General Topics
Case study and questions
Before reading the tutorial read the following case study and try to answer the questions. The answers can be found within the body of text.
A 66 year old man presents for carotid endarterectomy (CEA). He suffered an embolic stroke six weeks ago. Carotid duplex scanning has demonstrated that the left internal carotid artery is 95% occluded.
- A 66 year old man presents for carotid endarterectomy (CEA). He suffered an embolic stroke six weeks ago. Carotid duplex scanning has demonstrated that the left internal carotid artery is 95% occluded.
- Which systems will you examine?
- The patient asks you if there are any risks of the operation. What do you tell him?
- Should surgery be performed so soon after a stroke?
Past medical history reveals well controlled chronic obstructive pulmonary disease(COPD) and hypertension. He denies angina or symptoms of peripheral vascular disease. His hypertension is well controlled on atenolol, slow-release nifedipine and frusemide. He is also taking aspirin. He has made a complete recovery from his recent stroke. You discuss surgery under both general and local anaesthesia. The patient decides that he would like to remain awake.
What options are available for anaesthetising the surgical field for carotid endarterectomy? What are the side effects and complications of these?
Should he take all his usual medications on the morning of surgery?
Would you prescribe any sedative premedication?
Is routine invasive arterial blood pressure monitoring justified?
The patient arrives in the operating suite and after establishing monitoring you perform superficial and deep cervical plexus blocks.
What are the anatomical landmarks for these techniques?
Describe how you would perform a deep cervical plexus block.
Surgery commences and after about fifteen minutes the patient complains of pain in his jaw.
What would you do?
On application of the carotid cross clamps the patient loses consciousness and begins to obstruct his airway.
How would you manage the situation?
Following appropriate treatment, the patient regains consciousness and surgery continues uneventfully. You are called to recovery half an hour after completion of surgery as the patient is complaining of a headache. His blood pressure is 195/110.
- Would you treat his hypertension?
- If he became drowsy what investigations would you request?
The headache resolves and just as he is about to be transferred to the ward, he complains of difficulty breathing. On examining him, you notice a large swelling beneath the incision.
- What is your immediate management?
- How will you manage his airway for surgical re-exploration?
Background
Atherosclerotic disease is widespread in the western population, with increasing prevalence in the developing world. Risk factors include familial predisposition, smoking, diabetes, hypercholesterolaemia and hypertension.
Surgery to relieve atherosclerotic obstruction of the carotid arteries was first performed at St. Mary’s Hospital, London, in 1954. Since then, evidence for its effectiveness has accumulated (CEA is the most studied operation in surgical history). In 2003 nearly 140,000 carotid endarterectomies were performed in the USA (Halm).
The aim of CEA is to prevent embolic stroke. As with any prophylactic operation, careful evaluation of the relative benefits and risks of the procedure is required. Peri-operative combined mortality and major stroke risk is 2 – 5%. This is lowest for asymptomatic disease, intermediate for TIA and highest for completed stroke.
The North American Symptomatic Carotid Endarterectomy Trial (NASCET) and the European Carotid Surgery Trial (ECST) are both large randomized class 1 studies which have defined current indications for carotid endarterectomy. NASCET found that for every six patients treated with CEA, one major stroke would be prevented at two years (i.e. a “number needed to treat” (NNT) of six) for symptomatic patients with a 70 – 99% stenosis. Symptomatic patients with less severe carotid occlusion (50 – 69%) had a smaller benefit, with a NNT of 22 at five years (Barclay). In addition, co-morbidity adversely affects the outcome; patients with multiple medical problems have a higher post-operative mortality and hence benefit less from the procedure. The European asymptomatic carotid surgery trial (ACST) found that asymptomatic patients may also benefit from the procedure, but only the group with a high grade stenosis (greater than 80% stenosis in men; unclear in women). For maximum benefit patients should be operated on soon after a TIA or stroke, preferably within the first month.
Depending on definition and rigorousness of assessment, the reported peri-operative stroke rate can be as high as 35% (Rothwell). The majority of peri-operative strokes are caused by factors outside of the anaesthetist’s control such as intra-operative embolism and thrombosis at the endarterectomy site (Jacobowitz). Twenty per-cent of peri-operative strokes are due to haemodynamic failure, a factor often readily manipulated by the anaesthetist (Whitney).
CEA necessitates a period of carotid artery cross clamping. This may cause cerebral ischaemia if collateral blood flow via the circle of Willis is inadequate. In such patients cerebral perfusion is maintained by the use of a surgically inserted shunt to bypass the isolated section of carotid artery. The use of a shunt is associated with increased risk of embolic phenomena and arterial dissection.
Avoidance of cerebral ischaemia is essential. Various techniques are available for monitoring cerebral perfusion. Clinical observation of an awake patient is the gold standard monitor of cerebral function. In patients receiving general anaesthesia monitoring options include carotid artery stump pressure, middle cerebral artery blood flow trans-cranial Doppler analysis, near infrared spectroscopy, electroencephalography (EEG) and somatosensory evoked potentials. None of these techniques are entirely reliable.
KEY POINTS
- Surgery is performed to reduce future risk of stroke.
- CEA should be performed as soon as possible after a TIA or stroke.
- Vascular patients with high associated co-morbidity.
- High risk surgery; combined death and major stroke risk is 2-5%.
- Carotid cross clamping risks ipsilateral cerebral ischaemia.
- Significant cardiovascular morbidity – use invasive arterial blood pressure monitoring and control blood pressure pharmacologically.
- Awake surgery allows direct neurological monitoring.
- Suitable local anaesthetic techniques include cervical epidural, superficial and deep cervical plexus blocks and local infiltration by surgeon.
- Consider formal neurological monitoring when using general anaesthesia.
- Continue neurological and invasive haemodynamic monitoring into the post-operative period.
- If re-exploration is necessary this should be done under local anaesthesia
Local versus general anaesthesia for CEA.
A Cochrane review in December 2003 found only limited evidence from randomized controlled trials (seven studies, 554 patients). This showed reduction in incidence of post-operative wound haematoma with local anaesthesia (no difference in mortality or stroke risk). However, these were small, under-powered studies. Meta analysis of non-randomized studies (with questionable methodology) suggests reduction in death, stroke, myocardial infarction and respiratory complications (Rerkasem).
Perceived advantages of an “awake” technique are:
- Gold standard cerebral function monitoring
- Lower shunt insertion rate
- Improved haemodynamic stability
- Earlier detection and treatment of complications
- Intact cerebral autoregulation
- Lower re-exploration rate
- Less post-operative anxiety for surgeon and anaesthetist
- Less postoperative pain
- Useful for patients in whom general anaesthesia is undesirable, such as those awaiting heart valve surgery or coronary artery bypass grafting.
- Shorter hospital stay
However, CEA is currently performed routinely under general anaesthesia in many centres with excellent results. Proponents of this technique argue its advantages including tight arterial carbon dioxide control, cerebral protection afforded by volatile anaesthesia and avoidance of the need for emergency airway control if the patient loses consciousness. It has also been argued that some surgeons feel more comfortable operating on an anaesthetised patient. A UK based multi-centre randomized controlled trial comparing general with local anaesthesia for carotid surgery (the “GALA” trial) is approaching its target of 3000 patients. When published, its findings will hopefully provide more reliable guidance as to the optimal anaesthetic technique.
Local anaesthetic techniques
Blockade of the second, third and possibly fourth cervical dermatomes is required for surgery. This can be achieved by various techniques; cervical epidural, superficial and deep cervical plexus blocks (alone or in combination) and local infiltration by the surgeon. There have been no differences in outcome demonstrated between these techniques.
Cervical epidural
Although this technique is not familiar to most anaesthetists, it has found favour in a few areas, in particular France. A midline approach using the “hanging drop” method may be used followed by insertion of an epidural catheter, test dose and then 10 – 15ml of 0.5% bupivacaine with or without fentanyl 50 – 100mcg. However, this approach risks significant and frequent side effects and complications. Commonly observed problems are hypotension, bradycardia and alterations in respiratory function. The reported dural puncture rate is 0.5% and respiratory failure requiring intubation occurs in around 1%. These risks are unacceptable when safer techniques are available.
Superficial cervical plexus block
The four cutaneous branches of the cervical plexus emerge from behind the sternocleidomastoid muscle (SCM) at its mid-point (Erb’s point).
These nerves supply the area from the clavicle to the jaw line anteriorly and to the mastoid process posteriorly. Classical superficial cervical plexus block involves subcutaneous infiltration of 5-10ml of local anaesthetic along the posterior border of SCM around its midpoint. A modified or intermediate cervical plexus block involves puncturing the investing fascial layer before 10-20ml of local anaesthetic is injected.
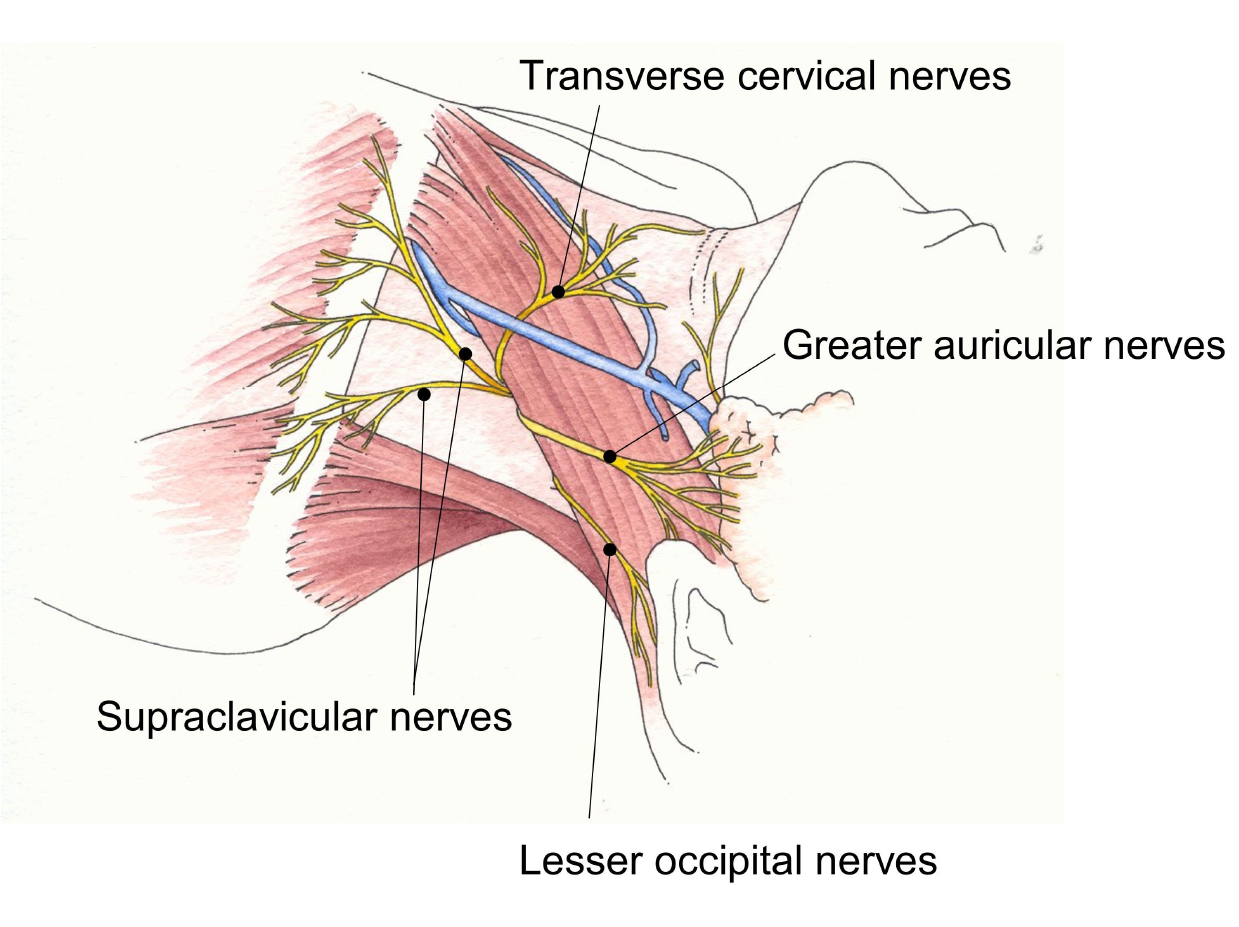
Conventional teaching is that local anaesthetic injected deep to the investing fascia of the neck cannot block the roots of the cervical plexus due to the impenetrable layer of deep cervical fascia. However, a recent cadaveric study demonstrated that an injection of methylene blue deep to the investing fascia did reach the nerve roots (JJ Pandit). This supports the observation that carotid surgery can be performed with the intermediate cervical plexus alone.
Deep cervical plexus block
The classical approach to a deep cervical plexus block (DCPB) was to perform separate injections at C2, C3 and C4. The single injection technique, described by Winnie, is simpler and equally effective. The aim is to deposit local anaesthetic on the roots of the cervical plexus. The interscalene groove is identified at the level of the superior curnu of the thyroid cartilage (C4) by moving the fingers laterally from sternocleidomastoid. A 25-50mm short beveled regional block needle is inserted medially, caudally and dorsally toward the contralateral elbow. This angulation minimizes the risk of needle advancement between the transverse processes of the cervical vertebrae and inadvertent vertebral artery or intrathecal injection. The needle should be advanced until contact with bone (transverse process of C4) is felt and 10ml of local anaesthetic injected after careful aspiration. The plexus is superficial, often only 10-20mm deep to the skin.
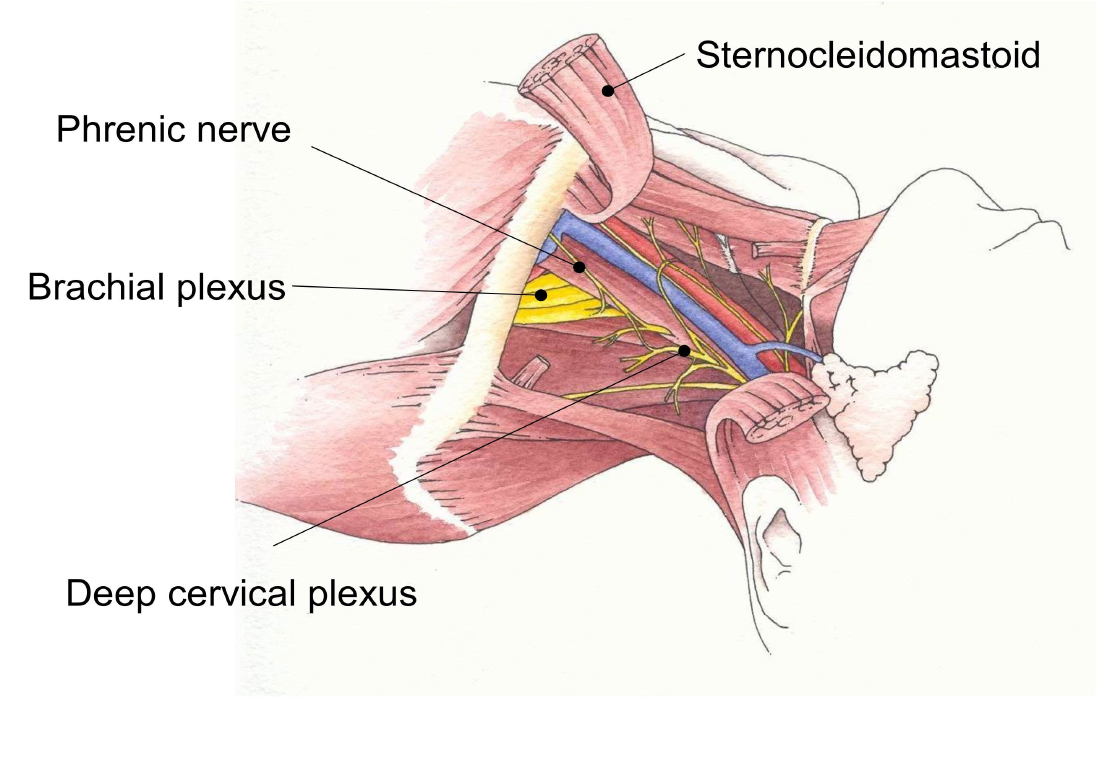 The phrenic nerve, recurrent laryngeal nerve and stellate ganglion are also commonly blocked by a deep cervical plexus block. It is contra-indicated in patients with severe respiratory disease.
The phrenic nerve, recurrent laryngeal nerve and stellate ganglion are also commonly blocked by a deep cervical plexus block. It is contra-indicated in patients with severe respiratory disease.
The mandibular branch of the trigeminal nerve sometimes contributes to innervation of the surgical site, particularly in patients with a high carotid bifurcation. This can result in discomfort from the retractor placed under the jaw. Local anaesthetic infiltration subcutaneously along the jaw line or an inferior alveolar nerve block have been shown to help in these patients. More than 50% of patients will require direct supplementation of local anaesthesia by the surgeon around the carotid sheath despite cervical plexus block. This is due to pain fibres running with the sympathetic nerves which are not always covered by cervical plexus block.
Local anaesthetic infiltration
The operation can be performed under local anaesthetic infiltration by the surgeon.
Pre-operative assessment
- Careful assessment of cardiovascular co-morbidity is required although functional cardiovascular investigations are not usually required. Blood pressure must be adequately controlled pre-operatively. The blood pressure must be documented.
- Assess suitability for surgery under local anaesthesia. Contra-indications include patient refusal and movement disorders such as Parkinson’s disease. Assess the patient’s ability to lie in a semi-recumbent position for two to three hours.
- Enquire about any respiratory impairment, including exercise tolerance. The phrenic nerve is almost always blocked when performing a deep cervical plexus block, which may precipitate respiratory failure in those with precarious lung function. DCPB should not be performed on such individuals.
- Contralateral recurrent laryngeal nerve palsy should also be identified (hoarse voice, history of thyroid surgery) as this nerve is susceptible to surgical damage and may be blocked with local anaesthetic infiltrated by the surgeon. Complete airway obstruction may result from bilateral palsy.
- Make a note of anti-anginal, anti-hypertensive and respiratory medication.
- All patients should be taking a statin and aspirin unless contra-indicated.
- A careful explanation of the procedure including neurological monitoring will help reduce anxiety and improve compliance.
- Warn the patient they may feel some intra-operative discomfort such as toothache or earache. An analogy to dental surgery under local anaesthetic is often helpful.
- Document pre-existing neurological deficits meticulously so that new ones may more easily be assessed.
- 12 lead ECG, full blood count, urea & electrolytes and group & save should be performed.
- Omit diuretic on the morning of surgery unless large doses are taken for heart failure. This may require the insertion of a urinary catheter.
- Patients taking clopidogrel require careful consideration. Its use is associated with increased incidence of post-operative wound haematoma. However, if discontinued peri-operatively the risk of thromboembolic coronary and cerebral events is increased. One option is to continue clopidogrel and be prepared to treat bleeding (for example aprotinin and platelet transfusion).
- Maintain all other cardiac medication including other anti-platelet drugs such as aspirin and dipyridamole. Sedative pre-medication is generally avoided as it hinders neurological assessment.
- Ensure tight glycaemic control in diabetic patients. Hyperglycaemia may worsen any neurological deficit.
- Allow clear oral fluids up to two hours pre-operatively and ensure the bladder has been emptied prior to leaving the ward.
Per-operative
- Minimum monitoring should include pulse oximetry, ECG, invasive blood pressure monitoring. If available, five-lead ECG is more sensitive at detecting myocardial ischaemia.
- The arterial cannula should be sited in the contralateral arm if possible.
- Insert one IV cannula for fluid and one for infusions (e.g. sedation)
- Maintenance of cardiovascular stability is paramount. Aim for a mean arterial pressure at baseline or slightly higher during cross clamping. Vasoconstrictors such as metaraminol (eg 0.5mg boluses) or phenylephrine (infuse at 1-6mg/hr) and anti-hypertensives such as labetalol (5mg boluses or continuous infusion at 20-160mg/hour) or GTN (infuse at x-xmcg.Kg-1.min-1) should be available to correct haemodynamic perturbations.
Management of the “awake” carotid:
- Sedation is useful for insertion of lines, siting blocks and during initial dissection and mobilisation of the carotid artery. A target controlled infusion of propofol is ideal. Discontinue sedation in good time prior to carotid cross clamping so that the patient is alert enough to co-operate with neurological monitoring.
- Perform local anaesthetic blocks as described above.
- Oxygen should be administered via nasal cannulae or a nasal sponge.
- Empty the bladder pre-operatively. A full bladder intra-operatively can cause discomfort and agitation. Intravenous fluid is rarely required.
- Use non-claustrophobic drapes to separate the surgical field from the patient’s face.
- An experienced member of staff should be used to monitor and reassure the patient. This is a vital role to detect neurological deficit at the earliest possible opportunity. Monitoring should consist of assessment of contralateral motor function (grip strength), speech and cognition (regular questioning). Neurological deficit may either be immediate following cross clamping or subtle and insidious, perhaps related to relative in intra-operative hypotension.
- I.V. unfractionated heparin (3000 – 5000iu) should be given before cross clamping of the carotid artery. Reversal of anticoagulation with protamine is generally avoided as it is associated with increased stroke rates.
- A slow rise in arterial blood pressure is frequently observed during carotid artery cross clamping.
- If neurological deficit occurs, consider pharmacological augmentation of blood pressure if the patient is hypotensive.
- Ensure that the airway is unobstructed and increase the FiO2
- If neurological recovery does not follow, a surgical shunt should be inserted immediately (10% of cases).
- Several types of shunt are available including Javid and Pruitt-Inahara varieties. The latter is held in place with inflatable balloons and has a side port which allows shunt pressure to be transduced.
- Intra-operative conversion to general anaesthesia is rare but may be necessary to maintain the airway if neurological recovery does not result from shunt insertion. Target controlled propofol infusion and insertion of a laryngeal mask airway is an ideal technique.

Management of CEA under general anaesthesia:
- Invasive arterial blood pressure monitoring is mandatory. A balanced general anaesthetic is used. Attempt to maintain the blood pressure close to baseline values.
- Tracheal intubation gives a reliable airway but a Proseal™ laryngeal mask airway may also be used in selected patients.
- Positive pressure ventilation is be used to ensure control of arterial carbon dioxide tension.
- A superficial cervical plexus block will reduce intra-operative opiate requirement and provide post-operative analgesia.
- Remifentanil infusion provides ideal titratable anaesthetic conditions with rapid emergence allowing prompt assessment of neurological function upon awakening.
Post-operative
- Arrangements should be made for extended (4-6 hours) post-operative observation in a suitable environment with continued close invasive haemodynamic and neurological monitoring.
- Any patient developing post-operative neurological deficit needs immediate surgical assessment. This should include prompt assessment of carotid artery patency with carotid artery duplex scanning to help assess the cause of neurological deterioration and suitability for re-exploration.
- If carotid duplex scanning is unavailable, the patient should be assumed to have developed carotid artery occlusion and taken back to theatre without delay.
- Oxygen should be administered overnight.
- Post operative hypertension is common. Treat any pain and ensure that a full bladder is not the cause. Blood pressure should be controlled to below preoperative levels (the cerebral vasculature becomes exposed to increased pressure and flow post-operatively). Use short acting anti-hypertensive agents such as labetolol (5mg increments up to 100mg maximum).
- Uncontrolled hypertension may result in hyperperfusion syndrome. This occurs because in the pre-operative state, the cerebral circulation has adapted to a significant carotid stenosis by maximal dilatation and loss of autoregulation. Following restoration of carotid artery patency, the abnormal vasculature is susceptible to damage from excessive flow and pressure. Hyperperfusion syndrome may be manifest as an occipitofrontal headache which may progress to seizures and cerebral haemorrhage. This may be fatal.
- Bleeding causing wound haematoma occurs in 5% of cases. Occasionally this may cause progressive airway obstruction. Supra-glottic oedema due to venous congestion is common and may make visualisation of the larynx difficult.
- In the event of a return to theatre, re-exploration of the wound should be performed under local anaesthesia if at all possible. The original regional block will still be working and should be supplemented with local infiltration by the surgeon if necessary. Airway obstruction will be rapidly alleviated by removal of skin staples/sutures.
- Most patients are discharged the day after surgery.
Summary
Anaesthesia for carotid endarterectomy is challenging and rewarding. Firm evidence for the best anaesthetic technique has yet to be published. Whether local or general anaesthesia is employed both anaesthetic and surgical expertise have a significant effect on outcome. Practice makes perfect!
Acknowledgements
Thank you to Dr. Alice Roberts for the use of her illustrations.
Further reading
- New York school of regional anaesthesia website – http://www.nysora.com/techniques/
References
- Barclay, L. AAN Updates Guidelines on Carotid Endarterectomy Neurology. 2005;65:794-801
- Halm, MR. et al, Revisiting the appropriateness of carotid endarterectomy.Stroke 2003;34(6):1464-1471
- Jacobowitz GR et al. Causes of Perioperative Stroke after Carotid Endarterectomy: Special Considerations in Symptomatic Patients Ann Vasc Surg. 2001;15:19-24
- Kim GE et al. The anatomy of the circle of Willis as a predictive factor for intraoperative cerebral ischemia (shunt need) during CEA. Neurol Res 2002;24:237-40
- Rerkasem K, Bond R, Rothwell PM. Local vs general anaesthesia for carotid endarterectomy. The Cochrane Database of Systematic Reviews 2004, issue 2. Art No CD000126
- Rothwell PM, Warlow CP. Is self-audit reliable? Lancet 1995;346:1623
- Stoneham M, Oxford handbook of Anaesthesia. Oxford University Press. Chapter 16;321-323
- Steneham, MD, Knigton JD. Regional anaesthesia for carotid endarterectomy Br JAnaesth 1999;82: 910-19
- Whitney EG et al. Inadequate cerebral perfusion is an unlikely cause of perioperative stroke Ann Vasc Surg. 1997;11:109-14
- Winnie AP et al. Interscalene cervical plexus block: a single-injection technique Anesth Analg. 1975 May-Jun;54(3):370-5.



