Neuroanaesthesia
KEY POINTS
- Awake craniotomy is commonly indicated for resection of tumours near or within the eloquent areas of the brain to maximize tumour resection while preserving the vital neurological functions.
- Efficient scalp anaesthesia is the cornerstone for a successful awake craniotomy procedure.
- The use of dexmedetomidine for sedation during awake craniotomy is gaining popularity due to its analgesic and sedative properties with minimal respiratory depression.
INTRODUCTION
Awake craniotomy (AC) is required for neurosurgical procedures when the patient is needed to be awake and cooperative during certain stage ofthe procedure. The history of AC dates back to ancient times when trepanation of the skull was used to treat epilepsy or to allow the escape of evil air, as revealed in archaeological findings.1
In modern history, Sir Victor Horsley performed the first AC in London in 1886 to excise an epileptic focus after brain stimulation. In the 20th century, Wilder Penfield—who was one of the pioneers in neuroscience—developed a technique of intermittent sedation and analgesia to facilitate intraoperative identification of epileptogenic foci and their excision.1,2
This tutorial discusses the rationale for using AC for brain surgery, perioperative management of the patients, theatre preparation, the different anaesthetic techniques, and the adverse events during AC.
INDICATIONS

Intraoperative electrical stimulation of the brain and cognitive monitoring of the awake person is the ‘‘gold standard’’ for cortical and subcorticalmapping of vital neurological functions, such as motor, sensory, speech, and cognitive cortices.3 AC is mostly indicated for excision of brain tumours near the eloquent areas of the brain to help achieve maximal resection of the tumour without compromising vital neurological functions (Table 1). This can reduce postoperative morbidity and improve the prognosis, which is directly related to the extent of tumour resection.4
AC is also used to minimise anaesthesia-induced interference during surgeries requiring intraoperative electrocorticography, such as duringexcision of epileptic foci, or to confirm the location of implanted electrodes during deep brain stimulation procedures during microelectrode recording and macro-stimulation.5 Cortical mapping can also be useful during resection of certain intracranial vascular lesions near critical areas or supplying important territories.6
ADVANTAGES
AC has several nonsurgical benefits over craniotomy done under general anaesthesia, especially when patients are selected appropriately. It can be associated with less postoperative nausea and vomiting, shorter hospital stays, and reduced hospital costs.3 It may allow for enhanced recovery after craniotomy with same-day discharge.3 Improved patient satisfaction and postoperative pain were also observed in patients who underwent AC.3,7
PREOPERATIVE PATIENT ASSESSMENT AND PREPARATION
The success of AC depends on appropriate patient selection. Apart from patient refusal, most contraindications to AC are relative and summarized in Table 1. The preoperative assessment is the ideal time to establish a good rapport with the patient, to explain the procedure in detail, and to answer patient questions.
Information leaflets and videos help relieve patient anxiety surrounding the procedure and give them an idea about what to expect on the day of surgery. The neurophysiologist and speech/language therapist usually meet the patient before the surgery to provide training on the tasks they have to perform intraoperatively.
Plasma levels of anticonvulsants should be checked before the surgery to ensure therapeutic efficacy. Steroids and antiepileptics should becontinued as scheduled, although the morning dose of antiepileptics is usually omitted on the day of AC for epilepsy surgery.5 Further details about perioperative management for craniotomy can be found in ATOTW 466 (‘‘Anaesthesia for Craniotomy and Brain Tumour Resection’’).8
INTRAOPERATIVE MANAGEMENT
Patient Positioning
Correct patient positioning is crucial during AC to ensure appropriate communication with the patient during cortical mapping.6 The head and neck of the patient should be kept in a relatively neutral position to enable the patient to vocalize for language testing and swallow secretions comfortably. Extreme neck rotation or flexion can complicate airway management by making intubation or insertion of supraglottic devices more difficult and should be avoided.
Theatre Preparation
Theatre preparation focuses on keeping the patient comfortable throughout the procedure to reduce the risk of failure of cortical mapping and the need for conversion into general anaesthesia. The operating table should be configured with adequate padding. Forced-air warming device as well as fluid warmers should be used to avoid hypothermia and intraoperative shivering.
During cortical mapping, the forced-air warmer is turned off to enable the patient to communicate clearly without the additional noise. The theatre temperature is turned up so that the patient remains warm during this period.
The use of transparent drapes is recommended, especially for claustrophobic patients.5 A different theatre layout is usually used during AC. The theatre bed is turned 90 degrees to the anaesthesia machine to allow access to the patient by the neurophysiologist and the anaesthetist.9 Again,to keep the noise level to minimum, warning signs such as ‘‘Awake craniotomy in progress’’ should be placed on theatre entrances to limit unnecessary staff flow. Additionally, the patient can request to have their mouth moistened for comfort and to prevent coughing whilst their head is secured in pins.
Ice-cold saline should be readily available on the scrub trolley for the surgeon to irrigate the brain in case of intraoperative seizures. Propofol should be readily available in case cold irrigation fails to terminate seizures.
INTRAOPERATIVE MAGNETIC RESONANCE IMAGING
AC can be conducted in in the interventional magnetic resonance imaging suite. Repeated imaging is obtained intraoperatively to determine theextent of tumour resection or the position of implanted devices.3 Providing anaesthesia in this suite has its own challenges to ensure safety of thepatient and staff, such as avoidance of ferromagnetic equipment, use of extra long ventilator tubing and infusion lines, and use of hearing protection devices.10
MONITORING
Besides routine monitors, an arterial line is often needed. Wide-bore intravenous access is essential, and all lines should be inserted on the ipsilateral side of the brain lesion if possible. This is to ensure that testing, such as upper/lower limb movement on the contralateral side can be done with ease. Monitoring depth of anaesthesia, such as with the bispectral index, can be helpful to adjust the level of sedation or anaesthesia and to allow faster patient awakening for cortical mapping.11 The use of urinary catheters is institution-specific as they can add to patient discomfort. However, if a urinary catheter is not placed, careful intraoperative fluid management must be initiated.
LOCAL ANAESTHESIA
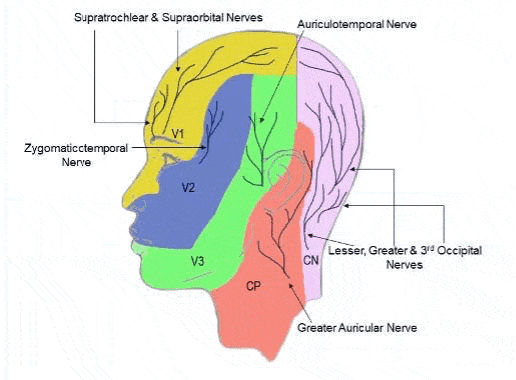
The pain-sensitive structures during craniotomy are the scalp, the periosteum, and the dura. The pia mater and the small cerebral vasculatures also receive trigeminal nerve supply. The scalp has a rich nerve supply arising from the trigeminal nerve supplying the anterior two thirds, and cervical spinal nerves (C2 and C3) supplying the posterior one third (Figure 1).5 Local anaesthesia (LA) of the scalp is the cornerstone for successful awake craniotomy.3 Patients with an effective scalp block will be comfortable and cooperative with minimal sedation during the surgery.

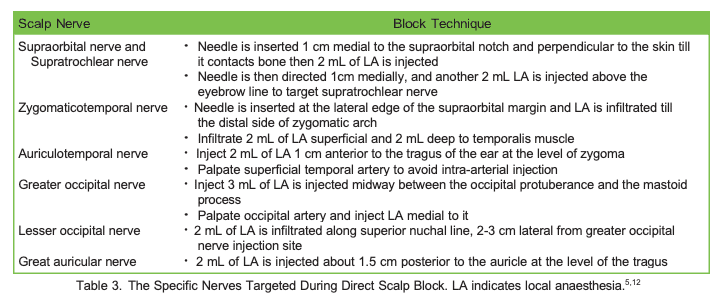
Different regional techniques have been described to provide LA to the scalp, but data are insufficient to support the superiority of one technique over another. The different techniques for scalp blocks are described in Tables 2 and 3 but combination of technique is common (eg, direct nerve block plus incision site infiltration). Bupivacaine, levobupivacaine, or ropivacaine are the commonly used LAs. Adrenaline in a concentration of 1:200 000 is usually added to LA solution to reduce the vascularity of the scalp and hence systemic LA absorption.5 Refer to ATOTW 480 on scalp block techniques.
ANAESTHESIA TECHNIQUES
Awake craniotomy procedure can be divided into 3 stages:
- craniotomy and exposure of the brain,
- cortical and subcortical mapping and excision of the brain lesion, and
- haemostasis and closure of craniotomy wound.
The patient needs to be awake and cooperative during stage 2. The stages before and after can be performed under local anaesthesia, sedation,or general anaesthesia.13 The commonly used anaesthesia techniques are the asleep–awake–asleep technique, the asleep–awake technique, or the conscious sedation approach.11
ASLEEP-AWAKE-ASLEEP TECHNIQUE
As its name would suggest, the patient initially receives general anaesthesia during the preparatory phase till opening of the dura. The patients is awakened for cortical mapping and neurophysiological monitoring. After completion of lesion resection, the patient is reanaesthetised during the third stage till the end of the surgery.
Total intravenous anaesthesia using target-controlled infusion of propofol and remifentanil is commonly used during the first stage of the surgery; dosage can be guided by careful titration during induction and use of intraoperative electroencephalogram monitoring. Target-controlled infusion technology is safe and can provide better drug titration to match individual needs with better cardio-respiratory stability.13
An alternative technique would be using volatile anaesthetic and remifentanil infusion. Total intravenous anaesthesia can have several potential advantages such as reduction of nausea and vomiting, smooth emergence, and short awakening times.3 It can also allow positive-pressure ventilation without the use of a muscle relaxant. Supraglottic airway devices (especially second-generation devices such as the ProSeal laryngeal mask or i-gel) are commonly used to secure the airway as they reduce the incidence of coughing and gagging during emergence from anaesthesia.5 They can be easily inserted to secure the airway while the patient’s head is in fixation pins during emergency situations or to re-anaesthetise the patient at the third stage of the procedure.14 After dural opening, the anaesthesia is stopped and the patient is awakened for neurophysiological monitoring and cortical mapping.
The advantage of the asleep-awake-asleep technique includes better control of the airway and ventilation and ensuring patient unawareness during the painful part of the procedure.11,13 In the case of longer procedures ( > 4 hours), asleep-awake-asleep might be a better option since the patient is more likely to cooperate with intraoperative testing if they are unconscious during the preparatory phase.3 Once the surgical resection is complete, the patient is again anesthetised using total intravenous anaesthesia and a supraglottic airway can be reinserted if needed.
A modification of this technique is asleep-awake approach, in which the patient is kept awake after brain lesion excision till closure of craniotomy wound. The patient anxiety usually lessens after lesion resection and mild sedation will be required to keep the patient reassured. It is recommended to supplement analgesia at that stage to ensure that the patient stays comfortable. This technique avoids airway manipulation while the head is fixed with pins in the Mayfield frame.5
CONSCIOUS SEDATION
The goal of the conscious sedation technique is to provide different levels of sedation depending on the stage of surgery while maintaining spontaneous ventilation and airway control during the procedure.11 Deeper levels of sedation are needed during the scalp block and initial craniotomy phase till dural opening. Sedation is then stopped or reduced for cortical mapping and testing. Once resection is complete,sedation is deepened to ensure patient comfort during the surgical closure. Sedation levels must be adjusted according to an individual’s needs as both oversedation and undersedation carry increased risk to the patient. Optimal sedation levels render a patient tranquil and able to give quick and clear response during mapping to provide meaningful neurological feedback with minimal hypoventilation and CO2 retention.3

Dexmedetomidine infusion (Table 4) or a combination of propofol and remifentanil target-controlled infusions are commonly used to provide sedation for AC. Dexmedetomidine is a selective α2 adrenoceptor agonist that is gaining popularity during AC. It has a sympatholyticaction as well as sedative, opioid-sparing, and some analgesic effects without significant respiratory depression. Also, it has minimal interference on neuronal functions and cortical mapping3,5,13. It can provide a comparable sedation efficacy to propofol and remifentanil infusion with fewer respiratory adverse events.15
Supplemental oxygen can be given by nasal prongs with an end-tidal CO2 sampling channel. There is a growing interest in using high-flow nasal oxygen in anaesthesia and intensive care, which can provide a higher ratio of arterial oxygen partial pressure to fractional inspired oxygen(PaO2/FiO2 ratio) than conventional oxygen therapy and might reduce respiratory-related complications during AC.16 Oxygen delivered via high-flow nasal cannulas is humidified and warmed, which can add to patient tolerance and comfort.
AWAKE TECHNIQUE
An alternative approach to AC relies only on effective LA without any sedation. This technique needs a highly motivated patient and short surgicaltime to be successful. Short-acting opioids such as remifentanil or patient-controlled analgesia can be used to supplement analgesia.17 Creating a good harmony within the operating room team, ensuring psychological support of the patient, and encouraging verbal and nonverbal communication are effective ways to avoid sedation during AC.3,18
COMPLICATIONS

AC is generally safe and well tolerated with low complication rates. Complications associated with AC are listed in Table 5. Failure of AC is uncommon (2%) and occurs mainly due to inadequate communication with the patient during the procedure, inappropriate patient selection, or persistent intraoperative seizures.5 Patients with bronchial asthma, remifentanil infusion, or prolonged surgery have been found to experience increased incidence of complications during AC.19 Cortical electrical stimulation during AC can induce focal or generalized seizures in about 8% of the cases but rarely results in AC failure.3,19 Seizures are usually self-terminated after stopping the stimulation or irrigation of the surgical field with ice-cold saline. Propofol or a thiopental bolus can be used to terminate persistent seizures but securing the patient’s airway might be needed if large doses are used.
Oxygen desaturation can result from airway obstruction or hypoventilation due to oversedation. It is more common with regimes using propofol and remifentanil infusions.3,5 Simple manoeuvres, such as jaw thrust, sedation adjustment, and oxygen supplementation are usually sufficient to resolve the problem. Different airway adjuncts including fibre-optic and video laryngoscopes should be readily available in case securing the airway is emergently needed. Laryngeal mask insertion has been found to have the highest success rate with shorter insertion time to secure the airway while the head is fixed in the Mayfield frame.14
Cerebral bulging (tight brain) may result from hypoventilation and CO2 retention and may require titration of sedation or control of the airway. Mannitol might be needed to reduce the brain oedema and improve the surgical field but will require the presence of urinary catheterisation.
It is not uncommon for the trigemino-cardiac reflex to be triggered during scalp block or LA infiltration. It is usually self-limited by cessation of the stimulation but might need anticholinergic medications to treat severe bradycardia.
SUMMARY
AC can carry many challenges for the patient as well as the anaesthesiologist. Different techniques have been described for anaesthesia and sedation management; however, good patient selection and preoperative preparation, effective local anaesthesia, and prompt treatment ofintraoperative complications are crucial for successful and safe procedure.
REFERENCES
- July J, Manninen P, Lai J, Yao Z, Bernstein The history of awake craniotomy for brain tumor and its spread into Asia.Surg Neurol. 2009;71(5):621-624.
- Todman Wilder Penfield (1891-1976). J Neurol. 2008;255(7):1104.
- Sewell D, Smith M. Awake craniotomy: anesthetic considerations based on outcome evidence. Curr Opin Anesthesiol. 2019;32(5):546-552.
- Bu L-H, Zhang J, Lu J-F, Wu J-S. Glioma surgery with awake language mapping versus generalized anesthesia: a systematic review. Neurosurg Rev. 2021;44(4):1997-2011.
- Jangra K, Rath Anesthesia for awake craniotomy: what is new? Curr Anesthesiol Rep. 2018;8(3):318-327.
- Bonhomme V, Franssen C, Hans Awake craniotomy. Eur J Anaesthesiol. 2009;26(11):906-912.
- Brown T, Shah AH, Bregy A, Shah NH, Thambuswamy M, Barbarite E, Fuhrman T, Komotar RJ. Awake craniotomy for brain tumor resection: the rule rather than the exception? J Neurosurg Anesthesiol. 2013;25(3):240-247.
- Keown T, Bhangu S, Solanki S. Anaesthesia for craniotomy and brain tumour resection. Accessed 1st April 2022. https:// resources.wfsahq.org/atotw/anaesthesia-for-craniotomy-and-brain-tumour-resection/
- Meng L, McDonagh DL, Berger MS, Gelb Anesthesia for awake craniotomy: a how-to guide for the occasional practitioner. Can J Anesth. 2017;64(5):517-529.
- Reddy U, White MJ, Wilson Anaesthesia for magnetic resonance imaging. Cont Educ Anaesth, Critic Care Pain. 2012;12(3):140-144.
- Burnand C, Sebastian Anaesthesia for awake craniotomy. Cont Educ Anaesth, Critic Care Pain. 2014;14(1):6-11.
- Osborn I, Sebeo ‘‘Scalp block’’ during craniotomy: a classic technique revisited. J Neurosurg Anesthesiol. 2010;22(3):187-194.
- Lobo F, Wagemakers M, Absalom, Anaesthesia for awake craniotomy. Br J Anaesth. 2016;116(6):740-744.
- Gruenbaum SE, Bilotta F, Bertasi TG, et al. Emergency airway management during awake craniotomy: comparison of 5 techniques in a cadaveric model. J Neurosurg Anesthesiol. 2022;34(1):74-78.
- Goettel N, Bharadwaj S, Venkatraghavan L, Mehta J, Bernstein M, Manninen P. Dexmedetomidine vs propofol-remifentanil conscious sedationfor awake craniotomy: a prospective randomized controlled Br J Anaesth. 2016;116(6):811-821.
- Yi P, Li Q, Yang Z, Cao L, Hu X, Gu High-flow nasal cannula improves clinical efficacy of airway management in patients undergoing awake craniotomy. BMC Anesthesiol. 2020;20(1):1-8.
- Ijaz MK, Moore,M. Remifentanil patient-controlled analgesia in awake craniotomy: an introduction of an innovative technique. J Neurosurg Anesthesiol. 2020;32(4):367.
- Hansen E, Seemann M, Zech N, Doenitz C, Luerding R, Brawanski Awake craniotomies without any sedation: the awake-awake-awake technique. Acta Neurochir. 2013;155(8):1417-1424.
- Abaziou T, Tincres F, Mrozek S, et al. Incidence and predicting factors of perioperative complications during monitored anesthesia care for awake craniotomy. J Clin Anesth. 2020;64: 109811.



