General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Deep brain stimulation can be used to treat:
a. Essential tremor
b. Dystonia and Tourette’s syndrome
c. Alcohol dependence
d. Obsessive-compulsive disorder
e. Depression - When managing patients with Parkinson’s disease for deep brain stimulator (DBS) implantation:
a. Patients should always take their usual anti-parkinsonian medications before surgery
b. Blood pressure control is important before and during the procedure
c. Deep paralysis is required during insertion of electrodes into the brain to avoid intracranial bleeding
d. Dexmedetomidine is commonly used as a sedative agent
e. Insertion of a urinary catheter is not always required - When managing patients with an implanted DBS:
a. A magnet should be readily available to deactivate the DBS during surgery
b. Bipolar diathermy can be used when the DBS device is active
c. External defibrillation is contraindicated when the DBS device is active
d. DBS devices can interfere with ECG monitoring
e. MRI is not absolutely contraindicated
Key Points
- Deep brain stimulators are increasingly being used to treat movement and psychiatric disorders.
- Careful patient selection improves successful outcome of DBS surgery.
- Multi-disciplinary preoperative preparation is essential to avoid serious complications.
- An “awake and sedate” technique is commonly used for DBS insertion.
INTRODUCTION
Deep brain stimulation is an increasingly common treatment for Parkinson’s disease (PD), other movement disorders, certain psychiatric conditions, and chronic pain syndromes. The insertion of a DBS is a minimally invasive procedure that places electrodes into specific locations of the brain for clinical testing, which are then connected to an implanted pacemaker (stimulator). Several anaesthetic techniques are currently used to perform this procedure depending on the experience of the local institution, individual practitioner, and patient-specific factors. Patients presenting for surgery with these devices require special consideration due to potential interference between the neurostimulator and other medical devices. This tutorial will review current anaesthetic techniques and recommendations for inserting and managing deep brain stimulators.
BACKGROUND
A common feature of movement disorders is the inability of patients to control their body’s movement due to an imbalance of neurotransmitters. PD is caused by the progressive degeneration of dopaminergic neurons in the basal ganglia. Historically, the surgical removal of lesions in deep brain structures (like the thalamus and globus pallidus) was an effective treatment for these disorders. Since the late 1960s surgical resection has become less popular due to its high complication rates and improved medical therapy.1
Thalamic deep brain stimulation was first developed for tremor control in the 1980s. Later on, sub-thalamic nucleus (STN) and internal globus pallidus (GPi) stimulation were also investigated. Targeting the STN can improve a wide range of parkinsonian symptoms and decrease the need for PD medication. The STN is currently the primary target for most patients with this disease. When STN deep brain stimulation is contraindicated, the thalamic route remains an option for patients with severe tremor, and GPi stimulation can be used to treat severe dyskinesia and dystonia.
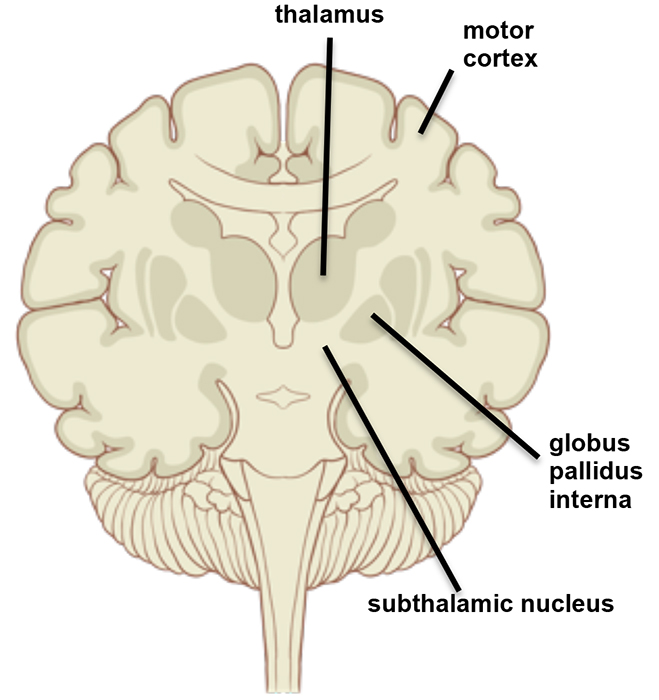
Figure 1: Common targets for surgery: globus pallidus, thalamus and subthalamic nucleus Source: Patrick J. Lynch, distributed under Creative Commons Attribution 2.5 License 2006
The DBS hardware has three main components:
- intracranial electrodes
- a programmable internal neurostimulator
- an extension cable connecting the DBS electrodes to the neurostimulator.
The surgical procedure consists of two stages:
Stage one involves mapping and placement of the intracranial electrodes.
Stage two involves implanting and connecting the neurostimulator, sometimes referred to as an implantable pulse generator (IPG). The whole procedure may be completed on the same day, or in a 2-stage procedure. The decision to separate the stages is usually based on the patient’s condition, team preference, and local hospital experience.
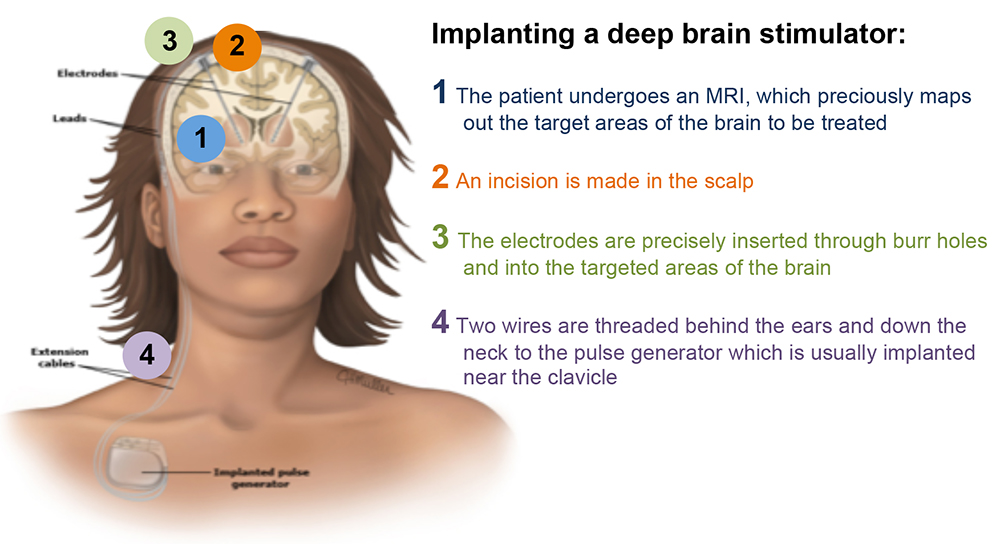
Figure 2: Components of a DBS. Reproduced with permission from UpToDate
PREOPERATIVE EVALUATION
Success in DBS surgery starts with careful patient selection during the preoperative evaluation. Determining suitability involves assessing a patient’s general physical condition, psychiatric history, and cognitive function. A multidisciplinary team involving anaesthetists, neurologists, neurosurgeons, neuropsychologists, and specialist nurses is essential to ensure all aspects of the patient’s care are evaluated. Contraindications include factors that either increase the operative risk, the risk of device malfunction, or those that may reduce the effectiveness of the DBS (Figure 3).
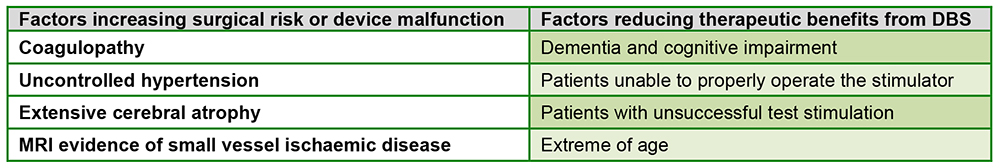
Figure 3. Factors that increase the operative risk or the risk of device malfunction, and those that may reduce the effectiveness of the DBS
The patient’s medication regimen, especially with PD and dystonia, needs to be carefully reviewed. Antiplatelet agents should be withheld, if possible, before and immediately after surgery. The need for chronic anticoagulation is not an absolute contraindication to surgery, but careful perioperative anticoagulation management is warranted. For diseasespecific drugs, clear instructions for continuation or discontinuation should be given to patients at the discretion of the multidisciplinary team, as some patients have to be in a “drug-off” state to elicit intraoperative mapping and clinical testing. If symptoms are severe, a reduced dose of their regular medication may be an alternative option.
Apart from ensuring the patient is medically optimised for the procedure, patients with PD commonly suffer from obstructive sleep apnoea or have an increased risk for aspiration. A more vigilant assessment for risks of aspiration and obstructive sleep apnoea are important when considering any sedative technique in this setting. If an awake surgical technique is decided on for the surgery, options of securing the airway at any stage of the procedure are important to have prepared in advance.
Patient cooperation during the awake part of the surgery is imperative for success (see below). Identification of factors which may complicate this (such as developmental delay, dementia, communication difficulties, claustrophobia, or previous difficulty with sedation) warrants special attention. Preoperative explanation of the intraoperative course can help reassure the patient, alleviate anxiety, identify potential intraoperative challenges, and improve cooperation during the procedure.
ANAESTHETIC MANAGEMENT OF DBS INSERTION
Stage one
A stereotactic frame is fixed to the patient’s head, usually under a combination of local anaesthesia/scalp block and sedation. General anaesthesia may be required if patients cannot remain still during the procedure. Once the frame is in place, an MRI scan of the brain is performed to establish the coordinates for precise electrode placement.
Intraoperatively, the stimulating electrodes are inserted via a burr-hole craniotomy. Unless the patient is not an appropriate candidate, monitored anaesthetic care with sedation is the most commonly used technique for this stage of the operation. Maintaining the ability to interact with the patient is essential to facilitate microelectrode localisation and clinical testing, which helps to confirm optimal electrode position, and detection of side-effects. The tests are more difficult to perform, and less accurate, under general anaesthesia. The other advantages of an awake technique include less haemodynamic fluctuations and postoperative nausea and vomiting, when compared to general anaesthesia.
Standard anaesthetic monitoring is recommended. Invasive arterial blood pressure monitoring is not necessary, but may be considered based on the clinical status of the patient.
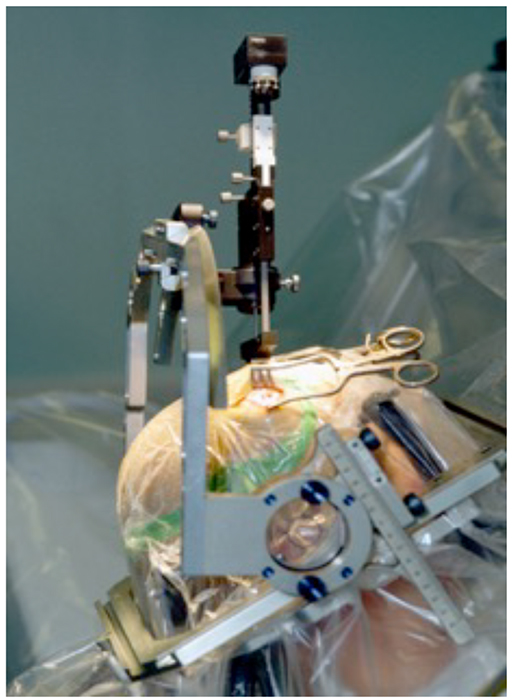
Figure 4. Electrode insertion during surgery
Intraoperatively, several key points need to be addressed:
- Oxygen is often used via a mask or nasal prongs attached to the frame.
- Temperature and positioning are important to ensure patient comfort.
- Draping should allow access to the patient’s face, arm and legs, maintain sterility, and avoid oxygen/carbon dioxide accumulation.
- A urinary catheter is not necessary, but conserving fluid administration is essential to avoid bladder over-distension.
- At all times during the procedure, tools to remove the head frame for emergency airway access must be available.
Stage two
The electrodes are connected to an external cable which is then tunnelled subcutaneously under the scalp, down the neck, and finally to the site of the IPG implantation. The IPG and battery unit are commonly implanted in the chest wall or abdomen under general anaesthesia. There are no specific anaesthetic drug considerations for IPG insertion.
Postoperative neurological observation is necessary after the first stage of the procedure, regardless of the type of anaesthetic technique utilised, as it is an intracranial operation. This is not as necessary for the second stage of the procedure, if it is carried out as a separate operation. Anti-parkinsonian medications should be recommenced as soon as possible, to avoid deterioration in motor and neurological function. Patients usually require assistance with daily activities for the first few weeks post-insertion, and are instructed not to drive for at least the first 6 weeks.
Choice of sedation regimen
Where available, dexmedetomidine has become a commonly used agent. It reliably provides sedation with minimal respiratory depression and attenuates intraoperative haemodynamic swings. Propofol is also commonly used during lead placement, especially in the STN, but the extent to which propofol interferes with microelectrode recordings (MER) is unclear. Propofol may cause dyskinesia and abolish tremor. Short acting opioids have minimal effect on MER, but high doses can cause worsening of rigidity. Benzodiazepines are not recommended as they abolish MER and interfere with stimulation testing.2 The sedation regimen used will vary by institution, but the goal is to minimize any effect on subcortical activity, thus optimising microelectrode recording and clinical testing (Figure 5).
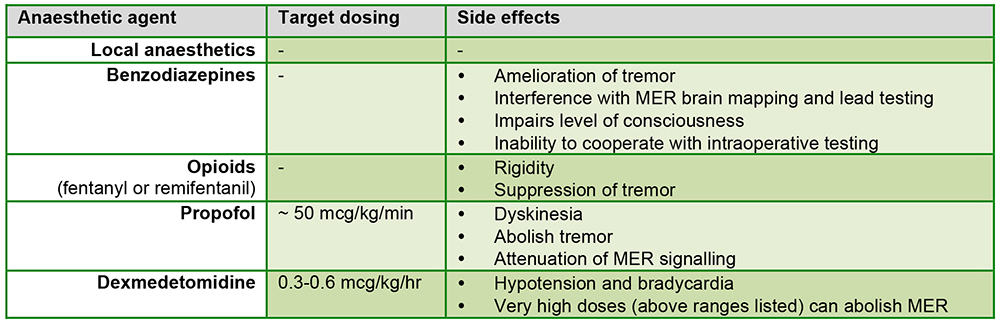
Figure 5. Table showing the side effects of commonly used anesthetic agents on MERs
DBS insertion is generally very safe but, as with all surgery, there are perioperative risks and complications.3 A small case series identified an intraoperative complication rate of approximately 7%. Major complications include intracranial bleeding (0.4-3.6%), seizures (0.8-4.5%), strokes, neurological deficit (0.3-0.6%), and post-procedural delirium. Other intraoperative complications include airway obstruction (1.6-5.5%), hypertension, hypotension, or venous air embolism (1.6-3.5%). Complications related to the device itself include infection, equipment failure, and electrode migration.
ANAESTHETIC MANAGEMENT OF PATIENTS WITH DBS
Preoperative preparation includes identification of the device and the severity of the patient’s symptoms when the DBS implant is turned off. Supplementation with oral medications may need to be considered if the device needs to be deactivated and symptoms are severe.
Figure 5. DBS Programmer
DBS systems may potentially interfere with monitoring and therapeutic equipment in the operating room, often producing artefacts on ECG readings. When diathermy is required, the neurostimulator may need to be disabled before induction of anaesthesia with the DBS Patient Programmer.
Bipolar diathermy should be used where possible, to reduce the risk of thermal injury to brain tissue around the implanted electrode or inadvertent reprogramming of the device. If monopolar diathermy is necessary, the grounding pad should be placed far away from the IPG and the lowest possible level of energy should be used, and only in short pulses. Short wave (microwave and ultrasound) diathermy should be avoided, as it has been linked to significant brain injury in patients with DBS.
Patients may experience rigidity once the device is switched off, and mechanical ventilation is sometimes necessary in the rare situation where severe rigidity interferes with their ability to breathe. Postoperatively, the device should (ideally) be turned back on before awakening.
OTHER CLINICAL CONSIDERATIONS
Guidelines for external defibrillation on patients with a DBS have not been established, but in critical situations, a DBS should not preclude their use. When needed, defibrillation paddles should be placed as far away from the neurostimulator as functionally possible. Interrogation of the DBS is recommended after external defibrillation.
Generally, MRI is possible in patients with implanted DBS devices, as long as manufacturer guidelines are followed and scan time is minimized. However, there are reports of electrode overheating, leading to brain injury, as well as damage and accidental reprogramming of the device. Consultation with your Radiologist is recommended prior to proceeding.
With current evidence, it is safe for patients to receive a DBS device when they have a cardiac pacemaker in situ. Advanced planning is paramount to minimise interference between the devices. As MRI may be contraindicated in patients with a cardiac pacemaker, stereotactic CT has been used for nuclei mapping as an alternative.
Electroconvulsive therapy, radiofrequency neuro-ablation, and peripheral nerve stimulation have also been shown to be safe in patients with a DBS in situ, if the stimulator is turned off and the probes are distant from the generator.
SUMMARY
- Deep brain stimulation is an expanding modality for treating movement and psychiatric disorders.
- An “awake and sedate” technique is most commonly used, as it facilities intraoperative mapping and neurological testing, with decreased complications.
- Dexmedetomidine is proving to be a popular anaesthetic agent for electrode implantation.
- Good preparation is essential for the management and insertion of deep brain stimulators.
ANSWERS TO QUESTIONS
- a. True: Deep brain stimulation is used to treat essential tremor.
b. True: Deep brain stimulation is used to treat other movement disorders, like dystonia and Tourette’s syndrome.
c. False: Currently there is no evidence for treating alcohol dependence with deep brain stimulation.
d. True: Deep brain stimulation has been used in obsessive-compulsive disorder with encouraging results.
e. True: Deep brain stimulation is an experimental therapy for treatment-resistant depression. - a. False: Most institutions prefer to withhold anti-parkinsonian drugs on the day of surgery to facilitate brain nuclei mapping and clinical testing. A reduced dose can be considered in patients with severe symptoms.
b. True: Blood pressure control is essential during the intraoperative period to maintain adequate brain perfusion and avoid intracranial haemorrhage.
c. False: “Awake and sedate” techniques are commonly used for electrode insertion. General anaesthesia is usually only considered when patients cannot tolerate the procedure awake. Deep paralysis is not routinely indicated.
d. True: Dexmedetomidine is commonly used as a sedative agent during deep brain stimulator implantation, as it has minimal effect on MER.
e. True: Insertion of a urinary catheter is not always required, but vigilant monitoring of fluid administration is essential to avoid bladder over-distension. - a. False: The DBS is programmed by an external, hand-held device, placed directly over the battery-stimulator unit. A magnet does not have any effect on the device.
b. True: When diathermy is required, the neurostimulator may need to be disabled before induction of anaesthesia. Bipolar diathermy should be used where possible to reduce the risk of thermal injury to brain tissue or inadvertent reprogramming of the device.
c. False: External defibrillation may damage the neurostimulator, but should not be withheld when needed.
d. True: DBS systems may potentially interfere with other monitoring and therapeutic electronic equipment.
e. True: MRI scanning anywhere on the body is possible with some of the new implanted DBS devices.
REFERENCES
- Miocinovic S, Somayajula S, Chitnis S, et al. History, applications and mechanisms of deep brain stimulation. JAMA Neurol 2013;70:163-171.
- Venkatraghavan L, Luciano M, Manninen P. Anesthetic management of patients undergoing deep brain stimulator insertion. Anesth Analg 2010;110:1138-1145.
- Poon CCM, Irwin MG. Anaesthesia for deep brain stimulation and in patients with implanted neurostimulator devices. Br J Anaesth 2009;103:152-165.
FURTHER READING
- Binder DK, Rau GM, Starr PA. Risk factors for haemorrhage during microelectrode-guided deep brain stimulator implantation for movement disorders. Neurosurgery 2005;56:722-732.
- Khatib R, Ebrahim Z, Rezai A, et al. Perioperative events during deep brain stimulation: the experience at Cleveland clinic. J Neurosurg Anesthesiol 2008;20:36-40.



