General Topics
KEY POINTS
- Free-flap surgery optimises functional recovery for patients suffering potentially disabling injuries.
- Complications from free-flap surgery may follow prolonged ischemia during flap elevation, slow establishment of surgical anastomoses, external compression of a completed flap, reperfusion injury, or infection.
- Key goals of anaesthesia management are to keep the patient safe while supporting perfusion and optimising oxygen delivery and favourable coagulation conditions to aid flap survival.
INTRODUCTION
Plastic and reconstructive surgeries aim to repair tissue defects and restore functional anatomy following disabling injuries. The goals are to cover a tissue defect, replace tissue loss from trauma, improve blood supply to an otherwise poorly vascularised bed, or improve sensation, as well as to provide padding over bony prominences.
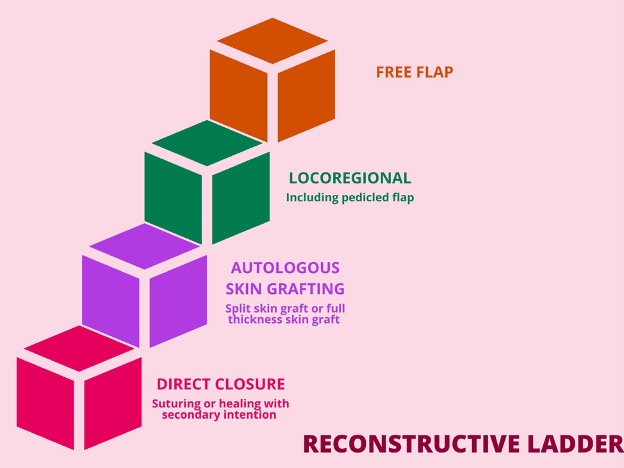
Figure 1. The reconstructive ladder.
A reconstructive ladder (Figure 1) is used to guide surgical wound closure. The ladder outlines increasing complexity of management for plasticsurgery. A flap is a vascularised block of tissue that is mobilised from its donor site and transferred to another location, adjacent or remote, for reconstructive purposes. In contrast to skin grafts, flaps retain their own vascular supply pedicle. In a pedicled flap, the donor tissue remains attached to the original donor site via an intact vascular pedicle and is transferred to cover a defect site. The final step of the ladder involves a freeflap. In contrast to pedicled flaps, free flaps are completely detached with division of the vascular pedicle and then blood supply is reestablished using microsurgical anastomoses of flap and recipient vessels. Given the complexity of this procedure, it carries the highest risk of flap failure.
Microvascular reconstruction involves 3 main stages:
- raising the flap with meticulous dissection,
- establishing microvascular reanastomoses of the flap to the recipient site, and
- insetting the flap and shaping the transferred tissue to close the defect.

Flaps can fail in 1% to 5% of cases, with the highest risk occurring in the first 2 days following operation.1 Local complications can occur because of ischemia, reperfusion injury, or infection (Table 1). The severity of microvascular damage and the risk of reperfusion injury are proportional to duration of primary ischemia time. Delayed establishment of microanastomoses once the flap is elevated increases complications owing to granulation and contracture formation.
OVERVIEW OF ANAESTHESIA FOR FLAP SURGERY
Anaesthetic management of microvascular reconstructive surgery can impact flap survival. Optimal flap perfusion and oxygen delivery are the key considerations.
Microcirculatory flow is essential to flap survival. Dissected vascular tissue is denervated and hence lacks intrinsic sympathetic tone. However, arteries and veins at both the donor and recipient sites still respond to physical, chemical, and humoral stimuli such as hypothermia, catecholamines, and drugs.3 Lack of intact lymphatic drainage increases interstitial oedema, predisposing to venous thrombosis-mediated flap failure.
![]()
Figure 2. Hagen-Poiseuille equation.
The Hagen-Poiseuille equation (Figure 2) can guide anaesthetic management. Anaesthetists can influence vessel radius (r) by altering smooth muscle tone as this significantly influences flow. Maintenance of blood pressure (ΔP ¼ difference between systemic arterial pressure and venous pressure) carries importance in the sense that it improves flow, but not at expense of vasoconstriction, which reduces vessel radius. Bloodviscosity (μ) should ideally be at low normal levels to facilitate higher flows.
![]()
Figure 3. Oxygen flux equation.
Oxygen delivery is critical for flap integrity (refer to oxygen flux equation, Figure 3). Decreased cardiac output, anaemia, and hypoxemia cancompromise viability of the flap. Postanastomotic distance from blood vessels to donor tissue is vital for oxygen transfer. Oedema from inflammation or excessive fluid therapy can increase diffusion distance, thereby compromising oxygen delivery and flap survival.
PREOPERATIVE ANAESTHETIC CONSIDERATIONS
Comprehensive multidisciplinary preanaesthetic assessment should aim to optimise patient fitness and identify factors that might compromise flap survival.
In poorly controlled diabetic patients, suboptimal donor blood vessel integrity and immunosuppression increase risk of surgical site infection. Haemoglobin A1C levels below 8% and blood glucose levels within 4 to 12 mmol/L are important.2 In renally impaired patients, dialysis timing to minimise fluid overload plus avoidance of high uric acid levels can aid wound healing. Patients with arterial insufficiency may warrant further investigation and consideration for revascularisation surgery prior to microvascular reconstruction.
Smoking cessation 4 to 6 weeks prior to surgery minimises risk of vasoconstriction and flap compromise as well as pulmonary complications.2 Nicotine-replacement therapy is contraindicated as it may reduce donor blood flow. Obese patients should consider weight-reduction strategies prior to flap surgery to improve surgical outcomes.
Cancer patients presenting for microvascular reconstructive surgery have multiple comorbidities, immunosuppression, malnutrition, and elevated thrombo-embolic risk. Radiotherapy can cause microvascular damage and make surgery more technically challenging. Hypoalbuminemia can contribute to wound dehiscence and interstitial oedema.
Preoperative haemoglobin levels of <10 g/dL have been associated with increased flap failure and thrombosis.4 Anaemic patients with haemoglobin >8 g/dL or haematocrit >30% should be considered for preoperative blood transfusion.
Contraindications for surgery are hypercoagulable conditions, such as sickle cell anaemia and polycythaemia, as they significantly increase risk of anastomotic thrombosis.5
INTRAOPERATIVE ANAESTHETIC CONSIDERATIONS
Intraoperative management has a major impact on surgical outcome and the role of the anaesthetist is central to this.
Monitoring
Due to the long duration of surgery, patients require a general anaesthetic with intubation. Adequate venous access and invasive arterial pressure monitoring help optimise flap perfusion and enable monitoring of haemoglobin and haematocrit through arterial blood gas analysis. Indwelling urinary catheterisation and core temperature monitoring aid cases in which volume control is complex. Central venous cannulation and cardiac output monitoring may further guide fluid therapy and vasoactive drug use.
Positioning
Positioning of the patient requires meticulous attention to avoid peripheral nerve and pressure injury. Careful positioning, padding, and passive intraoperative mobilisation with regular assessment of pressure areas are important.
Anaesthetic Technique
Volatile anaesthetic or propofol infusion can be used for maintenance. Each of the volatiles has its own advantages. Sevoflurane may attenuate ischemic reperfusion injury, isoflurane maintains microcirculatory flow, and desflurane has rapid offset in the context of long-duration surgery.5 Total intravenous anaesthesia may reduce stress-induced vasoconstriction, reducing postoperative nausea and vomiting, and produce a smoother recovery profile5 but may result in more movement because of lack of inhibition of spinal reflex so may require deeper muscle relaxation or remifentanil. Opioid options include remifentanil infusion or intermittent opioid. Key advantages of remifentanil include vasodilation, rapid control of arterial pressure, intraoperative analgesia, and less reliance on neuromuscular blockade, which can be useful if the surgery requires peripheral nerve monitoring, but postinfusion hyperalgesia may be problematic.5
Ventilation
Ensuring normoxia and normocapnia during the perioperative period are important for patient and flap survival. In both donor and recipient tissues, hypoxia induces catecholamine release and vasoconstriction. Hypocapnia results in vasoconstriction through local effect on vessels. Hypercapnia can lead to acidemia and reduce erythrocyte deformability as well as catecholamine release and subsequent vasoconstriction.2
Hemodynamic Goals
During the dissection phase of surgery, controlled hypotension may be required. This can be achieved by up-titrating the volatile agent,propofol, or remifentanil levels. Pharmacological agents that directly cause vasodilation are avoided due to risk of blood-flow steal away from the flap. During the microanastomotic and insetting phase, normotension targeting mean arterial pressure >70 millimeters of mercury (mmHg) ensures adequate flap perfusion.6
Fluid Therapy
A hyperdynamic circulation with high cardiac output, peripheral vasodilation, and a large pulse pressure difference provide optimal conditions for flap perfusion.
Various studies have demonstrated a direct relationship between increasing the quantity of intraoperative fluid and a rise in the rate of complications in free-flap surgery.7 An over restrictive fluid approach can result in hypotension and flap (and vital organ) compromise. Over zealous fluid administration to raise arterial pressure can cause interstitial oedema and compromise microcirculatory flow because of the lack of lymphatic drainage from free flaps.
Our recommendation is to use goal-directed fluid therapy, administering crystalloids or colloids based upon the patient’s response to a fluidbolus by looking at parameters such as pulse pressure variation, stroke volume variation, cardiac index, and systemic vascular resistance. Goal-directed fluid therapy achieving a stroke volume variation of <13% using a pulse contour analysis device improves oxygen delivery and avoids excess fluid.8 Goal-directed fluid therapy should also aim for urine outputs of >0.5 mL/kg/h and normal serum lactate.
Haematocrit
A target haematocrit of 30% to 35% achieves best balance between viscosity and blood flow while optimising oxygen carrying capacity.2Transfusion of packed red blood cells may be necessary to restore microcirculatory oxygenation.
Vasoactive Drugs
Effects of vasoconstrictive drugs on free-flap microcirculation is contentious. Vasoconstriction compromises viability of tissue flaps throughischemia and thrombosis in animal models.9 However, clinical studies in humans show that vasopressor use does not increase risk of flap loss, nor does it affect flap perfusion.10,11 Judicious use of metaraminol, ephedrine, or phenylephrine for normotension in patients with adequate volume replacement appears safe.5
Temperature Regulation
Hypothermia can be detrimental to the flap by inducing shivering, sympathetic vasoconstriction with suboptimal flap perfusion, coagulopathy, and increased risk of wound infection. Large tissue areas at both donor and recipient sites are exposed for prolonged periods during free-flap surgery resulting in hypothermia through evaporation. In addition, induction of general anaesthesia redistributes core temperature to peripheral tissues resulting in core temperature loss to colder external environments. It is important to prewarm patients prior to surgery to minimise redistribution of blood flow.
Higher ambient theatre temperature, forced-air warming devices, underbody warmers, and warmed intravenous fluids and blood products, as well as humidified gases, will help avoid hypothermia.
Thromboprophylaxis
Patients are at higher risk of developing thromboses due to prolonged immobilisation associated with lengthy free-flap procedures. Microvascular thrombosis can interfere with flap perfusion. Prophylactic perioperative unfractionated heparin (5000 units subcutaneously) inhibits thrombin aggregation and/or perioperative aspirin inhibits platelet function. In contrast, perioperative low-molecular-weight heparin confers no benefit on anastomotic thrombosis.2
Preventive therapy for deep venous thromboses and pulmonary embolism includes mechanical means (graduated compression stockings or pneumatic compression devices) and chemoprophylaxis with postoperative subcutaneous enoxaparin or unfractionated heparin for at least 14 days, when bleeding risk is considered minimal.2
Tranexamic acid
Intraoperative tranexamic acid use is popular as it reduces blood loss without increasing deep venous thrombosis. A recent literature review suggests tranexamic administration does not significantly increase the risk of flap complications.12
Analgesia
There are at least 2 operative sites in free-flap surgery and postoperative pain can increase sympathetic activity, and release catecholamines, which can compromise flap survival through vasoconstriction and vasospasm affecting recipient-site vessels. Optimal analgesia also facilitates early mobilisation and minimises thrombosis formation. Donor-site pain is often greater than the recipient-site pain, as the flap is insensate. Many patients may also become opioid tolerant and may develop chronic pain postoperatively. Multimodal, opioid-sparing analgesia is an ideal option.
The use of nonselective nonsteroidal anti-inflammatory drugs in free-flap surgery is contentious. In one observational study, ketorolac use forhead and neck free-flap surgery did not increase bleeding complications or risk of free-flap compromise.13 The selective COX-2 inhibitors, parecoxib and valdecoxib, may be detrimental to flap survival as they do not provide the antiplatelet effects of undifferentiated COX inhibitors.13However, randomised control trials are required to fully elucidate this area.
Regional anaesthesia in addition to general anaesthesia may be advantageous as it provides quality analgesia and regional sympatheticblockade, resulting in vasodilation with improved flap perfusion. Both central neuraxial and peripheral blocks may be appropriate. It is important to consider risk of hematoma formation for a neuraxial technique if an anticoagulant is being used perioperatively.
Emergence
Smooth emergence is achievable by many means. These include extubating in a deep plane of anaesthesia, or in a lighter plane while maintaining a low dose of remifentanil infusion or a bolus of intravenous lignocaine, or converting an endotracheal tube to a laryngeal mask airway to minimise airway stimulation. Straining, coughing, and retching can increase venous pressures and reduce flap perfusion and should ideally be avoided. A good antiemetic strategy may also contribute to smooth emergence.
POSTOPERATIVE CARE
Uncomplicated free-flap surgery patients can be managed safely without requiring high-dependency unit or intensive care unit level of admission.14
One of the key goals of care during the postoperative phase is to minimise secondary ischemia arising because of vessel spasm, intravascular thrombosis, and venous congestion secondary to hematoma or interstitial oedema. Measures to optimise flap perfusion intraoperatively can extend into the postoperative phase.
Regular flap monitoring using clinical observations are the ‘‘gold standard.’’ However, other adjuncts such as Doppler, implantable Doppler, microdialysis, florescence angiography, or near-infrared spectroscopy are useful postoperatively.15
Compromised flaps have a 75% salvage rate when managed in a timely manner.5 Definitive management of a failing flap is urgent re-exploration in theatre to reinstate microvascular anastomoses.
SUMMARY
Anaesthesia for tissue-flap surgery requires an approach aiming to optimise donor tissue perfusion and requires a goal-directed approach reflecting an understanding of physiological effects of anaesthesia technique together with techniques that support oxygen delivery, blood flow, and favourable coagulation.
References
- Novakovic D, Patel RS, Goldstein DP, et Salvage of failed free flaps used in head and neck reconstruction. Head Neck Oncol. 2009;1:33.
- Mak QH, Chan HT, Irwin Anaesthesia for plastic and reconstructive surgery. Anaesth Intensive Care Med. 2020;22(1):64-69.
- Adams J, Charlton Anaesthesia for microvascular free tissue transfer. Br J Anaesth CEPD Rev. 2003;3(2):33-37.
- Hill JB, Patel A, Del Corral GA, et Preoperative anemia predicts thrombosis and free flap failure in microvascular reconstruction. Ann Plast Surg. 2012;69(4):364-367.
- Nimalan Anaesthesia for free flap breast reconstruction. Br J of Anaesth. 2015;16(5) 162-166.
- Chalmers A, Turner MW, Anand R, et Cardiac output monitoring to guide fluid replacement in head and neck microvascular free flap surgery—what is current practice in the UK? Br J Oral Maxillofac Surg. 2012;50(6):500-503.
- Pearse RM, Ackland Perioperative fluid therapy. BMJ. 2012;344:e2865.
- Funk D, Bohn J, Mutch W, et Goal-directed fluid therapy for microvascular free flap reconstruction following mastectomy: a pilot study. Plast Surg (Oakv). 2015;23(4):231-234.
- Cordeiro PG, Santamaria E, Hu QY, et Effects of vasoactive medications on the blood flow of island musculocutaneous flaps in swine. Ann Plast Surg. 1997;39(5):524-531.
- Goh CSL, Ng MJM, Song DH, et al. Perioperative vasopressor use in free flap surgery: a systematic review and meta- analysis. J Reconstr Microsurg. 2019;35(7):529-540.
- Chen C, Nguyen MD, Bar-Meir E, et al. Effects of vasopressor administration on the outcomes of microsurgical breast Ann Plast Surg. 2010;65(1):28-31.
- Klifto KM, Hanwright PJ, Sacks Tranexamic acid in microvascular free flap reconstruction. Plast Reconstr Surg. 2020;146(4):517e-518e.
- Schug, Do NSAIDs really interfere with healing after surgery? J Clin Med. 2021;10(11):2359. doi:10.3390/ jcm10112359
- Panwar A, Smith R, Lydiatt D, et al. Vascularized tissue transfer in head and neck surgery: is intensive care unit–based management necessary? Laryngoscope. 2016;126(1):73-79.
- Kohlert S, Quimby AE, Saman M, et Postoperative free-flap monitoring techniques. Semin Plast Surg. 2019;33(1):13- 16.
This work by WFSA is licensed under a Creative Commons Attribution-NonCommercial-NoDerivitives 4.0 International License. To view this license, visit https://creativecommons.org/licenses/by-nc-nd/4.0/
WFSA Disclaimer
The material and content provided has been set out in good faith for information and educational purposes only and is not intended as a substitute for theactive involvement and judgement of appropriate professional medical and technical personnel. Neither we, the authors, nor other parties involved in its production make any representations or give any warranties with respect to its accuracy, applicability, or com- pleteness nor is any responsibility accepted forany adverse effects arising as a result of your reading or viewing this material and content. Any and all liability directly or indirectly arising from the use of this material and content is disclaimed without reservation.



