General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found in the body of the text.
- Answer True or False:
a. EVAR is a low risk procedure as it is minimally invasive
b. EVAR carries a lower operative risk than an open AAA repair
c. EVAR is advocated for older, less fit patients and open repairs for younger patients
d. EVAR is associated with fewer long term complications and re-interventions than open AAA repair - Answer True or False:
a. Surgery is considered when the AAA is greater than 5.5cm in diameter
b. EVAR requires yearly radiological surveillance of the graft
c. Significant blood loss can occur with EVAR
d. Emergency EVAR should never be done under LA - Answer True or False:
a. An increasing size of AAA is associated with an increased risk of rupture
b. There is a greater risk of spinal cord ischaemia with infra-renal EVARs compared to supra-renal EVARs
c. Neuraxial block is contra-indicated due to the use of heparin intra-operatively
d. There is a 10% conversion rate to an open procedure - The risk of renal impairment is increased with:
a. The use of carbon dioxide contrast
b. Increasing number of graft fenestrations
c. The use of sodium bicarbonate solution
d. Supra-renal EVAR compared to infra-renal EVAR
INTRODUCTION
Endovascular Aortic Aneursym Repair (EVAR) is increasingly being used for repair of Abodminal Aortic Aneurysms (AAA). It is the most common method of treatment for AAA in the USA (>70% elective AAA repairs) (1). As technology improves and surgeons and interventional radiologists are becoming more experienced, endovascular repair is being increasingly used for more complex juxta- renal and supra-renal AAA repairs, AAA ruptures and for repair of thoracic aneurysm and dissections.
ABDOMINAL AORTIC ANEURYSMS
The normal diameter of the aorta is 2cm and it is considered to be aneurysmal when it reaches 3cm in diameter. The prevalence of AAA in the UK is approximately 4% in males aged 65-74. Risk factors include age >60years, male gender (6 times more common than in females) and smoking (lifelong smokers are at 4 times greater risk than non-smokers). Other risk factors include hypertension, hypercholesterolaemia, family history and inherited connective tissue diseases such as Marfans or Ehlers Danlos (type IV).
The risk of rupture increases with increasing size of the aneurysm:
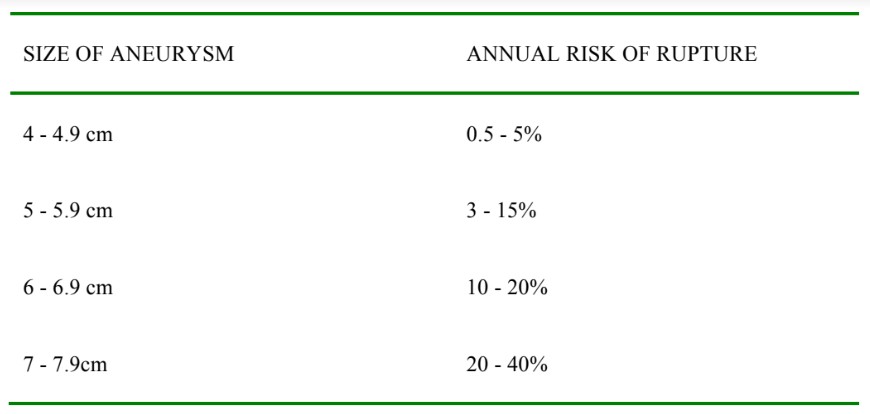
Table 1. Annual risk of rupture with size of aneurysm
With approximately 5% mortality rate following elective open AAA repair, elective surgery is usually only considered in AAA greater than 5.5cm or if is rapidly growing (>5mm in 6 months) (Nice guidelines EVAR 2009)
Surgical Procedure
Technique
A distinction should be made between anaesthesia and surgery for simple infra-renal aneurysm repairs and that for more complex infra-renal, para-renal and suprarenal aneurysms.
Repair of a simple infra-renal AAA is a minimally invasive surgical technique which involves catheterising the femoral or iliac arteries and passing one or more expandable grafts into the abdominal aorta under fluoroscopic guidance. The stent is deployed across the aneurysm thus preventing blood flow into the aneurysmal sac, which will eventually thrombose. Additional trouser grafts can be inserted into the common iliac arteries, overlapping the main graft. Infra-renal aneurysms which have an inadequate infra-renal neck to support the graft can be stabilised by using a stent graft which has a bare metal portion (known as a “crown of thorns”) extending above the renal artery ostia. It has been proven that these bare metal stents do not impede blood flow to the renal arteries.
Juxta-renal and supra-renal aneursyms can be treated with fenestrated grafts which have orifices for the coeliac, superior mesenteric and renal arteries as required. Once in place, further covered stents can be deployed via the orifices into the arteries. These fenestrated grafts are purpose-made, specific to the patient’s anatomy – which is determined by 3D reconstruction CT approximately 6 weeks prior to surgery and therefore they can only be used in elective cases. For more expedient surgery, standard chimney grafts can be inserted into the visceral or renal arteries and a main stent deployed over these into the aorta.

Figure 1. Stent graft with scallop for the superior mesenteric artery and fenestrations for the renal arteries. Crown of thorns extending up from the graft.
In some cases it may be required to insert the stents from above and the surgeons will cannulate a brachial or subclavian artery to achieve this. This possibility should be discussed with the surgeon before deciding on the site for arterial line placement.
The use of more complex grafts and an increasing number of fenestrations increases the skill required from the operator, and can increase the procedural time significantly. The fluoroscopy time and hence the dose of iodinated contrast used is also likely to be higher, increasing the risk of renal impairment. The use of carbon dioxide as a contrast agent is possible in patients with high risk of renal impairment however it carries the risk of localised accumulation and significant gas embolism. It also results in poorer images and requires a higher radiation dose per image so is not used routinely.
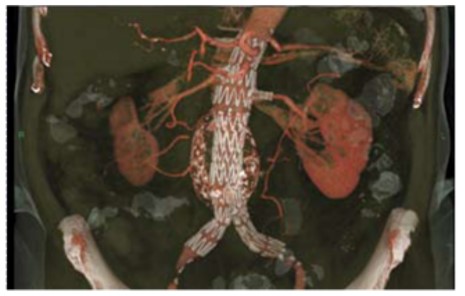
Figure 2. 3D CT reconstruction of complex EVAR graft
Blood Loss
Blood loss during a simple infra-renal EVAR is usually minimal – approximately 200ml, and intraoperative transfusion is rare. Prolonged procedures will have on-going blood loss from the access vessels and complex EVARs can result in significant blood loss. Use of cell salvage is prudent in complex EVARs and consideration should be given to the use of a rapid infusion device in high risk cases. With all EVARs there should be a high index of suspicion for occult blood loss from damage to the aorta, iliac, femoral or other access arteries and there should be a low threshold to carry out an angiographic assessment to identify any bleeding point. The highest risk period for acute rupture of the aneurysm is during stent deployment.
Risk of conversion to an open procedure is 2% per annum, with female patients and patients with a small body habitus having a higher incidence of abandoned procedures.
EVAR vs Open surgery
Potential benefits of EVAR over the conventional open surgical procedure are: less physiological disturbance, reduced blood loss, less pain, reduced length of ICU and hospital stay. For infra-renal aneurysms additional benefits include reduced procedural time and avoidance of general anaesthesia. Disadvantages of EVAR include potential conversion to GA in a patient deemed unfit for GA, increased long-term complications such as endoleak and graft migration and the requirement of lifelong yearly surveillance by CT scan or duplex ultrasound.
Two UK multicentre studies looked at outcomes between open and endovascular AAA repair from 1999 to 2003.
The EVAR 1 study showed that EVAR reduces the operative mortality (30 day mortality) compared to open surgery by a third and medium term aneurysm-related mortality, but offered no decrease in long-term all-cause mortality at 4 years. EVAR is also associated with increased re-interventions and complications but has the same health related quality of life as open procedures.
The EVAR 2 study concluded there was no long-term survival benefit of EVAR versus surveillance in patients deemed unfit for open repair and showed that there was considerable 30 day operative mortality with EVAR in these patients (9% mortality). The study recommended focusing on increasing patients’ physiological fitness. A number of patients in the surveillance group however, crossed over into EVAR group so potentially skewing results. The most recent NICE guidelines state that there is uncertain benefit of EVAR over surveillance in patients who are unfit for open repair.
Since these studies, increasingly complex aneurysms are being treated with EVAR and in patients who would otherwise be deemed unfit for open surgery. Also, technological improvements in stent design and operator experience have increased so studies and longer term follow-up is required to determine the long-term risks and benefits of EVAR.
Suitability for EVAR
Location and Expertise
Evidence shows a reduced mortality from elective AAA repair in centres which carry out more than 32 cases per year. In recent years there has been a reconfiguration of vascular surgery services in the UK to create fewer specialist centres with a greater concentration of resources and experience in the field.
The Vascular Society of Great Britain and Ireland state that all patients undergoing elective AAA repair should be discussed by a multidisciplinary team consisting of vascular surgeons, interventional radiologists and anaesthetist with an interest in vascular surgery. A joint decision should be made, weighing up factors such as technical issues, physiological fitness and life expectancy of the patient, short and long-term benefits and risks of the procedure and the patient’s wishes.
Technical Suitability
Suitability of the aneurysm for endovascular repair depends on its morphological features and is formally assessed by 3D reconstruction CT. Features of the aneurysm assessed include: the proximal neck (length, diameter, presence of thrombus, angle), Distal landing zone (length, diameter), the calibre, tortuosity and presence of aneurysms or thrombus in the Iliac arteries and access vessels.
Preoperative assessment
Patients having EVAR should undergo the same thorough preoperative assessment and workup as for an open procedure. The incidence of major cardiorespiratory disease, hypertension, diabetes and stroke is increased in this group of patients. 70% of patients with AAA also have coronary artery disease. The American College of Cardiology and American Heart Association (ACC/AHA)) joint guidelines are a useful tool for the evaluation and care of cardiac patients undergoing non-cardiac surgery. According to the guidelines infra-renal EVAR is an intermediate risk procedure. Complex EVARs should be considered high risk procedures.
In addition to standard tests of cardio-respiratory fitness, the Vascular Anaesthesia Society of Great Britain and Ireland (VASGBI) recommend that an objective assessment of the patient’s functional status should be made – with cardiopulmonary exercise testing performed if it is available. Medication should be reviewed and optimised – all patients should be on low-dose aspirin and a statin unless there are specific contraindications. Consideration should be made to stopping non-steroidal anti-inflammatory drugs.
Particular attention should be paid to the preoperative renal function as these patients are at high risk for acute kidney injury (AKI) postoperatively for a number of reasons including the use of IV contrast and para-renal stents. Strategies for minimising renal impairment such as maintaining adequate hydration, limiting contrast load and omitting nephrotoxic drugs should be considered and planned for.
Current guidelines by the UK Renal Association on prevention of contrast induced nephropathy recommend that pre-procedural volume expansion with 0.9% sodium chloride or isotonic sodium bicarbonate should be used in patients at risk. There is conflicting evidence of benefit of one solution over the other. Meta-analyses have not shown compelling evidence for the use of N-Acetyl cysteine in the prevention of contrast induced AKI.
In general, younger, fitter patients undergo open AAA repair as they are more likely to cope with the physiological demands of open surgery and will avoid the longer-term complications and yearly surveillance associated with EVAR. EVAR is usually advocated for older patients with more significant co-morbidities who are deemed unfit for open repair as there is a reduced operative mortality compared with an open procedure.
- Objective functional assessment of cardiovascular fitness should be made (eg. CPEX)
- High risk of renal impairment:
- Stop nephrotoxic drugs
- Ensure adequate hydration
- Limit contrast dose
Intraoperative Management
Site and Setup
All EVARs should be carried out in a hybrid theatre with appropriate imaging facilities and the ability to convert to an open procedure if required.
Some hybrid theatres are not within the main theatre complex, so attention must be paid to the issues of remote-site anaesthesia. Protection from ionising radiation is important and additional mobile lead shielding should be placed between the patient and theatre staff, and an adequate distance from the x-ray tube should be maintained. This can make access to the patient more difficult so all IV lines and monitoring cables should be of sufficient length.
Monitoring
In addition to AAGBI minimum monitoring, an arterial line should be placed prior to induction of anaesthesia on the contralateral side to any surgical upper limb access site. Large bore IV access should be gained as there is potential for significant blood loss if converted to an open procedure. Central venous access should be inserted in complex or lengthy procedures and cardiac output should be monitored where appropriate. Temperature and hourly urine output should be monitored.
General vs Regional Anaesthesia
The use of general anaesthesia or neuraxial block is acceptable for EVAR. Short, infra-renal procedures have also been performed under local anaesthetic with sedation. Robust evidence of mortality or morbidity benefit of one technique over another is lacking. Factors to consider when making a decision include patient co-morbidities, use of antiplatelet or anticoagulant drugs, duration of surgery, the ability to lay flat and patient preference. The use of intraoperative heparin is not a contraindication to inserting an epidural however consideration should be given to the timing of removal post-operatively when the patient may still be significantly anti-coagulated. During prolonged procedures awake patients can become restless, requiring sedation or even conversion to GA. Prolonged femoral and internal iliac artery occlusion can cause ischaemic pain in the legs and buttocks respectively. Short periods of apnoea during the procedure are required to improve the image quality in digital subtraction angiography, so the ability of the patient to perform a breath-hold should also be considered. Other risks and benefits of regional versus general anaesthesia are documented elsewhere.
In open AAA repair, heparin is given to prevent thrombosis associated with stasis distal to the aortic cross-clamp. There is a lack of trial data regarding the use of heparin in EVAR, however experience with open vascular procedures means that most surgeons will apply the same practice and request systemic heparinisation routinely – given after the access vessels are cannulated. In centres where the activated clotting time (ACT) can be measured the aim is to maintain an ACT of 2-2.5 times the baseline (approximately 200-250 seconds). The surgeons may request reversal with protamine at the end of the procedure.
Complications of EVAR
Complications of EVAR can be divided into immediate and long term complications (see table 2).
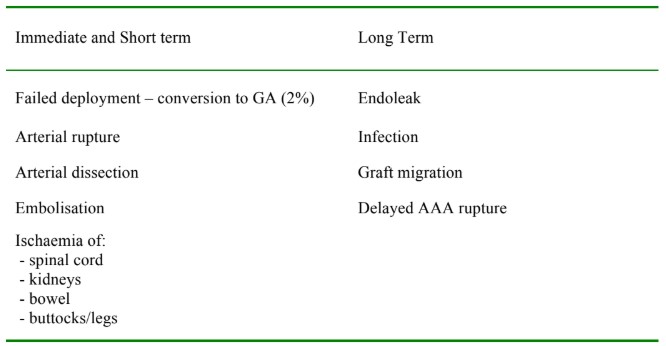
Table 2. Complications of EVAR
Endoleak
The reported incidence of secondary procedures in EVAR varies from 10-30% with the most common indication being endoleak.
The types of endoleak are classified as follows:
Type I – At sites of graft attachment
Type II – At sites of branches (i.e. back bleeding into the aneurysmal sac)
Type III – Graft defect eg. Hole in the graft
Type IV – Graft wall porosity
Type I and II leaks can cause on-going bleeding into the aneurysm leading to rupture. Endoleaks can be managed in a number of ways including conservative management with increased surveillance, coil embolisation, placement of graft cuffs, ligation of feeding collateral arteries, open repair and redo-EVAR.
Spinal Cord Ischaemia
Spinal cord ischaemia (SCI) following infra-renal EVAR is rare: 0.21%, however the risk of SCI in complex, supra-renal abdominal EVARs is increased due to a number of factors. Long stent grafts will occlude more of the thoracic and lumbar collateral arteries to the spinal cord. The largest collateral artery, the artery of Adamkeiwicz arises directly from the aorta anywhere from T5 – L5 (although most commonly from T9-T12) so it is more at risk from occlusion with stent grafts which extend supra-renally. Collaterals to the spinal cord also arise from the internal iliac, inferior mesenteric (IMA) and middle sacral arteries caudally. The IMA is invariably occluded during infra-renal EVAR. Hypoperfusion, thrombosis and microembolism of atheromatous material into the collateral arteries are also thought to be contributory factors to SCI in abdominal EVARs. Factors increasing the risk of microembolism include a prolonged procedural time, extensive manipulation of the intravascular catheters and perioperative embolisation the lumbar arteries. Patients who have had previous AAA repair are also at greater risk of SCI.
Spinal drainage of CSF can be used to prevent or treat SCI, by increasing the perfusion pressure to the spinal cord (perfusion pressure = MAP – CSF pressure). Pre-operative spinal drains are occasionally inserted for complex abdominal EVARs if the patient is thought to be at particularly high risk of SCI. A high index of suspicion should be maintained in all patients post-operatively and a spinal drain inserted if neurology develops.
Emergency EVAR
An increasing number of ruptured AAAs are being treated with EVAR. Several case series have suggested a lower mortality with EVAR compared with open repair in ruptured AAA however this may be due to selection bias with more stable patients undergoing EVAR. (A multi-centre randomised controlled trial is currently underway looking at this (IMPROVE trial).
There is a risk of conversion to an open procedure, so preparation should be as for an emergency open repair. The anaesthetic perioperative management for open ruptured AAA repair is well covered elsewhere in the literature.
In brief, the preoperative anaesthetic management should include a rapid, targeted assessment, large bore IV access, cross-match and hypotensive resuscitation. Equipment for large volume, rapid blood transfusion and cell salvage should be prepared, as well as infusions of inotropes and vasodilators. Arterial access should be gained if possible before the start of the procedure.
Smooth co-ordination and communication between the anaesthetic, surgical, radiology and theatre teams is vital to ensure minimal delay and optimal outcome in this group of patients. In stable patients a pre-operative CT scan is acquired and a joint decision is made between the surgeons and radiologists as to the suitability of the aneurysm for endovascular repair.
The first surgical step is often to cannulate the access vessels and insert an expandable aortic balloon into the supracoeliac aorta under local anaesthesia. The balloon acts as a cross-clamp which can be deployed to gain haemorrhage control if required. After this the procedure can continue under local anaesthesia with judicious use of sedation, or general anaesthesia. Neuraxial anaesthesia is not appropriate for ruptured AAA repair.
Emergency EVARs can be carried out solely under local anaesthesia and some studies have suggested improved outcomes with local anaesthesia alone, however a number of factors may prevent the patient tolerating the procedure, including:
- Abdominal pain from expanding haematoma
- Respiratory insufficiency from increased intra-abdominal pressure
- Ischaemic pain in legs and buttocks
- Agitation from hypotension and metabolic disturbance.
Induction of anaesthesia causes loss of abdominal tone and can lead to loss of tamponade of the aneurysm with catastrophic bleeding ensuing. If general anaesthesia is required, it must not be induced until the surgical team is prepared to deploy the aortic balloon or open the abdomen rapidly and apply an aortic cross-clamp.
Post operatively the patient should be nursed in an intensive care or high dependency care environment.
- Excellent teamwork and communication is vital in EVAR for ruptured AAA
- Prepare for an open procedure
- Consider local anaesthesia alone in appropriate patients until haemorrhage control is gained
Summary
Patients undergoing endovascular AAA repair have a greater incidence of major comorbidities and should undergo thorough preoperative assessment and optimisation within a multidisciplinary setting. A distinction should be made between simple infra-renal EVAR and complex supra-renal EVARs which can carry a greater perioperative risk.
Anaesthetic preparation should be carried out with the awareness that there is a risk of major haemorrhage and conversion to an open procedure can occur.
Good team-work and communication is essential when carrying out EVARs and this is even more vital during emergency EVARs.
ANSWERS TO QUESTIONS
- FTTF
- TTTF
- TFFF
- FTTT
WEBLINKS AND FURTHER READING
- NICE: Abdominal Aortic Aneurysm – endovascular stent grafts: guidance. http://guidance.nice.org.uk/TA167/Guidance/pdf/English
- ACC/AHA 2007 Guidelines on perioperative cardiovascular evaluation and care for non-cardiac surgery. Anesth Analg. 2008 Mar;106(3):685-712
- The Vascular Anaesthesia Society of Great Britain and Ireland
- http://www.vasgbi.com/
- Endovascular Aneurysm repair trials. http://www.evartrials.org/
- Abdominal Aortic Aneurysm Quality Improvement Project
- http://www.aaaqip.com/aaaqip/index.html
- Clinical Practice Guidelines for endovascular Abdominal Aortic Aneurysm repair: Written by the Standards of Practice committee for the Society of Interventional Radiology, and Endorsed by the Cardiovascular and interventional radiological society of Europe and the Canadian Interventional Radiology Association. http://www.sirweb.org/clinical/quality.shtml
- Endovascular aneurysm repair versus open repair in patients with abdominal aortic aneurysm (EVAR trial 1): randomised controlled trial. Lancet. 2005 Jun 25-Jul 1;365(9478):2179-86
- Endovascular aneurysm repair and outcome in patients unfit for open repair of abdominal aortic aneurysm (EVAR trial 2): randomised controlled trial. Lancet. 2005 Jun 25-Jul 1;365(9478):2187-92.



