Paediatric Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the tutorial.
- How is an apnoea in a pre-term infant defined?
- Which of the following will increase pulmonary vascular resistance in a neonate?
a. Hypoxia
b. Hypercarbia
c. Isoflurane
d. Nitrous oxide - Foetal haemoglobin will shift the oxyhaemoglobin dissociation curve in which direction?
a. Left
b. Right
What clinical effect will this have? - Which of the following statements are true?
a. Babies born prior to surfactant development are prone to developing respiratory distress0 syndrome
b. Pain pathways do not develop until 36 weeks gestational age
c. Theatres should be pre-warmed to 24°C for a pre-term
d. A neonate of <1200g will require a size 2.5mm endotracheal tube
e. Pressure controlled ventilation is preferred to volume controlled ventilation
f. Pre-term babies can be have day case operations at 45 weeks post gestational age
g. Intravenous caffeine can be given at 100mcg/kg to try and reduce the incidence of post operative apnoeas
INTRODUCTION
Premature babies are defined as those born before 37 weeks gestation and account for about 13% of UK births. Those born prior to 28 weeks gestational age are classified as extreme premature neonates. Many of these babies weigh less than 2500g (low birth weight); those weighing less than 1000g are defined as extremely low birth weight.
The clinical course and long term prognosis of these infants depends on the gestational age (GA) at birth, with increasing morbidity and mortality associated with lower GA. Advances in modern neonatal intensive care mean that premature babies commonly survive from as early as 24 weeks GA. Premature infants may be hospitalised for many weeks post delivery and may present for surgery linked to their prematurity, but also for incidental operations at a young age.
Common procedures requiring anaesthesia include:
- PDA ligation
- Vitrectomy or laser for retinopathy of prematurity
- Inguinal hernia repair
- Laparotomy for NEC or bowel perforation
- CT/MRI scanning
Anaesthesia for premature babies can present many challenges, particularly related to immature physiology and incomplete organogenesis. Some maturation continues after birth, and surgery should be delayed if at all possible, particularly until respiratory stability can be achieved.
In order to be able to provide safe anaesthesia for this age group, it is important to understand the special problems facing the preterm baby.
PHYSIOLOGY
Respiratory system
Full maturation of the respiratory system occurs around 36 weeks gestational age. Pulmonary surfactant is produced from 32-34 weeks and lack of surfactant is one of the key problems seen in premature infants. Surfactant is necessary to lower surface tension in alveoli, to facilitate alveolar opening and prevent alveolar collapse at end expiration. Babies born prior to 28 weeks develop respiratory distress syndrome (RDS) (previously known as hyaline membrane disease), the severity of which may be ameliorated by giving antenatal steroids to mothers in preterm labour to induce surfactant production. Artificial surfactant is given immediately after birth to preterm infants to reduce the risk of RDS.
Modern practice is to use nasal continuous positive airway pressure (ncpap) for respiratory support in premature neonates, but some of these babies, particularly VLBW infants, will require ventilation and oxygen supplementation for prolonged periods. Those exposed to artificial ventilation or greater than 28 days of oxygen therapy may develop bronchopulmonary dysplasia (BPD) (also known as chronic lung disease of prematurity, CLD). This is thought to be due to a combination of volutrauma, barotrauma, oxygen toxicity and accumulation of secretions. BPD is characterised by increased oxygen requirements, reduced lung compliance, reversible airway obstruction and increased susceptibility to infections such as bronchiolitis.
Premature infants are prone to apnoeas. These are defined as pauses in breathing of greater than 20 seconds or loss of effective breathing associated with a bradycardia. These can occur as a result of airway obstruction, loss of central respiratory drive, or more commonly a combination of both. Apnoea is more common in the presence of hypoxia, sepsis, intracranial haemorrhage, heart failure, and drugs such as prostaglandins, opioids and anaesthetic agents. Babies with anaemia, a history of apnoea, neurological disease and lung pathology are more susceptible to developing apnoeas after anaesthesia, and this increased susceptibility may occur up to 60 weeks post conception. Apnoeas usually occur within the first 12 hours after surgery but may persist for up to 72 hours.
Babies have a high oxygen consumption in order to meet their high metabolic rate. They are unable to increase their tidal volume, instead compensating for increased respiratory demands through a raised respiratory rate, which can lead to early fatigue. They have a reduced functional residual capacity which is smaller than their closing capacity rendering them dependant on PEEP and prone to rapid desaturations.
Cardiovascular System
Delayed closure of the ductus arterious is common in premature infants and leads to left to right shunt from the aorta to the pulmonary artery, increased pulmonary blood flow and cardiac failure with low diastolic blood pressure. PDA is a risk factor for necrotizing enterocolitis, intraventricular haemorrhage and CLD. Oxygen reduces pulmonary vascular resistance and increases pulmonary blood flow further – high-inspired oxygen concentration should be avoided. Conventional treatment for PDA is fluid restriction and diuretics. Medical closure may be attempted with non-steroidal anti-inflammatory agents (NSAIDs), or the duct may be closed by surgical ligation via thoracotomy.
Renal system
Preterm infants have an impaired ability to concentrate urine, so cannot tolerate under and over hydration. They are unable to retain sodium, have a poor response to aldosterone and have high levels of antidiuretic hormone. For this reason they are prone to hyponatraemia. Water loss is common in preterm infants due to the large body surface area and thin skin, particularly in the first few days of life.
Drug excretion is also delayed due to an immature renal system.
Gastrointestinal system
Premature babies have limited glycogen stores and immature hepatic function. They are particularly prone to developing hypoglycaemia during starvation and blood sugars should be regularly checked. Hyperglycaemia should also be avoided as this will predispose to dehydration. Drug metabolism is immature and drug doses and dose intervals should be modified.
Premature babies are prone to develop necrotising enterocolitis (NEC). This is an inflammatory condition that may affect any part of the gut, usually the terminal ileum, caecum and ascending colon. The features of NEC include abdominal distension, blood stained stool, and sepsis with possible gut perforation. Risk factors include prematurity, hypoxia, early feeding with formula milk and colonization of the gut with pathogenic bacteria.
Central nervous system
Intraventricular haemorrhage occurs in 25% of very low birth weight infants, mostly within the first 72 hours of life. Intraventricular haemorrhage may be complicated by ventricular dilation, which can progress to hydrocephalus, parenchymal infarction, or cystic periventricular matter injury. The risk factors include RDS, hypotension, aggressive fluid resuscitation, fluctuating blood pressure and hypertonic infusions. Periventricular leucomalacia describes changes in white matter associated with hypoxia, hypotension, severe hypocarbia, large PDA, toxic injury or infection. It is a strong predictor for later cerebral palsy.
Research has shown pain receptors develop by 20 weeks GA and pain pathways develop by 26 weeks. A foetus of 26 weeks may demonstrate a stress response in response to stimulation. There has been recent concern about that anaesthetic agents may have an effect on the developing brain leading to later memory and learning impairment. Only essential surgery should be performed in early life, and ketamine should probably be avoided in premature babies.
Retinopathy of prematurity
Retinopathy of prematurity (ROP) is due to retinal ischaemia and proliferation of retinal vessels at the junction of the vascular and non-vascularised retina. It is seen in LBW infants and is caused by vasoconstriction of retinal vessels due to exposure to high concentrations of supplemental oxygen. It can lead to retinal detachment, fibrosis and is a common cause of blindness in children, but can be prevented by good neonatal care and avoiding exposure to high concentrations of oxygen.
Haematological system
A premature baby has a lower level of haemoglobin than a term baby, with a greater proportion of this in the form of foetal haemoglobin. Foetal haemoglobin has a reduced ability to release oxygen to the tissues therefore blood transfusions may be required to meet a recommended target haematocrit of 40-45% in order to aid oxygen delivery.
Temperature
Control of the thermal environment is essential in the premature baby. They are particularly vulnerable to heat loss due to their large surface area to body weight ratio, increased evaporative losses through the thin, poorly keratinised skin, inability to shiver and limited heat production from brown fat metabolism. . Over or under heating these babies will lead to hypoglycaemia and increased oxygen requirements, increasing the likelihood of apnoeas.
PRE-OP ASSESSMENT
A full preoperative assessment with a detailed history from both the parents and the notes is essential.
Particular points to ascertain are:
- Gestational age at birth and the current gestational age
- Weight
- Periods of mechanical ventilation, CPAP and oxygen therapy and the duration
- Apnoeas – frequency, duration, possible triggers
- Co-morbidities, particularly cardiac
- General health, growth and development
- Previous operations
- Medications including oxygen
Examination must include airway assessment – this can warn of potential difficult intubation. If already intubated the size and length of the endotracheal tube should be noted with chest auscultation carried out for confirmation of correct tube placement.
Recent investigations including haemoglobin, haematocrit, platelets, electrolytes and a coagulation profile should be checked and should be within the normal limits for a premature neonate. A crossmatch should be taken where blood loss is anticipated to be greater than 10% of blood volume. All premature babies should have an echocardiogram performed before surgery, which should be noted as well as current medication including inotropic support
Starvation times should be minimised to prevent hypoglycaemia and dehydration. Starvation times should be logged as deficits will need to be corrected intraoperatively
At the preoperative assessment, parents should be informed about the possibility for post operative ventilatory support particularly if the child is extubated and there is a history of recent apnoeas.
INTRA-OPERATIVE MANAGEMENT
Anaesthesia
The infant may already be intubated and ventilated prior to arrival in the operating theatre. If not, inhalational induction is often preferred as intravenous access may be difficult, particularly if the infant has undergone multiple procedures since birth. A range of uncuffed tubes should be available – a neonate of <1200g may need a 2.5mm tube, with increasing diameters of tubes available as the weight increases. The tube position after intubation should be confirmed by auscultation and, as a guide a 2kg baby has the tracheal tube positioned at the gum margin at the 8cm mark. There is an increased incidence of difficult intubation in premature infants, and if the infant has undergone prolonged ventilation, there is a possibility of subglottic stenosis. Modified rapid sequence induction may be considered, but may be associated with rapid desaturation made worse by distortion from cricoid pressure. An orogastric tube is useful after intubation to decompress the stomach and to minimise splinting of the diaphragm and facilitate ventilation. The position of the endotracheal tube should be verified whenever the baby has been moved.
Ventilation may be complicated by poor compliance due to previous mechanical ventilation and subsequent chronic lung disease. Barotrauma should be avoided as well as excessive oxygen concentrations as the latter will predispose to retinopathy of prematurity. A balanced anaesthetic technique should be used, and deep volatile anaesthesia avoided (myocardial depression).
Research has shown that neonates have intact pain pathways from early gestation and therefore need analgesia titrated to the level of pain. A multimodal approach should be employed, however this is not always straightforward in the premature infant. Paracetamol is commonly used although, due to immature hepatic pathways, the intravenous dose should be adjusted:
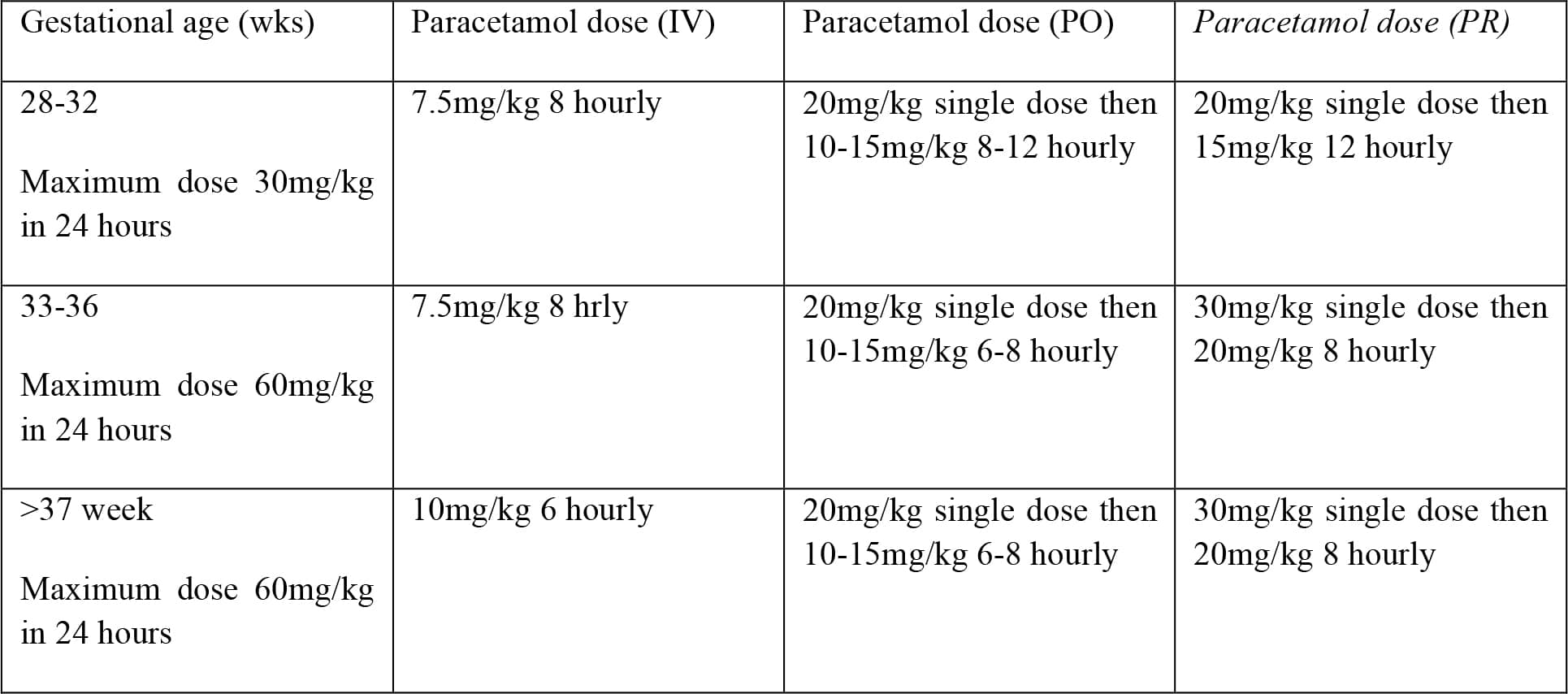
NSAIDs for analgesia are contra-indicated due to renal immaturity. In the case of duct dependent systemic or pulmonary circulation NSAIDs may cause premature closure of a PDA. The use of opiates should be minimised if planning to extubate the infant at the end of surgery as these will lead to an increased incidence of postoperative apnoeas. Where opiates are necessary, they should be short acting such as fentanyl. The use of local anaesthetic techniques is encouraged, for instance local infiltration by the surgeon, or where possible a caudal epidural or spinal.
Theatre/equipment
The environment used should be appropriate for anaesthetising babies and have all the necessary equipment and staff for this role. For infants <1 kg, interventions such as duct ligation may be performed in the NICU. The anaesthetist requires a trained assistant who is experienced and competent in dealing with small preterm babies.
Full monitoring must be available as per AAGBI guidelines. Ideally there should be 2 oxygen saturation probes – in patients with a patent PDA one should be placed on the right hand and one on the lower limb to compare pre ductal and post ductal levels. ECG via neonatal electrodes, non-invasive blood pressure, capnography (either by end tidal or transcutaneous) and temperature monitoring are mandatory. Invasive arterial blood pressure monitoring should be considered in babies receiving inotropic support or where major haemorrhage is possible.
A paediatric breathing circuit, such as a Mapleson F, should be available with suitably sized airway devices and adjuncts, which should all be checked prior to starting the case, as per the WHO surgical safety checklist. Dead space must also be minimised. Babies should all be supported with positive pressure ventilation to prevent exhaustion. Hyperventilation, hyperoxia, high peak inspired pressures and barotrauma should be avoided. If using a ventilator, it should ideally be able to deliver pressure-controlled ventilation with PEEP.
The theatre should be pre-warmed to achieve a temperature of at least 25°C. In addition to this, active warming devices, such as overhead heaters should be used as well as a paediatric heat moisture exchange. All fluid and blood products should be warmed prior to administration. The surgical team need to ensure that any irrigation fluid is warmed prior to use. Heat loss should be minimised by preventing unnecessary exposure. Surgical drapes should be lightweight, preferably plastic allow the baby to be clearly seen.
Fluid, Blood and Glucose
Fluid balance, electrolyte and acid base deficits should all be corrected prior to surgery although in some cases this is not possible. Fluid administration should be divided into replacing deficits and covering on-going losses.
To replace a fluid deficit, an isotonic (0.9% sodium chloride or Hartmann’s solution) fluid bolus of 10-20ml/kg should be given, with reassessment following the bolus. If 40ml/kg replacement has been given or loss of 10% of the circulating blood volume has occurred, the next bolus should be blood at 10ml/kg.
Blood glucose should be closely monitored. Hypoglycaemia is common in this patient group, particularly where parenteral nutrition has been discontinued pre-operatively. There is some debate over absolute values which are ‘too low’ but a widely accepted cut off is a blood sugar of <2.6mmol/L. Glucose can be given as a bolus of 1- 2ml/kg of 10% glucose in addition to maintenance fluid or with the maintenance fluid using a solution containing 10% glucose at 4ml/kg/hr . An alternative method would be to add 25ml of 50% glucose to 500ml Hartmann’s solution to create a 2.5% solution. Regular blood sugars should be checked in order to confirm normoglycaemia.
On-going fluid losses should be calculated by assessing suction pumps and swabs in addition to clinical signs such as heart rate, blood pressure, capillary refill time and urine output. These should be replaced by isotonic fluid or blood if the loss is greater than 10%. In extremely premature infants and those with cyanotic heart disease the haematocrit should be maintained at 35-40%/
Special Considerations
Depending on what type of surgery is being performed, certain aspects of anaesthesia should be taken into consideration.
- Inguinal hernia repair:
- Normally carried out once weaned from mechanical ventilation
- Consider caudal or ilio-inguinal block
- PDA ligation:
- Positioning for surgery as for left thoracotomy
- Sudden ligation of the duct may cause a large surge in systemic BP and increase the risk of intraventricular haemorrhage. The duct should be clamped gently or the concentration of the volatile could be temporarily increased.
- Laparotomy:
- Often for NEC, therefore very sick baby.
- Correct any acid base abnormality and coagulopathy pre op (be prepared to give FFP and platelets)
- Aggressive fluid therapy is usually required due to 3rd space losses. Admission to critical care post op.
- CT/MRI:
- Non stimulating procedure for investigating potential cerebral damage
- Can often be achieved without pharmacological intervention by feeding the infant and then securely wrapping them. However, if this fails the infant should be anaesthetised and the airway secured with an endotracheal tube
and ventilated to control the CO2 and minimise changes in ICP.
POST-OPERATIVE CARE
A decision needs to be made over whether the baby should remain intubated or whether extubation is possible with the neonatal unit kept informed of this. This decision should be made taking into consideration the preoperative state of the baby as well as the type of surgery performed. If plans are made to extubate, it should only occur once the baby is fully awake and managing adequate tidal volumes without support.
The main postoperative risk to these infants is that of apnoea. Premature babies under 60 weeks gestational age need to be kept in a high dependency unit for at least 12 hours post operatively and for a further 12 hours following any apnoeic period. Continuous apnoea alarm monitoring must be available and IV caffeine at 10mg/kg has been given to try and reduce the incidence of apnoeas. CPAP may well be useful at this stage as it can distend the chest wall and therefore trigger stretch receptors.
ANSWERS
- An apnoea is a pause in breathing of greater than 20 seconds or loss of effective breathing associated with bradycardia.
- a) and b)
- a) left – oxygen is bound more avidly but has a reduced ability to release it to the tissues
- a), d) and e)
REFERENCES AND FURTHER READING
- Bayley G, Walker I. Special considerations in the premature and ex-premature infant. Anaesthesia and Intensive Care Medicine 12(3): 91-94
- Santin RL, Porat R. Apnea of prematurity. Available at www.emedicine.com/ped/topic1157.htm
- Walther-Larsen S, Rasmussen LS. The former preterm infant and risk of post-operative apnoea: recommendations for management. Acta Anaesthesiol Scand 2006; 50: 888-93
- APA Consensus Guideline on Perioperative Fluid Management in Children v 1.1 September 2007



