General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Below what glomerular filtration rate is renal replacement therapy usually commenced?
- 100ml/min/1.73m3
- 30ml/min/1.73m3
- 15ml/min/1.73m3
- True or false, a positive crossmatch between the donor and recipient is an absolute contraindication to transplantation?
- When is immunosuppression is started?
- Pre-operatively
- Intra-operatively
- Post-operatively
INTRODUCTION
Since the first successful human kidney transplant in 1954 renal transplantation has become the treatment of choice for most patients with end-stage renal disease. Renal disease and its associated comorbidities can provide the anaesthetist with a myriad of problems and these patients require thorough preparation for surgery and careful peri-operative management. This tutorial briefly looks at the identification of both recipients and donors for transplant and their peri-operative anaesthetic management.
PATIENT POPULATION
Patients presenting for renal transplantation present a challenge to the anaesthetist relating to both the chronic kidney disease (CKD) and its underlying cause. A variety of conditions result in CKD including most commonly, diabetes, hypertension, glomerulonephritis and polycystic kidney disease. The decline in glomerular filtration rate to less than 15ml/min/1.73m3, also known as end stage renal disease (ESRD)1, typically results in the initiation of renal replacement therapy, either by haemodialysis or peritoneal dialysis.
All patients with end stage renal disease should be considered for renal transplantation. For suitable patients, renal transplantation results in better long-term survival compared to renal replacement therapy across most age groups and aetiologies of ESRD2. Absolute contraindications to consideration for transplant are listed in box 13:

Box 1: Absolute contraindications to renal transplantation
Over recent years the demand for renal transplants has continued to rise, the limited availability of cadaveric kidneys means some patients on the waiting list have a significant wait for a transplant. This has resulted in an increased number of living donors. Living donation usually occurs between persons who know each other, either live related transplants, where the individuals are related by blood, or live unrelated transplants, where the individuals are not related by blood but often have close ties.
The organs donated between April 2008 and March 2009 are shown below in table 23:
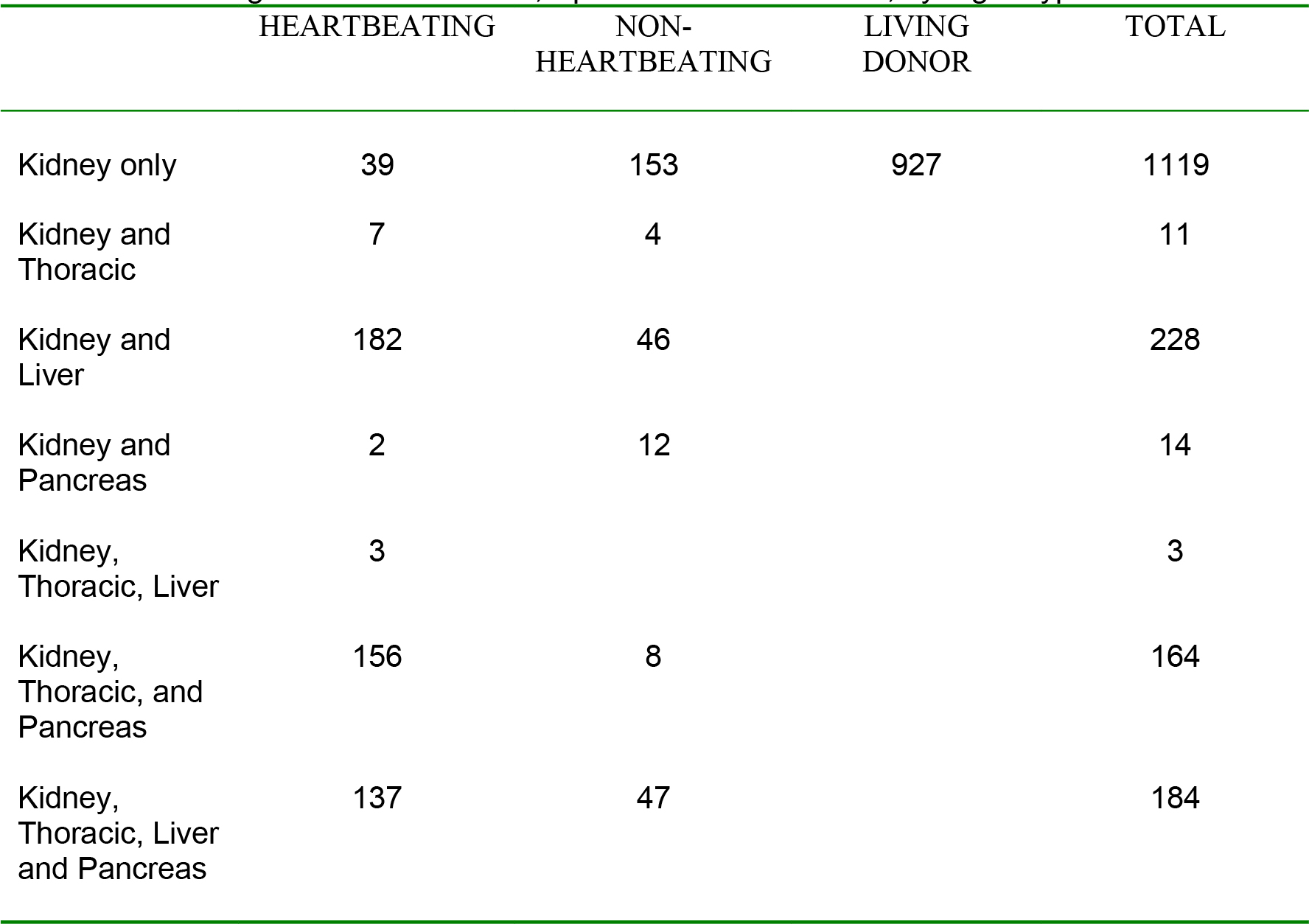
Table 1: Solid organ donors in the UK, April 2008 – March 2009, by organ types donated
Once entered into the transplant programme the patients undergo extensive evaluation. Since these patients can wait up to two years for their transplant the patients usually have undergone all appropriate investigations and should be adequately worked up when they present for their transplant.
MATCHING
Matching of the organ to recipient can be divided into three phases4:
Blood type matching
Using the four major blood types A, B, AB and O, as with blood transfusion, type O is the universal donor and Type AB is the universal recipient.
Tissue matching
This relates to genetic matching between donors and recipients. Currently six specific antigens, called major histo-compatibility complex, are defined in each donor and recipient. The best compatibility is a six-antigen match between donor and recipient, this match occurs 25 percent of the time between siblings and occasionally at random in the general population. Long term outcomes relate to matching, with six antigen match having best outcome, then 5 antigen match and so on. However, immunosuppressant medications have improved to the extent that even poor tissue matches now appear to respond and survive similarly
Crossmatching
Very sensitive and final test performed on a kidney donor and recipient. The technique allows definition of how a transplant recipient may respond to particular cells or proteins of the donor kidney. This has allowed for possible detection of a recipient who would reject an organ. A negative crossmatch means the recipient has not responded to the donor and the transplant can go ahead. A positive cross match means the recipient has responded to the donor and the transplanted kidney is likely to be rejected. Recent advances have seen progress in successful transplantation across a positive crossmatch1.
ALLOCATION AND TIMING
Allocation of grafts is based on both matching of the grafts to recipients and a waiting list system, with special priority given to children to minimize the impact of CKD on growth1.
Timing of the transplant is important as future graft function is directly related to warm ischemic time (harvest and transplant time), cold ischemic time (storage time, especially if greater than 24 hours), transplanted graft perfusion and pre-donation graft state5. Living donation is a scheduled event and allows optimal preparation of the donor and recipient alongside minimizing organ preservation time.
PRE-OPERATIVE ASSESSMENT
As discussed previously both the cause and effects of CKD on the patient are of interest to the anaesthetist. Cardiovascular disease and peripheral vascular disease have a higher incidence in these patients compared with the general population. There is also a higher prevalence of hypertension, whether this is a cause or result of the CKD remains debatable2. When creatinine clearance falls to less than 10 mL/min uraemia can develop, although some patients may be symptomatic at higher clearance levels. In combination with uraemia the hypertension causes left ventricular hypertrophy and dilatation and evidence of cardiac failure may well be present. Uraemia itself can have multi-system effects (Table 3)5:

Box 2: System effects of uraemia
When an organ becomes available and a recipient has been identified they need to be fasted and prepared for theatre. This often involves dialysis. The timing of the dialysis has implications. Recent dialysis can lead to electrolyte disturbance, in particular hypokalaemia, fluid depletion and residual anticoagulation. These factors should all be considered in the pre-operative assessment.
PERI-OPERATIVE MANAGEMENT
Whilst transplant surgery has been carried out under regional anaesthesia, general anaesthesia with paralysis and controlled ventilation is the current method of choice. Premedication addressing both anxiety and the increased incidence of gastric paresis and reflux in this patient population should be considered.
Alongside instituting standard monitoring, central venous access should be gained to allow monitoring of the central venous pressure (CVP), core temperature should also be monitored throughout. Invasive arterial pressure monitoring is not required as standard in these patients and should be avoided unless clinically indicated as it poses a risk to future fistula sites.
A state of immunosuppression is induced immediately after anaesthesia, just prior to the operation. Different units have different regimens. Immunosuppression is then maintained postoperatively. Antibiotics are also given before incision.
OPERATIVE MANAGEMENT
Future graft function is directly influenced by transplanted graft perfusion. The general aims are to keep the patients warm and well perfused, maintain an adequate mean arterial pressure and optimising fluid status. The optimal CVP at the point of reperfusion has been debated, previously aggressive fluid loading and a CVP >15mmHg had been recommended. Subsequent evidence suggest a CVP of 7- 9mmHg may offer equally good graft function but have a lower risk of volume overload6.
Other drugs are often used intra-operatively to try to improve graft function. Different units have different protocols and the evidence to support their use is not strong. Most commonly used are dopamine, furosemide and mannitol:
Dopamine; given as an infusion from induction, in a low dose infusion (typically 0.5 – 3 mcg/kg/min).
Furosemide; given as a bolus prior to reperfusion, in a varying dose depending on local protocol (40 – 250mg) is aimed at inducing diuresis, promoting urine flow in the graft and so avoiding oliguria. This can occasionally promote massive diuresis resulting in difficult fluid management post operatively.
Mannitol; as an intravascular volume expander and osmotic diuretic it is given with the aim of promoting renal blood flow. Again given as a bolus pre reperfusion at a dose of 0.25 – 0.5 g/kg depending on local protocol.
POST-OPERATIVE MANAGEMENT
The vast majority of patients will be woken up and extubated post operatively. After leaving recovery they are nursed in high dependency areas. The aim of a warm, well-perfused patient, that was initiated pre-operatively, continues in the post-operative period. Fluid regimes are often variable, taking into account the previous hour’s urine volume and changes in CVP. In an otherwise stable patient, oliguria should trigger surgical review and consideration of urgent Doppler imaging of the grafts blood supply.
Analgesia is often provided by an intra-operative bolus of morphine, followed postoperatively by regular paracetamol and morphine. Patient Controlled Analgesia protocols, if available, are well tolerated in this group. In the event of a failing graft this regime should be reviewed due to the potential accumulation of morphine metabolites. Due to their potential renal toxicity, non-steroidal anti inflammatories should be avoided.
SUMMARY
The number of patients undergoing renal transplantation continues to increase. This patient population presents various challenges to the anaesthetist, and awareness of the pre-operative and peri-operative management of these patients is important for all trainees to appreciate.
REFERENCES
- Collins B, Johnston T. Renal Transplanation: Urology; Oct 2009
- Salifu M, Otah K; Perioperative Management of the patient with chronic renal failure: Jan 2008.
- UK transplant statistics: www.uktransplant.org.uk
- Peters T; RENALIFE 2001 Special edition; Vol 17.
- Rang, S; West, N; Howard, J; Cousins, J: Anaesthesia for Chronic Renal Disease and Renal Transplantation: European Association of Urology and European Board of Urology Update series 4 (2006): 246-256
- De Gasperi A, Narcisi S, et al. Periopertive fluid management in kidney transplantation: is volume overload still mandatory for graft function? Transplant Proc 2006;38:807-9
ANSWERS TO QUESTIONS
- C
- FALSE
- A



