Paediatric Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Which of the following statements are correct?
a. Blood sparing techniques are surgical techniques aimed to reduce blood loss during an operation
b. Blood transfusions in children have a higher incidence of adverse events than adults
c. Non-infectious risks of blood transfusions account for 87-100% of all fatal incidents
d. The majority of paediatric adverse transfusion incidents are due to human error - Which of the following techniques are easily applicable to a paediatric patient?
a. Clean surgical technique
b. Perioperative autologous blood donation
c. Controlled hypotension
d. Hypervolaemic haemodilution - A transfusion algorithm/protocol
a. Should be written by the surgeons who are responsible for the blood loss
b. Should only be applicable to theatres where blood loss occurs
c. Should have restrictive transfusion triggers
d. Should include advice about optimizing anaemic patients before surgery - Which of the following statements about antifibrinolytics are true?
a. Aprotinin has been shown to have serious side effects in adults, but not in children
b. Tranexamic acid is as effective as aprotinin in reducing perioperative blood loss and transfusion
c. Antifibrinolytics have been shown to significantly reduce blood loss in major paediatric surgery
d. Antifibrinolytics are safe and should be given to every patient
Key Points
- Blood transfusions are associated with considerable risk which is why blood conservation techniques should be employed whenever possible
- Blood sparing strategies should be multidisciplinary and multimodal: a mix and match approach of pre-, intra- and postoperative techniques should be tailored to the individual patient.
- Communication between all specialities involved is key to a successful outcome
INTRODUCTION
Blood-sparing techniques are defined as all strategies and techniques aimed to reduce blood loss and reduce the need to transfuse blood and blood products. This is done in a multidisciplinary and multimodal approach.
The reasons for applying blood conservation strategies are:
- Risks and side effects of blood transfusion
- Infectious
- Non-infectious
- Low supply of blood products
- Few donors
- Short storage time for paediatric patients
- Refusal of transfusions by patients for religious or personal reasons
Blood transfusion
Blood transfusions can be beneficial and sometimes life-saving, but remain associated with significant risks and side effects.
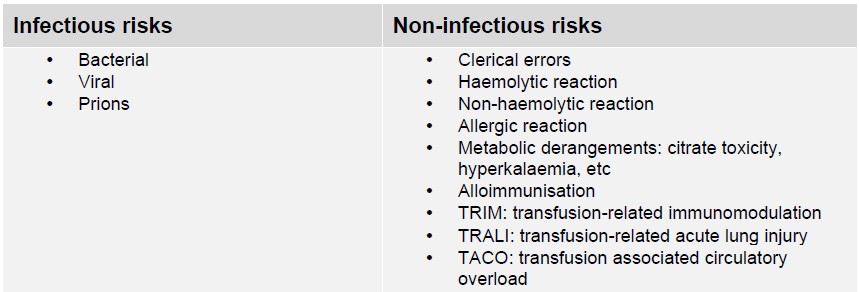
Table 1: Infectious and non-infectious risks of blood transfusions
Risks and side effects differ significantly between industrialised and developing countries. In developing countries, transfusion-transmissible infections dominate, in industrialised countries, immunological reactions, overtransfusion and clerical errors are the main problem.
The overall safety of blood components has increased in recent years, especially in industrialised countries, particularly in relation to infection risks, but there are still considerable non-infectious risks, particularly in the paediatric population. The incidence of adverse outcome is 37:100,000 RBC issued for infants, 18:100,000 for children <18yrs and 13:100,000 for adults (Serious Hazards of Transfusion SHOT data, UK).
Children not only have a higher incidence of adverse events, but a worse outcome as well. In view of these risks, blood transfusions should be kept to a minimum and only be given after careful consideration of the risks and benefits of transfusion.
PERIOPERATIVE
Blood conservation strategies begin long before the operation. Health professionals should be aware of the risks of transfusion and should avoid unnecessary and inappropriate transfusions as well as overtransfusion. Several strategies have been developed to decrease blood use, but they also carry risks and bear costs. Most of these methods are available for children, but their use and efficiency will depend on patient age and type of surgery.
These strategies can be classified into three areas:
- Increasing pre-operative red blood cell mass
- Decreasing peri-operative blood losses
- Optimizing transfusion practice
A combination of all three strategies is the best approach, depending on the risk assessment for the individual patient, and the type of surgery involved. The higher the expected blood loss, the greater the benefit from blood-conservation strategies.
Some techniques are applicable to all types of surgery (e.g. surgical attention to reducing blood loss by clean surgical technique, pressure dressings etc), while some are best applied in surgical specialities with large expected blood loss, such as cardiac, craniofacial or scoliosis surgery. The more advanced strategies, such as pre-operative autologous donation, may only be possible in specialist centres with appropriate logistical support.
Optimizing transfusion practice
Adoption of restrictive transfusion triggers and regular audit of transfusion policies and algorithms has been shown to reduce blood transfusion without increasing adverse events. However, the optimal transfusion threshold in children in terms of haemoglobin or haematocrit that is applicable to every situation is unclear. Acute blood loss in theatre is a dynamic process and several aspects need to be considered before a transfusion.
Adolescents and adults compensate for acute blood loss by increasing their cardiac output. Smaller children have a limited ability to increase cardiac contractility, therefore rely on increasing their heart rate to compensate for blood loss. Blood transfusion should be considered in infants <4-6 months when they have lost 20-25% of their blood volume, rather than 30-35% of blood volume, as in older children and adults.
The decision to transfuse should be based on the ongoing losses (speed and amount of blood loss and an estimate of the duration of surgery), pre-existing conditions, such as cardiac malformations or previous cardiac surgery, and signs of end-organ impairment, such as oliguria.
In case of rapid blood loss, frequent laboratory measurement of haemoglobin can be misleading, because by the time the results get reported, the haemoglobin might already have changed depending on the speed and amount of blood loss and the amount of replacement fluid. A quick and easy way of near patient testing are machines like Haemocue or blood gases (arterial or venous). Blood gases have the advantage of providing Glucose and lactate levels and electrolytes.
In anaemic patients without acute blood loss (e.g. postoperative patients), transfusion should be given to maintain tissue perfusion and oxygenation. Again, there are no absolute numbers in terms of haemoglobin or haematocrit. The age of the patient, pre-existing disease and clinical condition need to be taken into account.
Generally, the same transfusion threshold is used for normovolaemic infants over 4-6months of age as children, adolescents and adults. Studies have shown that a restrictive transfusion policy using a haemoglobin threshold of 70g/l is not associated with increased morbidity or mortality in postsurgical PICU patients over 4-6 months1.
For ventilated neonates with FiO2<0.35and premature babies, a transfusion threshold of haemoglobin 100g/l is recommended. For babies with cyanotic heart disease, congestive heart failure or children with a FiO2 requirement >0.35, the haemoglobin threshold might be higher, up to 120g/l.
A detailed transfusion policy should include advice about optimizing pre-operative conditions, such as anaemia, as well as advice about medications such as aspirin or other anticoagulants.
It should be applicable to an operating department environment and also at ward level: if the patient is transfused on the ward because of a low haematocrit but is otherwise asymptomatic, the efforts of the whole team will be undone. This emphasizes the importance of a standardized multidisciplinary transfusion algorithm, which is known and adhered to throughout the hospital. A comprehensive multimodality blood conservation programme adapted to the specific surgical requirements represents the best approach to reduce the use of allogeneic blood transfusions.
PREOPERATIVE
Patient factors
Patient risk factors for blood transfusion should be assessed at the preoperative assessment, particularly if the planned surgery is associated with potential blood loss. Patient factors to consider are:
- Starting haemoglobin/haematocrit, adjusted for age and weight
- Co-morbidities: cardiac or pulmonary conditions, diabetes, renal problems, gastroenterology problems, etc
- Pre-operative treatment with anticoagulants
- Clinical signs of anaemia or hypovolaemia
- Type of surgery. Operations associated with a high expected blood loss are craniofacial, scoliosis and cardiac surgery.
Any modifiable risk factors should be addressed prior to surgery: correct anaemia or hypovolaemia, stop anticoagulants if possible; discuss any problems that might have arisen with the appropriate specialist (cardiologist, haematologist, surgeon, etc).
Medication
Children may be taking drugs that interfere with coagulation, such as aspirin or warfarin, particularly children with cardiac abnormalities. Stopping anticoagulants might not be possible due to the underlying condition (e.g. cardiac conditions such as shunts or artificial valves), but all non-essential medication should be stopped in a timely fashion.
The decision to stop anticoagulants should be made by carefully weighing the risk of thrombo-embolism against the risk of increased intra- and postoperative bleeding. The clinical team responsible for the perioperative care of the patient, for example, cardiologist, haematologist, anaesthetist, surgeon must discuss the pros and cons of stopping anticoagulants.
The most common anti-platelet medication in children is aspirin. In most cases it doesn’t need to be stopped before minor surgery (e.g. for excision of skin lesion). Its benefits usually outweigh the perceived bleeding risks. If it can be stopped, it should be stopped 5-7 days before surgery. Platelet function tests are not necessary.
Newer anti-platelet drugs are either ADP receptor inhibitors (Clopidogrel, Ticlopidin) or Glycoprotein IIb/IIIa receptor inhibitors (Abciximab). They are usually prescribed alongside aspirin and a decision has to be made whether both can be continued, both stopped, aspirin alone continued or a replacement medication started (heparin). This decision should be made by the cardiologist, haematologist, surgeon and anaesthetist on a case-by-case basis. There aren’t enough studies in the paediatric patients to support a recommendation for all cases at the moment2.
The most common oral anticoagulant used in children is warfarin, a vitamin K antagonist. Newer oral anticoagulants either act by direct inhibition of thrombin (Dabigatran, Ximelagatran)or direct inhibition of Factor Xa (Rivaroxaban). These drugs have been shown in adults to be as effective as warfarin with a better safety profile. The use and recommendations for children have been extrapolated from adult studies, as there are few or no studies in the paediatric population2. If a patient is on a new anticoagulant, a paediatric haematologist should be consulted about the stopping or changing of the drugs perioperatively.
If anticoagulation needs to be continued, a detailed plan about the management of the medication needs to be made and communicated clearly to all members of the team, including the patient and the parents.
In patients receiving warfarin, anticoagulation usually needs to be continued, and so is changed perioperatively to an IV heparin infusion (unfractionated heparin), which has a shorter half-life. Patients can therefore be anticoagulated until shortly before the operation starts.
The following is a guideline describing how to change warfarin to heparin8:
- Day 3 before surgery:
- Take last dose of warfarin
- Day 2 before surgery:
- Omit warfarin
- Day before surgery:
- Give loading dose of 100U/kg of unfractionated heparin, followed by an infusion of heparin 25U/kg/hr.
- Check APTT 4hrs after start of heparin; it should be 60-90 sec.
- If the APTT is too low, increase the heparin infusion by 10% and re-check after 4 hrs.
- If desired APTT is reached, do daily APTT levels.
- Day of surgery:
- Check INR: for most surgery INR should be <2; if higher, consult with surgeon/ haematologist/cardiologist
- Stop heparin 4hrs before surgery. In some cases where there will be a long period of time preparing the child for surgery, for instance repeat cardiac operations, some surgeons are happy for the heparin to be stopped on arrival in the anaesthetic room, with skin incision approximately 1.5 hrs afterwards
- Re-start heparin 1-2 hrs after surgery, continue with heparin infusion as instructed by the surgeon
- If no other instruction: start usual warfarin dose 24hrs post-surgery
- Day 1 postop:
- Continue heparin infusion, give regular warfarin
- Day 2 postop:
- Check INR: if at normal pre-op level, stop heparin. If INR too low, continue with heparin infusion and re-check INR daily. Stop heparin once INR is at desired level.
Anaemia
Anaemia should be investigated and treated, depending on the aetiology, although this may delay the operation.
Chronic childhood anaemia in industrialised countries is usually associated with diet, or pathologies such as cancer or the effects of chemotherapy; in developing countries malnutrition, parasite infestation or infections (malaria) dominate. The same transfusion thresholds as in acute anaemia apply.
Iron and erythropoietin can be used in anaemic children to increase red cell mass before an operation, although studies show either no change or decrease in allogeneic blood transfusions. In order to be successful, children need to be able to generate a good bone marrow response. Oral iron and erythropoietin supplementation over 2 weeks in adults with a normal haemoglobin resulted in an increase in the haemoglobin by 10g/l.
Intravenous iron increased the haemoglobin by up to 60g/l in a study in adults with anaemia due to chronic bleeding who refused transfusion.
The use of iron and erythropoietin, as well as erythropoietin alone, has been used to increase pre-operative haematocrit, and to increase the units of blood collected for pre-operative autologous blood donation.
Pre-operative autologous blood donation
Pre-operative autologous blood donation programs have been used to augment blood supply and alleviate the risks of allogeneic blood transfusions, particularly the risk of alloimmunisation. Children receiving autologous donations tolerate a greater intraoperative allowable blood loss if the haematocrit has returned to baseline or higher before surgery.
The administrative structure for this is complex and involves multiple specialities. Pre-operative autologous donation eliminates the risks of allogeneic blood transfusion, but there are additional risks associated with clerical errors or infectious contamination.
Although described in children as young as 6 months, most pre-operative donation programs are for children >20kg1. These programs vary widely with variable inclusion criteria. Smaller children need a general anaesthetic for the donation and need to be able to generate an adequate bone marrow response to increase their haemoglobin/haematocrit. As mentioned before, erythropoietin alone and in combination with iron has been shown to increase red cell mass prior to surgery.
Surgery must be synchronised with the donations in view of the short storage time for paediatric patients. Waiting lists and cancelled surgery due to patient factors (common cold, chest infections) are common in paediatric anaesthesia, which can cause wastage percentages of autologous blood up to 64%, making this an inefficient system to reduce perioperative blood transfusion in many cases.
INTRAOPERATIVE
Surgical technique
One of the most important techniques to reduce blood loss is an active surgical approach to bleeding.
Prevention of ongoing blood loss is key. There are multiple ways to achieve this:
- Use of vasoconstrictors: Adrenaline-soaked swabs or local anaesthetics with adrenaline cause vasoconstriction and therefore less blood loss
- Tourniquet
- Clean surgical technique: diathermy, ligation, fibrin patches, ultrasound, etc
- Pressure dressings
Postoperative bleeding should be addressed in the same way: early exploration and second look operations decrease the amount of blood lost. Delayed re-exploration is associated with higher risk of complications and transfusion.
Acute normovolaemic haemodilution
Acute normovolaemic haemodilution (ANH) involves taking blood from the patient after induction of anaesthesia and replacing it with crystalloid or colloid to maintain normovolaemia. Reducing the haematocrit in this way decreases viscosity and improves tissue perfusion by decreasing peripheral vascular resistance, causing an increase in blood flow. Blood lost intraoperatively has a lower haematocrit, so less red cell mass will be lost. After blood loss has stopped, the collected blood is transfused back to the patient.
This method is recommended for patients with multiple antibodies for whom it is often difficult to get allogenic blood crossmatched. Since the blood is kept at room temperature, there are no side effects of cold storage, such as decreased platelet function or high potassium content. The efficiency of this technique depends on the amount of blood collected and the blood volume lost by the patient.
ANH might not be applicable to very young patients as infants are less able to compensate for anaemia by increasing stroke volume compared to adults. In babies younger than 6 months ANH is not recommended as the dilutional anaemia is poorly tolerated due to the presence of HbF, which has reduced ability to unload oxygen to the tissues. ANH is used to good effect in adolescents for scoliosis surgery and children undergoing craniosynostosis surgery. However, a recent meta-analysis of 42 RCT suggested only modest benefits from ANH1.
Hypervolaemic haemodilution
Hypervolaemic haemodilution (HH) is dilution of blood volume without withdrawing blood first. It is usually achieved with colloid (approx. 10-15ml/kg) to reach a pre-determined haematocrit level, usually 25%. Care should be taken to prevent circulatory overload and circulatory failure due to cardiac inability to deal with the increase in volume.
Studies in adults and children found HH efficient in reducing the need for allogeneic blood transfusion and studies comparing ANH and HH found almost no difference in postoperative haematocrit.
Controlled hypotension
Blood pressure is lowered to a pre-defined mean arterial pressure, usually 50-60mmHg. Various drugs have been described to achieve this: in paediatric anaesthesia deepening of the anaesthetic with anaesthetic vapours or TIVA is usually sufficient. In PICU, beta blockers, nitroprusside, nitroglycerin, nicardipine have been used to good effect. This is an inexpensive but effective method to decrease intraoperative blood loss.
The limitations of this technique are hypovolaemia, elevated ICP and decreased end-organ blood flow. Hypocapnia should be avoided and a higher FiO2used to assist oxygen delivery.
Cell salvage and autotransfusion
Cell salvage systems can be applied to infants and children and are widely used, especially in surgery with major blood loss, such as cardiac, craniofacial and scoliosis surgery. Intraoperative blood salvage was found to significantly decrease the necessity for transfusions and transfusion volumes in several studies. Small centrifugation bowls have been introduced, so that the technique is now safe and efficient method to use, even in infants <10kg and /or <1 yr of age. The system can be run continuously in operations with major ongoing blood loss.
Cell salvage can be done postoperatively from drains as well, but special care has to be taken with aseptic collection and processing of salvaged blood.
Antifibrinolytics
The use of antifibrinolytics remains a controversial issue. In the references at the end of this article I’ve included two major studies: the 2008 Cochrane Review of antifibrinolytic agents3 and a similar 2009 metaanalysis about the effect of aprotinin, tranexamic acid and aminocaproic acid in the paediatric population4.
Aprotinin, a serine protease inhibitor, was withdrawn from the market in 2007, following the publication of several studies revealing serious side effects in adults. The most widely known study is the BART trial (Blood conservation using Antifibrinolytics in a Randomized Trial), looking at adult high-risk patients undergoing complex cardiac surgery. It showed an increase in 30-day mortality, myocardial infarction, right heart failure and cardiogenic shock. Interestingly, these results could not be confirmed in the paediatric population: a recent study looking at aprotinin for congenital cardiac surgery in over 12,000 paediatric patients has found no increase in mortality nor rate of dialysis5.
Studies in paediatric patients have been looking at major surgery with significant blood loss, such as cardiac or scoliosis surgery: several studies compared different antifibrinolytics and found no major differences in terms of side effects or efficiency in reducing perioperative blood loss and transfusions1. There were no additional side effects of aprotinin when compared to tranexamic acid. A meta-analysis in major paediatric surgery found that tranexamic acid (TEA) was as effective as aprotinin in reducing perioperative blood loss and transfusion6.
A 2008 Cochrane Review of studies in scoliosis surgery looking at aprotinin, tranexamic acid and aminocaproic acid showed that all three antifibrinolytics decreased the amount of blood transfusion3. The authors recommended a high dose regimen for all 3 drugs, as these were the regimens used in the studies included in the review and found no adverse effects. Another study looked at tranexamic acid vs placebo in scoliosis surgery: it showed that estimated blood loss (EBL) was reduced in the TEA group compared with placebo, although the amount of transfused blood did not differ7.
POSTOPERATIVE
Postoperative blood losses after major surgery such as cardiac, major orthopaedic surgery or craniofacial reconstruction can be significant. Pressure dressings and drains can be deceiving as bleeding can occur obliquely into the body, into the thoracic or abdominal cavity or soft tissue. Drains can be blocked by blood clots. Constant vigilance is necessary and suspected blood loss should elicit a prompt response. Early decision for surgical second look in case of ongoing or abnormal bleeding avoids unnecessary transfusions. Patients with re-exploration are at higher risk of complications if the time to re-exploration is prolonged.
Blood salvage via cell saver systems can be done postoperatively from drain losses. As mentioned above, meticulous asepsis must be maintained for collection and re-transfusion.
Abnormal clotting should be corrected with medication such as vitamin K, calcium or antifibrinolytics. Postoperative anaemia can be augmented with iron and erythropoietin.
FUTURE ALTERNATIVES?
Alternatives to blood transfusion can be grouped into five categories:
- Cell-free, haemoglobin-based oxygen carriers
- Artificial red cells based on liposomes or encapsuled in nanoparticles
- Fluorocarbon-based solutions to dissolve oxygen
- Universal red cell (modified human red cells)
- Red cells derived from stem cells
Although all of these alternatives have been and are being investigated, at present only stem cell-derived erythropoesis looks promising. Ex-vivo production of red cells from stem cells is currently under research. The difficulties lie in expanding the laboratory conditions to a larger scale for mass production and potential adverse effects.
ANSWERS TO QUESTIONS
- a. False: blood-sparing techniques are not just surgical. It is a multidisciplinary approach.
b. True
c. True
d. True - a. True
b. False: PAD is a complicated procedure, involving multiple specialities and requiring meticulous timing, which is difficult to achieve with paediatric patients.
c. True
d. True - a. False: A transfusion protocol is a multidisciplinary policy and should have input from multiple. Specialities, such as anaesthetics, haematology, ward doctors and nurses, etc
b. False: A good transfusion policy includes advice about pre- and postoperative care of patients with low haemoglobin.
c. True
d. True - a. True
b. True
c. True
d. False: Like all other drugs, antifibrinolytics should only be given after careful risk/ benefit consideration
WEBLINKS
REFERENCES and FURTHER READING
- Lavoie J. Blood transfusion risks and alternative strategies in pediatric patients. Pediatric Anesthesia 2011; 21:14-24
- Dabbous MK, Sakr FR, Malaeb DN. Anticoagulant therapy in pediatrics. J Basic Clin Pharma 2014;5: 27-33. Available from: http://www.jbclinpharm.org/text.asp?2014/5/2/27/134947
- Tzortzopoulou A, Cepeda MS, Schumann R et al. Antifibrinolytic agents for reducing blood loss in scoliosis surgery in children. Cochrane Database Syst Rev 2008; 3: 3CD006883
- Schouten ES, van de Pol AC, Schouten ANJ et al. The effect of aprotinin, tranexamicacid and aminocaproic acid on blood loss and use of blood products in major pediatric surgery: a meta-analysis. Pediatr Crit Care Med 2009; 10: 182–190.
- Pasquali SK, Hall M, Li JS et al. Safety of aprotinin in congenital heart operations: results from a large multicenter database. Ann Thorac Surg 2010; 90: 14–21.
- Schouten ES, van de Pol AC, Schouten ANJ et al. The effect of aprotinin, tranexamic acid, and aminocaproic acid on blood loss and use of blood products in major pediatric surgery: a meta-analysis. Pediatr Crit Care Med 2009; 10: 182–190.
- Sethna NF, Zurakowski D, Brustowicz RM et al. Tranexamic acid reduces intraoperative blood loss in paediatric patients undergoing scoliosis surgery. Anesthesiology 2005; 102: 727–732.
- Rees P, Grech V: Perioperative anticoagulation for children with prosthetic mechanical valves. Images Paediatr Cardiol. 2000 Apr-Jun; 2(2): 41–42



