) fluids, preeclampsia and Valsalva manoeuvres during labour
- Reduced time to desaturation following apnoea secondary to reduced functional residual capacity and increased oxygen consumption
- Increased risk of oesophageal regurgitation and pulmonary aspiration due to decreased lower oesophageal sphincter tone
- Situational pressure to expedite delivery
- Remote locations of obstetric units, where experienced help or additional personnel are not immediately available.
One study found the incidence of failed intubation in obstetric patients to be 2.3 per 1000 general anaesthetics (1:443).24 The Obstetric Anaesthetists Association and Difficult Airway Society guidelines recommend the following in the management of obstetric airways:25
- Thorough preoperative evaluation, including airway assessment
- Antacid prophylaxis with H2 blocker (eg famotidine), prokinetic (eg metoclopramide), and nonparticulate antacid (eg sodium citrate)
- Preoxygenation for 3 minutes in the ramped position with left uterine displacement (a 208-308 head-up tilt increases the functional residual capacity and apnoea time, and facilitates laryngoscopy in obese patients)
- Rapid sequence induction with cricoid pressure using thiopentone 3 to 5 mg/kg IV or propofol 2 to 2.5 mg/kg IV for induction, succinylcholine 1 to 1.5 mg/kg IV, or rocuronium 1 mg/kg IV for neuromuscular blockade, and intubation with a 6.0 to 7.0 endotracheal tube
Recent advances in airway management include videolaryngoscopy, transnasal high-flow oxygenation which extends safe apnoea time, and second-generation supraglottic airway devices. Local protocols for difficult airway management should be available and adapted to available resources.
The hypertensive response to laryngoscopy in patients with preeclampsia, eclampsia, or raised intracranial pressure can result in cerebrovascular or cardiovascular complications. Several preventive strategies include IV administration of beta-blockers (e.g. labetalol or esmolol), vasodilators (e.g. hydralazine, nitroglycerine, or nicardipine), opioids (e.g. fentanyl, alfentanil, or remifentanil), lignocaine, and magnesium during induction prior to laryngoscopy. However, hydralazine, lignocaine, magnesium bolus and calcium channel blockers (other than nicardipine) are not recommended for this purpose because of potential side effects or lack of demonstrated efficacy. Authors of a focused review conclude that it may be a combination of drugs from different classes that leads to optimal hemodynamic stability.26 Given the favourable pharmacologic profiles, wide availability, and predictability with few reports of serious maternal or foetal effects, esmolol 1.5 mg/kg IV or nitroglycerine 2 µg/kg IV,
combined with propofol 2 mg/kg IV are recommended depending on maternal hemodynamic variables at the time of induction of anaesthesia.26 Labetalol and remifentanil are reasonable alternatives.
Accidental Awareness Under GA
Accidental awareness under GA during CD is 30 times more common than for all anaesthetics, with almost half of episodes occurring between induction and the start of surgery.27 Predisposing factors for accidental awareness under GA included rapid sequence induction, use of thiopentone and immediate administration of neuromuscular blockers without confirming loss of consciousness, obesity, and omission of opioids at induction.25 A prolonged induction-intubation time may result in awareness if additional doses of induction agent are not administered. After induction of anaesthesia, an adequate alveolar concentration of volatile agent should be established before the effect of the induction agent has worn off.
Extubation
Attention to detail at the time of extubation is equally as important as intubation. Three out of 6 anaesthetic-related deaths in a national maternal mortality report were due to postoperative respiratory failure; 2 of these patients had CD under GA and the third patient was under spinal anaesthesia.28 In another report, 5 anaesthesia-related maternal deaths were attributed to airway obstruction or hypoventilation that occurred during emergence and recovery.29 Morbid obesity, inadequate postoperative monitoring, and care pathways were major contributing factors in both of these reports. Thus, emergence and extubation are critical periods because of the risk of regurgitation, vomiting, pulmonary aspiration, hypoventilation, and airway obstruction; therefore obstetric patients in particular should be fully awake, breathing spontaneously on 100% oxygen and able to protect their airway prior to extubation.
POSTOPERATIVE ANALGESIA
Multimodal analgesia should be utilised and include regular administration of acetaminophen in combination with nonsteroidal anti-inflammatory drugs such as diclofenac or ibuprofen unless contraindicated, with opioids available as required. Transversus abdominis plane blocks are effective in cases where long-acting neuraxial opioids have not been administered (eg GA CDs in patients without an [or with an ineffective] epidural catheter); however, they only affect somatic sensation so supplemental IV opioids are required to provide analgesia for visceral involvement. Infiltration of the incision site with long-acting local anaesthetic solution can also be an effective addition to a multimodal regimen.30
MANAGEMENT OF EMERGENT CD
- GA with a rapid sequence induction technique and cricoid pressure is potentially indicated for a category 1 CD as this is likely the quickest method to facilitate safe delivery.
- A technique referred to as a rapid-sequence spinal has been described when spinal anaesthesia is performed and surgical anaesthesia attained within 6 to 8 minutes.31 Components of this rapid-sequence spinal technique include the following:
* Delegate activities (IV cannulation and applying patient monitors)
* Use no-touch technique for spinal needle insertion (mask, hat, and sterile gloves, without sterile gown), single wipe of chlorhexidine solution and drying with swab for skin preparation
* Simplify spinal drug preparation by increasing the dose of hyperbaric bupivacaine and omitting opioids
* Make only 1 attempt at spinal placement unless obvious correction allows a second attempt
* Commence surgery on a rising block (≥T10)
* Be prepared to administer immediate GA if the spinal is inadequate - For patients with a functioning epidural catheter, dosing with a fast-acting local anaesthetic solution such as 2% lidocaine with 1:200 000 adrenaline (with or without 75-100 µg fentanyl) has a mean time to be ready for skin incision of approximately 10 minutes.21 In an emergency, the top-up can be initiated in the delivery suite with the anaesthetist directly observing and monitoring the patient. Addition of 8.4% preservative-free sodium bicarbonate further reduces the onset time.22
PREVENTION AND MANAGEMENT OF SPINAL-INDUCED HYPOTENSION
Aortocaval compression in the supine position can have significant haemodynamic effects in mid-to-late pregnancy but the majority of patients compensate with an increase in collateral blood flow and peripheral vasoconstriction. Autonomic (sympathetic) blockade secondary to NA can lead to severe spinal-induced hypotension.
Prevention of spinal-induced hypotension is important because placental perfusion is dependent on maternal blood pressure, and low Apgar scores and foetal acidosis correlate with the severity and duration of hypotension.32 Maternal blood pressure should be measured at 1-minute intervals after administration of NA (until delivery) to identify hypotension, and after delivery if the patient is stable without evidence of haemorrhage, the time interval between blood pressure measurements can be increased to every 3 to 5 minutes.
International Consensus Guidelines for spinal-induced hypotension have made the following recommendations:32
- Blood pressure should be maintained
- 90% of baseline.
- a-Agonist drugs are the most appropriate drugs for prevention and treatment of postspinal hypotension; phenylephrine and metaraminol have been shown to cause less foetal acidosis than ephedrine. It is recommended to start phenylephrine prophylactically as an IV infusion, immediately after the spinal is administered (commence at 25-50 µg/min and titrate to effect).
- If the patient becomes hypotensive with a normal heart rate, a bolus dose of phenylephrine (eg 50-100 µg IV) and an increase in the infusion rate may be required.
- If the patient is hypotensive with a low heart rate, a bolus dose of ephedrine (eg 3-6 mg IV) may be administered; if the heart rate is very low, administer glycopyrrolate (eg 200 µg IV) or atropine.
- Left lateral uterine displacement (by tilting the table or placing a wedge under the right hip) is recommended to relieve aortocaval compression. If there is severe, symptomatic hypotension, manual left displacement of the uterus should be performed in addition to IV fluid boluses and administration of vasopressors.
- Note: patients with preeclampsia usually experience less hypotension and require lower doses of vasopressors following administration of spinal anaesthesia for CD, but the guideline recommends having a phenylephrine infusion drawn up and connected ready to start, if required.
In low-resource settings, mephentermine can be used because of its low cost and availability. Mephentermine is a sympathomimetic agent that is slightly more potent than ephedrine with a similar effect.33
UTEROTONIC DRUGS
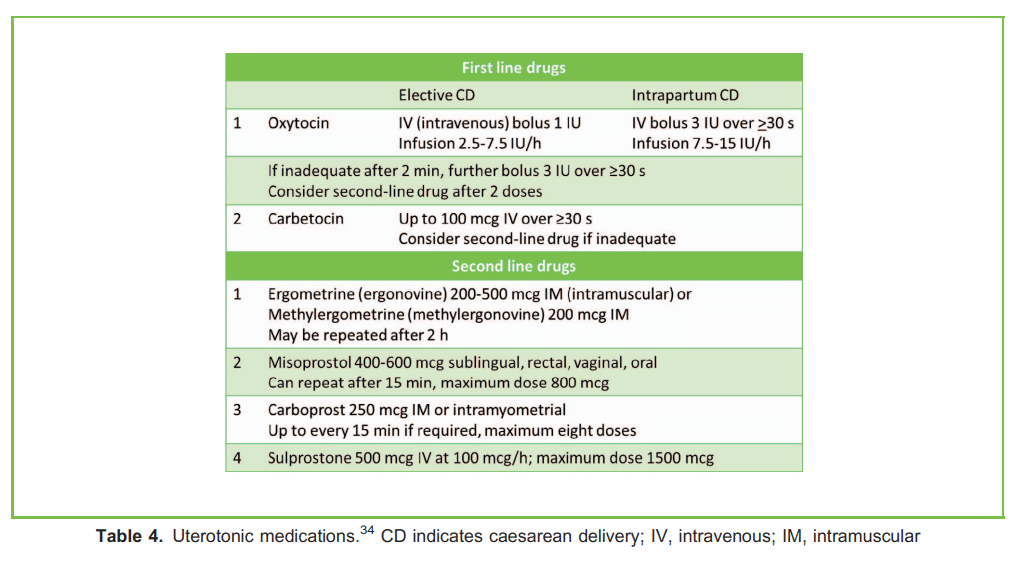
Oxytocin (or carbetocin) is the first-line uterotonic drug to prevent and treat uterine atony and subsequent postpartum haemorrhage. Because of its adverse cardiovascular effects that include hypotension, tachycardia, and pulmonary hypertension, large IV bolus doses of oxytocin are not recommended. Dose requirements during elective CD are lower compared with patients undergoing an intrapartum CD (eg labouring patients who have received an oxytocin infusion during labour). Recent guidelines recommend a small bolus dose immediately after delivery (eg 1 IU for an elective CD, and 3 IU for an intrapartum CD) followed by a titrated IV infusion (eg starting at 2.5 IU/h and 7.5 IU/h respectively).34 Oxytocin (10 IU intramuscularly [IM]) or a fixed dose combination of oxytocin and ergometrine (5 IU/500 µg IM) may be given after delivery in low-resource settings where infusion pumps are not available.35 Carbetocin has a more sustained action and is an alternative uterotonic drug to oxytocin. Second-line uterotonic drugs include methylergonovine, carboprost, and misoprostol, which should be considered if 2 doses of 3 IU of oxytocin are ineffective (see Table 4).34
OTHER CONSIDERATIONS
Although full details of other considerations are beyond the scope of this tutorial, the Enhanced Recovery after Caesarean protocols aim to improving maternal postoperative recovery, promote maternal-infant bonding, and optimise perioperative outcomes.36
- Preoperative elements: Limit fasting intervals, nonparticulate liquid carbohydrate loading, patient education, lactation/ breastfeeding preparation and support, haemoglobin optimisation36
- Intraoperative elements: Optimise IV fluids, prevent and treat spinal anaesthesia–induced hypotension, maintain normothermia, optimise uterotonic administration, administer antibiotic prophylaxis, administer intra- and postoperative nausea and vomiting prophylaxis and treatment, initiate multimodal analgesia, promote breastfeeding and maternal-infant bonding, delay cord clamping36
- Postoperative elements: Promote early oral intake, glycaemic control, early mobilisation, resting periods, and return of bowel function; remove urinary catheter early; administer venous thromboembolism prophylaxis and multimodal analgesia; facilitate early discharge; remediate anaemia; support breastfeeding36
Summary
CD is one of the commonest surgeries performed globally.1 Surgical and anaesthetic practices should be optimised to achieve the best possible maternal and neonatal outcomes.
NA for CD is associated with lower maternal and neonatal morbidity and mortality compared to GA.5 Small-gauged pencil-point needles (25 gauge or smaller) should be used to minimise the incidence of postdural puncture headache. Prophylactic measures to avoid spinal-induced hypotension are important to maintain utero-placental perfusion and foetal physiology. Opioids added to local anaesthetic solution augment the quality of intraoperative NA and provide postoperative analgesia.
Potential difficulties with airway management and accidental awareness under GA are the main concerns during GA for CD. Extubation and emergence are also critical periods, and appropriate postoperative monitoring and care
pathways should be in place
REFERENCES
1. Weiser TG, Haynes AB, Molina G, et al. Size and distribution of the global volume of surgery in 2012. Bull World Health Organ. 2016;94(3):201-209F.
2. Betran AP, Torloni MR, Zhang JJ, et al for the WHO Working Group on Caesarean Section. WHO statement on caesarean section rates. BJOG. 2016;123(5):667-670.
3. World Health Organization. WHO Statement on Caesarean Section Rates (WHO/RHR/15.02). Geneva: World Health Organization; 2015.
4. Knight M, Bunch K, Tuffnell D, et al. Saving lives, improving mothers’ care— lessons learned to inform maternity care from the UK and Ireland confidential enquiries into maternal deaths and morbidity 2014-16. Oxford, UK: National Perinatal Epidemiology Unit, University of Oxford; 2018:34-41.
5. Kim WH, Hur M, Park SK, et al. Comparison between general, spinal, epidural, and combined spinal-epidural anesthesia for cesarean delivery: a network meta-analysis. Int J Obstet Anesth. 2019;37:5-15.
6. Royal College of Obstetricians and Gynaecologists. Classification of urgency of caesarean section—a continuum of risk. Good Practice Guidance No 11. Accessed October 12, 2019. https://www.rcog.org.uk/globalassets/documents/guidelines/goodpractice11classificationofurgency.pdf
7. Hansen AK, Wisborg K, Uldbjerg N, et al. Risk of respiratory morbidity in term infants delivered by elective caesarean section: cohort study. BMJ 2008;336(7635):85-87.
8. Roberts CL, Nicholl MC, Algert CS, et al. Rate of spontaneous onset of labour before planned repeat caesarean section at term. BMC Pregnancy Childbirth, 2014;14:125.
9. Siddiqui N, Friedman Z, McGeer A, et al. Optimal hand washing technique to minimize bacterial contamination before neuraxial anesthesia: a randomized control trial. Int J Obstet Anesth. 2017;29:39-44.
10. Campbell JP, Plaat F, Checketts MR, et al. Safety guideline: skin antisepsis for central neuraxial blockade. Anaesthesia. 2014;69(11):1279-1286.
11. Broadbent CR, Maxwell WB, Ferrie R, Wilson DJ, Gawne-Cain M, Russell R. Ability of anaesthetists to identify a marked lumbar interspace. Anaesthesia. 2000;55(11):1122-1126.
12. Locks Gde F, Almeida MC, Pereira AA. Use of the ultrasound to determine the level of lumbar puncture in pregnant women. Rev Bras Anestesiol. 2010;60(1):13-19.
13. Carvalho B, Durbin M, Drover DR, Cohen SE, Ginosar Y, Riley ET. The ED50 and ED95 of intrathecal isobaric bupivacaine with opioids for cesarean delivery. Anesthesiology. 2005; 103(3):606-612. 14. Sng BL, Han NL, Leong WL, et al. Hyperbaric vs. isobaric bupivacaine for spinal anaesthesia for elective caesarean section: a Cochrane systematic review. Anaesthesia. 2018;73(4):499-511.
15. Ali MA, Ismail S, Sohaib M, Aman A. A double-blind randomized control trial to compare the effect of varying doses of intrathecal fentanyl on clinical efficacy and side effects in parturients undergoing cesarean section. J Anaesthesiol Clin Pharmacol. 2018;34(2):221-226.
16. Bang YS, Chung KH, Lee JH, et al. Comparison of clinical effects according to the dosage of sufentanil added to 0.5% hyperbaric bupivacaine for spinal anesthesia in patients undergoing cesarean section. Korean J Anesthesiol. 2012;63(4):321-326.
17. Cowan CM, Kendall JB, Barclay PM, et al. Comparison of intrathecal fentanyl and diamorphine in addition to bupivacaine for caesarean section under spinal anaesthesia. Br J Anaesth. 2002;89(3):452-458.
18. Sultan P, Halpern SH, Pushpanathan E, et al. The effect of intrathecal morphine dose on outcomes after elective cesarean delivery: a meta-analysis. Anesth Analg. 2016;123(1):154-164.
19. Fettes PDW, Jansson J-R, Wildsmith JAW. Failed spinal anaesthesia: mechanisms, management, and prevention. Br J Anaesth. 2009;102(6):739-748.
20. Patel S, Robertson B, McConachie I. Catastrophic drug errors involving tranexamic acid administered during spinal anaesthesia. Anesthesia. 2019;74(7):904-914.
21. Hillyard SG, Bate TE, Corcoran TB, et al. Extending epidural analgesia for emergency caesarean section: a meta-analysis. Br J Anaesth. 2011;107(5):668-678.
22. Lam DT, Ngan Kee WD, Khaw KS. Extension of epidural blockade in labour for emergency Caesarean section using 2% lidocaine with epinephrine and fentanyl, with or without alkalinisation. Anaesthesia. 2001;56(8):790-794.
23. Riley ET, Papasin J. Epidural catheter function during labor predicts anesthetic efficacy for subsequent cesarean delivery. Int J Obstet Anesth. 2002;11(2):81-84.
24. Kinsella SM, Winton AL, Mushambi MC, et al. Failed tracheal intubation during obstetric general anaesthesia: a literature review. Int J Obstet Anesth. 2015;24(4):356-374.
25. Mushambi MC, Kinsella SM, Popat M, et al. Obstetric Anaesthetists’ Association and Difficult Airway Society guidelines for the management of difficult and failed tracheal intubation in obstetrics. Anaesthesia. 2015;70(11):1286-1306.
26. Pant M, Fong R, Scavone B. Prevention of peri-induction hypertension in preeclamptic patients: a focused review. Anesth Analg. 2014;119(6):1350-1356.
27. Pandit JJ, Andrade J, Bogod DG, et al.; Royal College of Anaesthetists and the Association of Anaesthetists of Great Britain and Ireland. The 5th National Audit Project (NAP5) on accidental awareness during general anaesthesia: summary of main findings and risk factors. Anaesthesia. 2014;69(10):1089-1101.
28. Lewis G, ed. The Confidential Enquiry into Maternal and Child Health (CEMACH). Saving Mothers’ Lives: Reviewing Maternal Deaths to Make Motherhood Safer: 2003-2005. The Seventh Report on Confidential Enquiries into Maternal Deaths in the United Kingdom. London: CEMACH; 2007.
29. Mhyre JM, Riesner MN, Polley LS, Naughton NN. A series of anesthesia-related maternal deaths in Michigan, 1985-2003. Anesthesiology. 2007;106(6):1096-1104.
30. Kerai S, Saxena KN, Taneja B. Post-caesarean analgesia: what is new? Indian J Anaesth. 2017;61(3):200-214.
31. Kinsella SM, Girgirah K, Scrutton MJ. Rapid sequence spinal anaesthesia for category-1 urgency caesarean section: a case series. Anaesthesia. 2010;65(7):664-669.
32. Kinsella SM, Carvalho B, Dyer RA, et al. International consensus statement on the management of hypotension with vasopressors during caesarean section under spinal anaesthesia. Anaesthesia. 2018;73(1):71-92.
33. Mitra JK, Roy J, Bhattacharyya P, et al. Changing trends in the management of hypotension following spinal anesthesia in cesarean section. J Postgrad Med. 2013;59(2):121-126.
34. Heesen M, Carvalho B, Carvalho JC, et al. International consensus statement on the use of uterotonic agents during caesarean section. Anaesthesia. 2019;74(10):1305-1319.
35. World Health Organization, WHO Recommendations: Uterotonics for the Prevention of Postpartum Haemorrhage. Geneva: World Health Organization; 2018. Accessed January 15, 2021. https://apps.who.int/iris/bitstream/handle/10665/277276/9789241550420-eng.pdf?ua¼1&ua¼1
36. Bollag L, Lim G, Sultan P, et al. Society for Obstetric Anesthesia and Perinatology: Consensus Statement and Recommendations for Enhanced Recovery After Cesarean. Anesth Analg. 2021 May 1;132(5):1362-1377



