General Topics
QUESTIONS
Try to select the correct answers from the following statements. The answers can be found at the end of the article, together with explanations.
- Atrial Fibrillation:
a. The incidence increases with age
b. On an ECG is identified by the absence of p waves and a regularly irregular rhythm.
c. A CHADS2 score of greater than 3 suggests a high risk of thromboembolic complications
d. All patients in AF require treatment with warfarin
e. The a- wave on the JVP is more prominent in patients with AF - Atrial Fibrillation treatment options:
a. β-blockers are first choice agent for all patients with AF
b. Digoxin is a useful agent for acute rhythm control
c. Amiodarone is first line agent in haemodynamically unstable AF
d. Nifedipine is an effective treatment option in AF
e. After elective cardioversion and restoration of sinus rhythm, warfarin can be stopped immediately afterwards - The following may precipitate AF:
a. Hypokalaemia
b. Hyperkalaemia
c. Hypomagnesia
d. Hypovolaemia
e. Sepsis
INTRODUCTION
Atrial Fibrillation (AF) is the commonest arrhythmia. It is associated with increasing age and results in significant morbidity. The management of AF is not straightforward and requires consideration of several aspects including the recognition of precipitating factors, appropriate anticoagulation, and choosing a suitable method to either restore sinus rhythm or achieve rate control. This article aims to cover the key points regarding management of AF during the perioperative period.
The estimated prevalence of AF is 1-2%, and is increasing in developed countries as the population ages. It is difficult to estimate the perioperative incidence of AF, but it has been reported to be as high as 30% after coronary artery bypass graft (CABG) surgery. It is associated with significant morbidity and mortality, which includes an increased risk of heart failure, stroke and prolonged hospitalisation.
Medical management is centred round three core strategies:
- Assessment of the thromboembolic risk by using appropriate risk stratification scores and implementing anticoagulation if deemed necessary
- Restoration of sinus rhythm or rate/symptom control
- Treatment of the precipitating factors
Surgical patients can present in AF in a variety of ways and a useful way of classifying it in this population is:
- Pre-existing AF
- New onset AF
- Paroxysmal atrial fibrillation (PAF)
From a management perspective these various presentations of AF can be further classified according to whether they occur pre-operatively, intra-operatively or post-operatively.
PRE-EXISTING ATRIAL FIBRILLATION
Pre-operative management
According to the National Institute for Health and Care Excellence (NICE) guidelines on pre-operative investigation, an ECG should be considered for patients over the age of 40 who are assessed as ASA II or above, and is recommended for patients with a history of cardiovascular disease. A resting ECG will reveal AF by the absence of P waves and QRS complexes that occur in an irregularly irregular rhythm (figure 1). For patients with known AF, a transthoracic echocardiogram (TTE) is useful to ascertain the ejection fraction, exclude valvular heart disease (especially aortic or mitral valve involvement), and to look for regional wall motion abnormalities (RWMA).
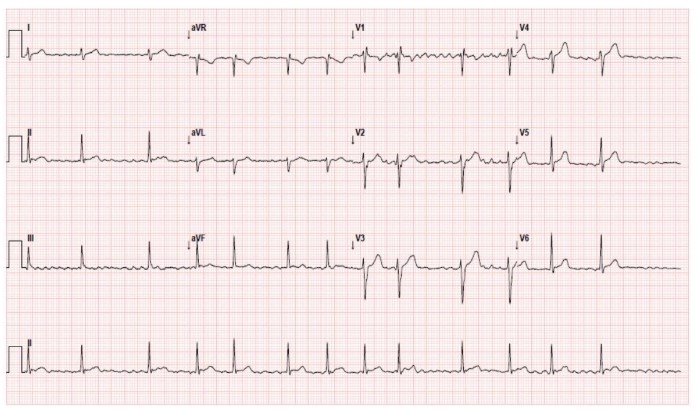
Figure 1: A 12 Lead ECG showing narrow QRS complexes which are irregularly irregular and are not preceded by P waves.
Patients with persistent or permanent AF should have their heart rate adequately controlled prior to an elective operation. This is often achieved by beta blockade or centrally acting calcium channel antagonists (CCB). The benefits of beta blockade are well documented, and include reduction in myocardial infarctions and the development of new AF. However recent evidence has shown there may be an increased mortality and risk of stroke in patients taking beta blockers, so cessation of medication should be based on individual cases prior to surgery. Medications such as verapamil and diltiazem are often omitted on the day of surgery owing to their associated hypotension and profound bradycardia under anaesthesia, but there are no clear guidelines and decisions must be individualised. If the decision is made to continue current rate control medication, then patients should take their medication at their usual times despite nil by mouth restrictions.
The other important aspect to consider is anticoagulation. Scoring systems such as CHADS2, and the newer CHA2DS2-VASc (table 1), stratify patients based on the NICE guidelines, and the scores predict the patient’s annual risk of stroke (table 2). Patients at low risk (CHADS2 score < 1) require no anticoagulation, while those at high risk (CHADS2 score of 2 or more) require warfarisation to a target INR of 2-3, or an equivalent level with new anticoagulants such as Dabigatran. Traditionally, patients with an intermediate score (CHADS2 of 1-2) were offered a choice between aspirin or warfarin, but aspirin has now been shown to be inferior to warfarin in stroke prevention. Therefore anyone with a score > 1 should probably also be warfarinised if there are no contraindications. Prior to surgery it is important to ensure that anticoagulation is managed appropriately.
The risk of thromboembolism in patients who discontinue anticoagulation for a short period (≤ 5 days) before an invasive procedure must be weighed against the risk of bleeding if these agents are continued. Therefore, the thrombotic and bleeding risks should be carefully assessed before deciding on how best to modify the anticoagulation. There is no good evidence that a short period of warfarin interruption is harmful for low to intermediate risk patients. For patients at very high risk for thrombosis (eg, CHADS2 ≥4, CHA2DS2-VASc ≥5), consider the use of bridging anticoagulation with intravenous heparin, therapeutic dose low molecular weight (LMW) heparin (eg, enoxaparin 1 mg/kg subcutaneously twice daily), or an intermediate-dose bridging regimen (eg, enoxaparin 40 mg subcutaneously twice daily). For patients undergoing low bleeding risk procedures (e.g.skin lesion excisions), there is no need to alter their anticoagulation treatment, provided the INR is within the therapeutic range. A useful strategy would be to employ sub-therapeutic doses of LMW heparin in low and intermediate risk patients, which is a safe compromise between the risks of bleeding and thrombosis.
For patients at a high risk of procedural bleeding warfarin should be stopped five days preoperatively to achieve an INR ≤1.5. If bridging anticoagulation is used then unfractionated heparin should be stopped 4-5 hours before surgery, and LMW heparin stopped 24 hours before surgery. Therapeutic dose intravenous or LMW heparin should not be restarted postoperatively until at least 24 hours after surgery and delayed longer (48-72 hours) if there is major surgery, a high risk of bleeding, or if there is any evidence of on-going bleeding. When warfarin is restarted the INR should be in the therapeutic range for at least 48 hours before heparin is discontinued.
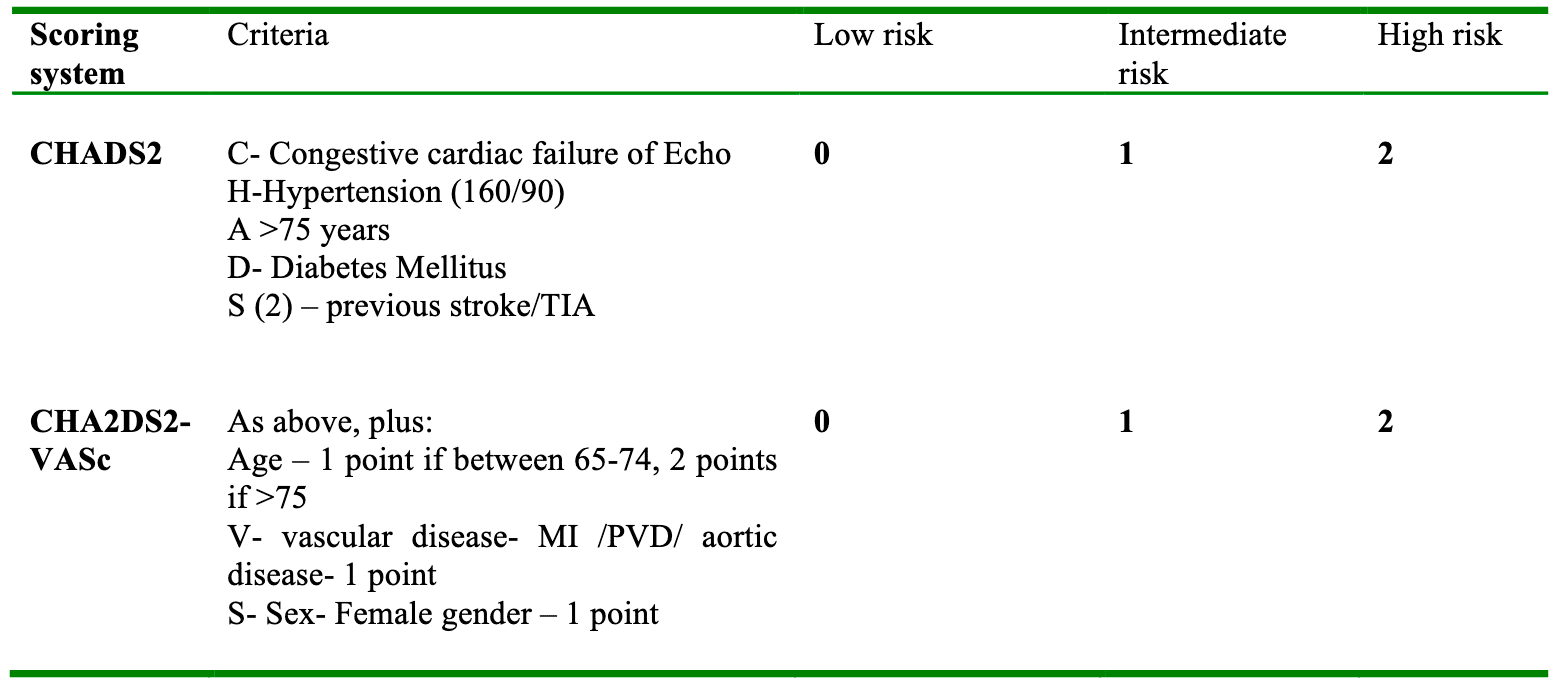
Table 1: Risk stratification scores adapted from BMJ BAFTA trial. A score of one point is added for each risk factor. The CHA2DS2-VASc system incorporates additional risk factors.
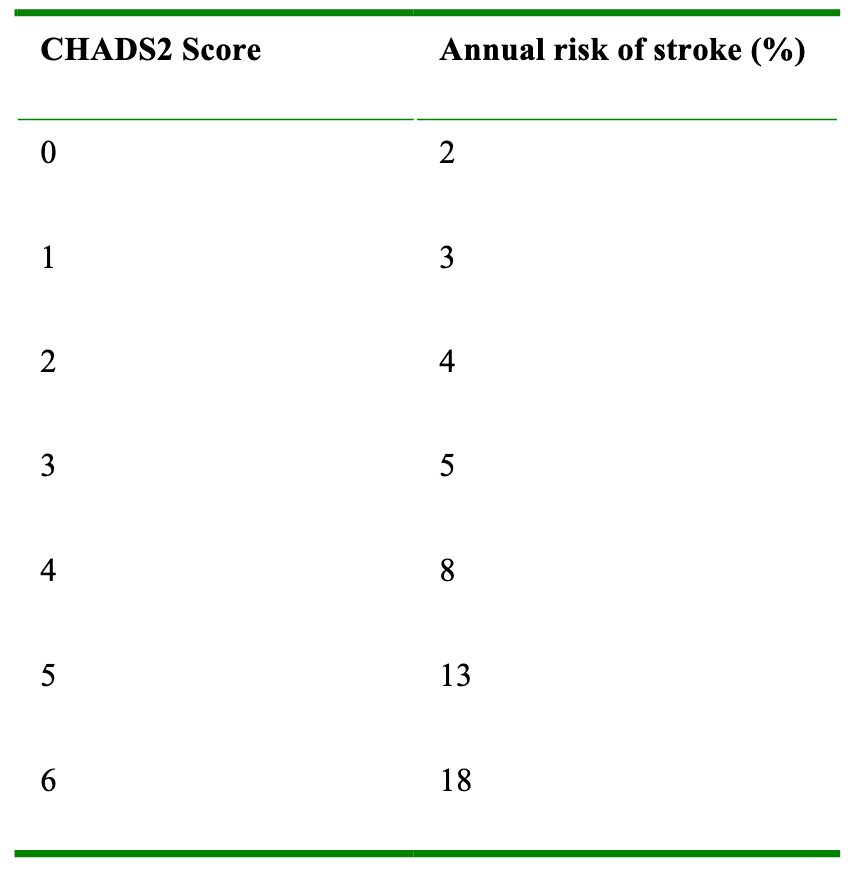
Table 2. Percentage increase in risk of stroke per annum with an increase CHADS2 score
Intra-operative management
AF with a normal ventricular rate is usually asymptomatic and does not cause any major anaesthetic problems. However AF with a rapid ventricular response can cause significant cardiovascular complications, including hypotension, rate related myocardial ischaemia, heart failure and pulmonary oedema. Normal atrial activity accounts for at least 10% of ventricular filling, which can increase up to 40% at higher heart rates. The elderly population are more dependent on atrial filling owing to reduced elasticity of the ventricles. For this reason the adverse effects of AF with a fast ventricular response are more profound in the elderly and those with underlying cardiovascular disease. The first step in managing these patients is to identify any precipitating causes (table 3) and attempt to correct these. Treatment should be commenced when adverse signs develop, such as rate related ischaemia evidenced by ST segment changes on continuous ECG monitoring, or hypotension secondary to the tachycardia. There are no clear guidelines as to which pharmacological agent is preferred, but intravenous options are given in table 4. All intravenous drugs should be titrated to effect, and have the common side effect of exacerbating hypotension.
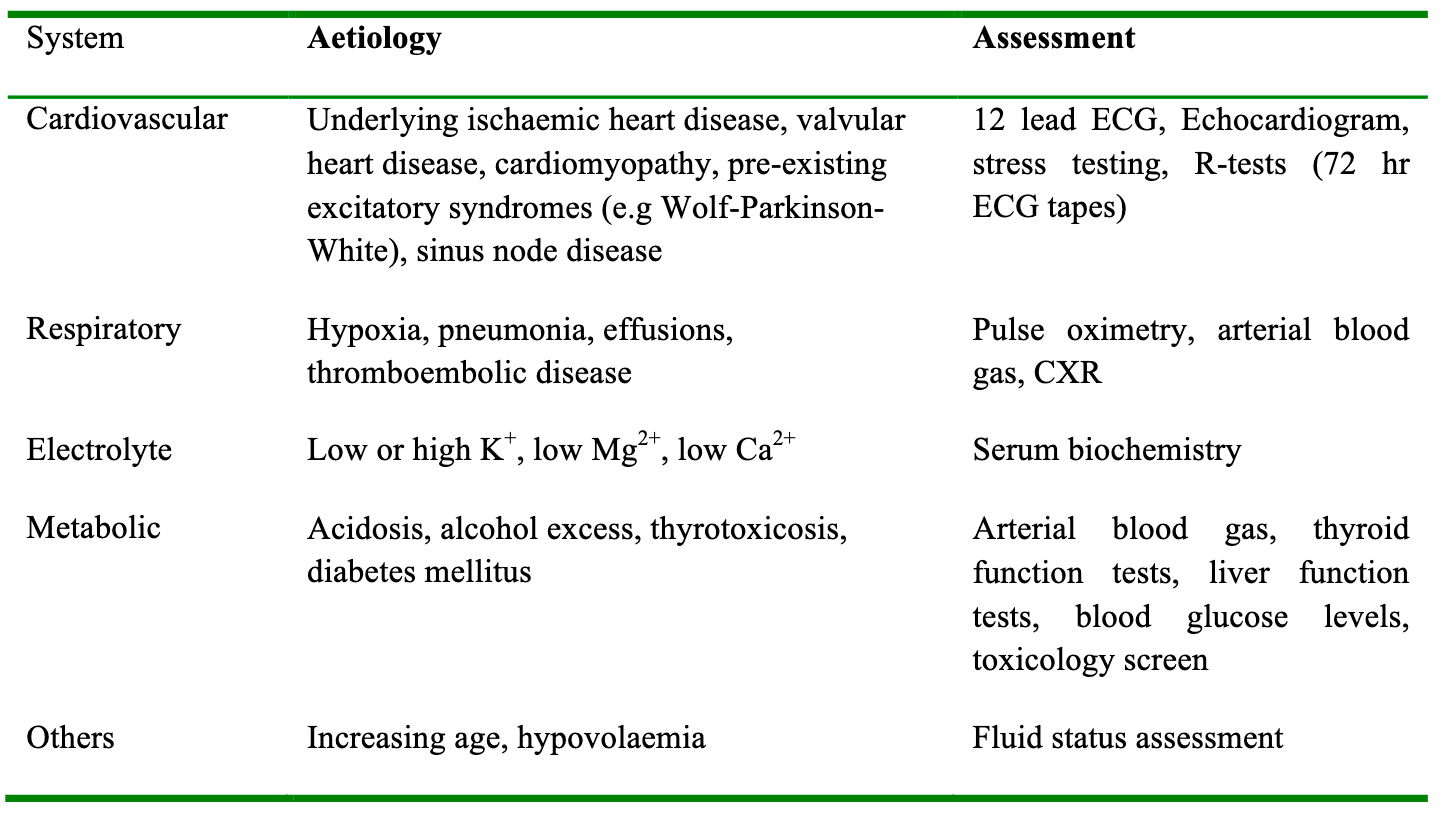
Table 3. Risk factors for the development of atrial fibrillation in patients undergoing surgery
Post-operative management
Patients should continue their usual rate control medication peri-operatively and timing of recommencement of anticoagulation should be discussed with the surgical team. A haematologist’s opinion should be sought for patients at high risk of thromboembolism as well as at high risk of bleeding complications. All post-operative patients are at an increased risk of thromboembolic disease, which further adds to the risk factors described by the CHADS2 score. A strategy for prevention of thromboembolism often includes low molecular weight Heparin (LMWH), which is maintained while warfarin is restarted and a therapeutic INR range is attained. Unfractionated heparin may be a preferred option for patients who are at a high risk of both bleeding and thromboembolic complications, because it can be reversed and titrated to effect more easily.
NEW ONSET ATRIAL FIBRILLATION
Pre-operative management
There are multiple risk factors for the development of new onset AF (table 3), which carries a higher risk in patients with pre-existing cardiac disease. Patients should always be optimised before undergoing elective surgery, and there is frequently opportunity for some optimisation in patients listed for emergency surgery. In the case of time-critical emergencies, management may have to occur intra-operatively to avoid delay.
Intra-operative management
In new onset AF the most important factor in order to guide management is the time of onset (in order to calculate the duration of the episode of AF). Usually this can be fairly accurately deduced since most patients have continuous ECG monitoring instituted at the start of any anaesthetic. Also, pre-anaesthetic assessment by pulse check and a recent 12-lead ECG may help in determining the time of onset. The significance of the duration of AF relates to the risk of developing atrial thrombus after 48 hours. Within the first 48 hours the risk of thrombus formation is low and is unlikely to lead to embolism after reversion to sinus rhythm. However, if the AF duration exceeds 48 hours, or if no clear time onset can be determined, thrombi may have formed in the fibrillating atria and there is a significant risk of embolism into the cerebral circulation if sinus rhythm is re-established.
It is important to correct any precipitating factors such as sepsis, hypovolaemia, hypoxia, and electrolyte imbalances, as well as any de-compensation of underlying disease such as cardiac disease or poorly controlled diabetes. Some factors can be promptly corrected intra-operatively, such as hypovolaemia, hypoxia and electrolyte disturbances, but others (such as sepsis) take longer to resolve so that the 48-hour safety window may be breached. If cardioversion to sinus rhythm is contemplated beyond 48 hours, cardiac imaging by transthoracic echocardiogram (TTE) or preferably transoesophageal-echocardiogram (TOE) should be performed to exclude thrombus formation in the atria.
If new onset AF is of less than 48 hours duration and precipitating factors have been corrected as far as possible, then medical treatment may be necessary without the need for anticoagulation. There are various agents available for intravenous use (table 4).
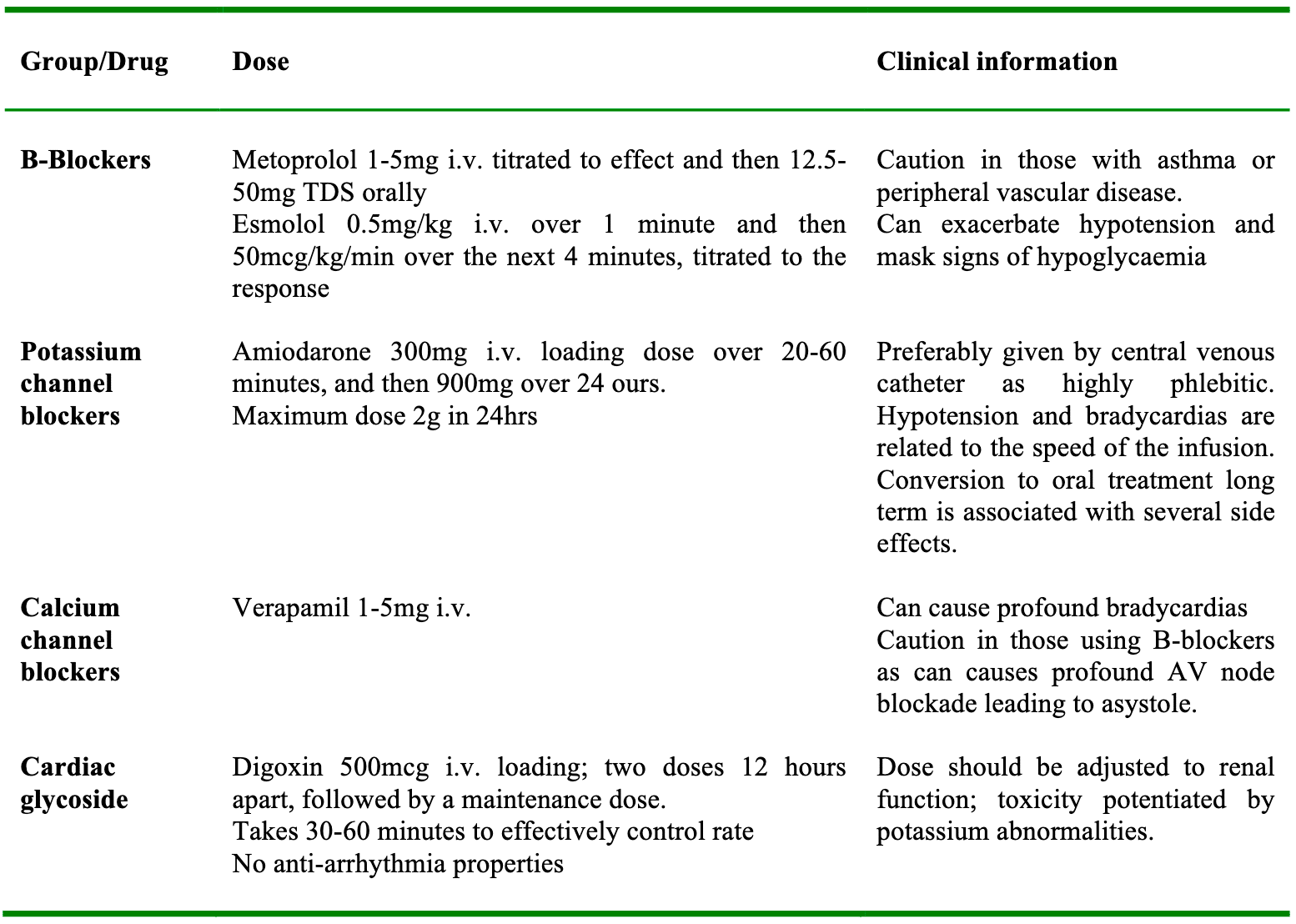
Table 4. Common pharmacological agents used to treat AF.
The quickest method for converting new onset AF back to sinus rhythm is by direct current cardioversion (DCCV). However, this is normally reserved for patients who are haemodynamically unstable. DCCV requires a defibrillator that is able to deliver a synchronized shock at an appropriate energy, as well as adequate sedation or anaesthesia for the patient. The recommended dose for emergency cardioversion of AF is 120-150 Joules with a biphasic defibrillator, which can be increased with subsequent shocks. The reason for synchronising the shock is to minimise the chance that the shock is delivered during the relative refractory period of the cardiac cycle (corresponding to the T wave), which can precipitate ventricular fibrillation.
For patients where precipitating factors have been treated and time of onset is uncertain or is beyond 48 hours, management should be directed at rate control, especially if the patient is significantly tachycardic or symptomatic. Anticoagulation should be commenced at the same time for the reasons discussed above.
Post-operatively
Patients with new onset AF who have responded to medication such as B-blockers or Amiodarone may require maintenance doses to ensure sinus rhythm is maintained. Correction of precipitating factors is important to prevent further relapses of AF. To optimise ongoing care there should be clear documentation of all circumstances relating to the AF, including treatment and potential causes, so that the physicians responsible for their post-operative care are fully informed. If rate or rhythm control is successful and patients are asymptomatic then further outpatient assessment by echocardiogram and referral to an atrial fibrillation clinic should be arranged. Issues that will need to be addressed include maintenance of rate control and anticoagulation to a therapeutic INR range of 2-3 for at least 4-6 weeks prior to a planned DCCV. During this time the patient should be under the care of cardiologists where possible. If the DCCV with three synchronised shocks fails to cardiovert the rhythm, they should be maintained on their anticoagulation treatment depending on their risk stratification (table 1). After successful DCCV there is a chance that patients may revert back to AF, which is why they should maintain anticoagulation to an INR of 2-3 for at least 4 weeks post cardioversion. At this point reassessment should occur and anticoagulation stopped if sinus rhythm is maintained. Anticoagulation in the immediate post operative period may be a difficult decision, and the risk of bleeding should be balanced against the risk of thromboembolic disease, giving an individualised decision for every patient
PAROXYSMAL ATRIAL FIBRILLATION (PAF)
This can be a difficult group of patients to manage. By definition PAF arises spontaneously and reverts back to sinus rhythm without an apparent cause or precipitating event. To make the diagnosis patients must have had previous episodes of AF which may have prompted investigations. The diagnosis is normally made by a 72 hour rhythm strip (R-test), or sometimes by echocardiogram. The AF can be asymptomatic, but some patients may complain of chest pain, shortness of breath, irregular palpitations and pre-syncope. These patients should be managed in the same way as patients with pre-existing AF.
Treatment options vary but the same basic rules discussed above apply. Those who have runs of AF with a rapid ventricular response (> 100 bpm) are frequently placed on rate control medication. The decision regarding anticoagulation is governed by the risk of thrombo-embolic disease according to risk stratification tools such as CHADS2. Paroxysmal atrial fibrillation needs to be optimised prior to surgery using the same principles as any patient with pre-existing AF. Given its paroxysmal nature, conversion to sinus rhythm shouldn’t be attempted unless echocardiography has excluded evidence of emboli to minimise the risk of a stroke. Other methods of treating PAF include Flecainide or, in a specific group of patients, radiofrequency ablation.
SUMMARY
- Identify whether the atrial fibrillation is pre-existing or new, and if so define the time of onset
- Precipitating factors should be identified and corrected
- DC Cardioversion should be considered for patients who are haemodynamically compromised
- A decision should be made whether rhythm or rate control is preferred
- Consider anticoagulation in patients at high risk of thromboembolism
- Arrange appropriate follow up
ANSWERS
- Atrial Fibrillation:
a. True
b. False: absence of P waves and an irregularly irregular rhythm
c. False: two or more risk factors indicate anticoagulation may be required
d. False
e. False: the a wave on the JVP corresponds to atrial contraction, which is absent in AF - Atrial Fibrillation treatment options:
a. False: β-blockers have to be used cautiously in those with asthma, peripheral vascular disease and diabetes.
b. False: digoxin, has a slower onset that other IV agents available, and so is used in the chronic setting, particularly those with heart failure
c. False: unstable patients should be treated by cardioversion
d. False: Nifedipine is a peripherally acting calcium channel antagonist and is used to prevent vasospasm or reduced systemic vascular resistance in hypertensives.
e. False: warfarin should be continued for a minimum of 4 weeks post elective DCCV because of the risk of reverting back into AF - The following precipitate AF
a. True
b. True
c. True
d. True
e. True
REFERENCES
- Camm AJ, Kirchof P, Lip GYH et al. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J 2010;31:2369-429
- NICE Guidelines: Preoperative tests for elective surgery: 2003
- NICE CG36 AF 2006 guidelines
- ALS guidelines for management of tacharrythmias. https://www.resus.org.uk/pages/periarst.pdf
- Lip G, Douketis J. Management of anticoagulation before and after elective surgery. UpToDate, 2014



