Intensive Care Medicine
INTRODUCTION
When thinking about fluid within the body we are essentially thinking about water. Tight regulation of the balance between water intake and output, and its distribution, is vital to the optimal function of every organ system within the body. In a wide variety of illnesses and during surgery, disturbances to this fine balance occur which must be identified and corrected to prevent deterioration, complications and to promote recovery.
Care of unwell patients with body water abnormalities and patients undergoing surgery, are encountered daily by medical practitioners. Therefore it is important to have a good understanding of the physiology of normal fluid homeostasis and what happens when these mechanisms fail or are overcome.
QUESTIONS
These are for you to test your current knowledge and understanding of body fluid physiology prior to reading this tutorial. The answers can all be found in the following text.
- What is the volume of extracellular fluid in a healthy 75 Kg male? Where/how is this fluid distributed?
- What is the normal osmolality of plasma? How can you estimate plasma osmolality?
- What do you understand by tonicity?
- What are the main sources of fluid loss from the body? What volume is lost via these routes in each 24 hour period?
- If a normally hydrated 75 Kg male patient is kept ‘Nil By Mouth’ for 24 hours, what volume of water would have to be given by alternative routes to maintain hydration, and what electrolytes should the water also contain to replace ongoing losses?
HOW MUCH FLUID IS THERE IN THE BODY, AND HOW IS IT DISTRIBUTED?
The amount of water in the body varies with a patient’s age, weight, and sex (Table 1). Total body water (TBW) accounts for 60% of an adult male’s total body weight; a normally hydrated 75 kilogram man will consist of approximately 45 litres of water. To simplify matters, all subsequent calculations will be based on this ‘ideal’ person with a total body water of 45 litres.

Table 1. TBW variation with age
Total body water is distributed throughout the body, and can be thought of as being in different ‘compartments’ or ‘spaces’. The compartments are separated from each other by membranes that regulate flow of water between each compartment and thereby control the amount of water which can exist in each of the compartments.
The largest fluid ‘compartment’ which accounts for two-thirds (30L) of TBW is within the cells of the body and is called intracellular fluid (ICF). The remaining one-third (15L) of TBW is outside the cells or “extracellular fluid” (ECF). The extracellular fluid is divided into several other compartments (Figure 1);
- 10L (approximately 2/3) exists in the spaces between cells and is termed interstitial fluid (ISF)
- 3.5L exists as blood plasma in blood vessels and is termed intravascular fluid
- The final 1.5L comprises the transcellular fluid which is made up of intraocular fluid, cerebrospinal fluid (CSF), urine in the bladder, and fluid within the lumen of the bowel
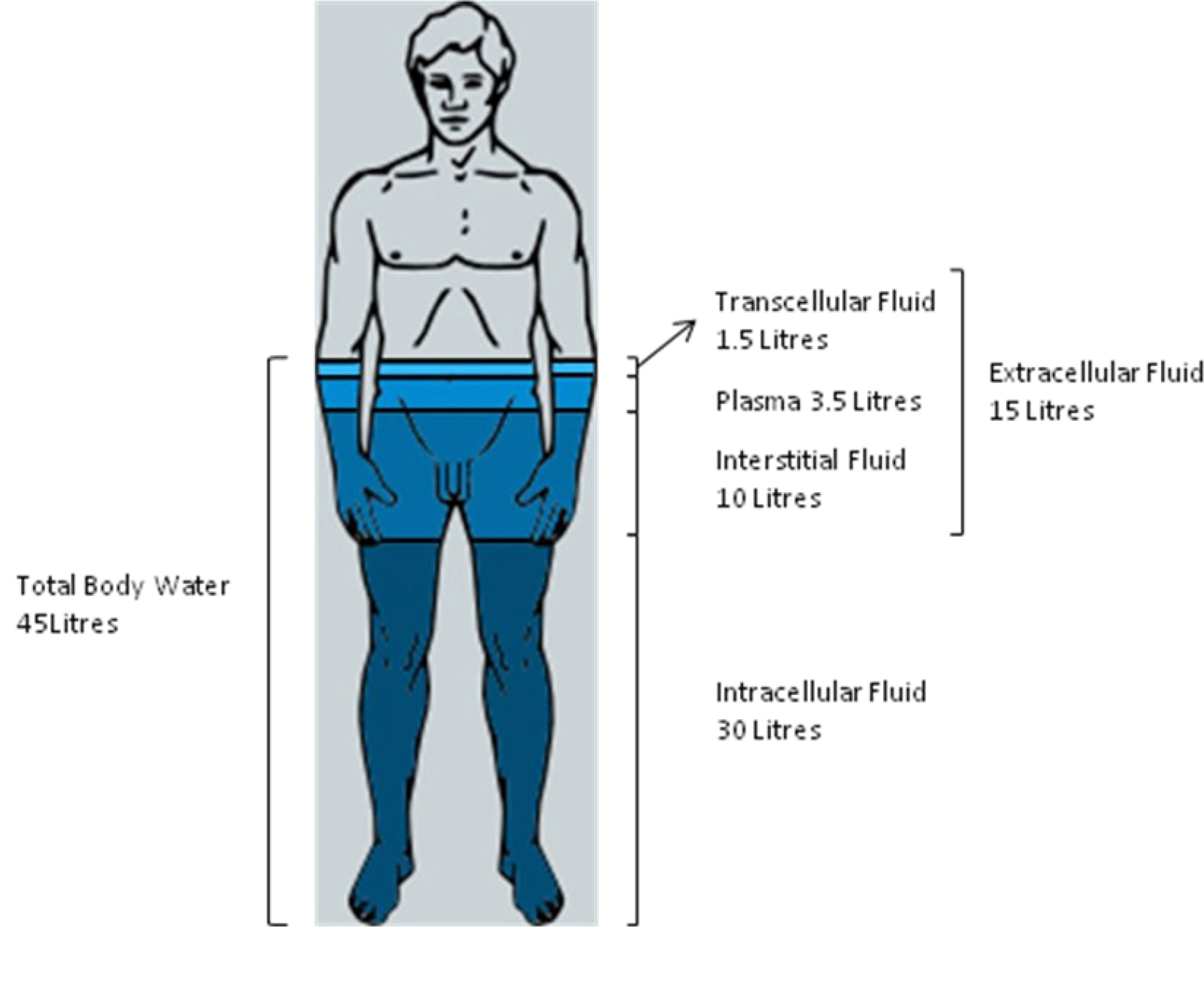
Figure 1. Relations of the body fluid compartments.
ESSENTIAL PRINCIPLES
In order to fully understand the distribution and movement of water within the body there are some definitions, principles, and concepts that must first be understood:
Solvents, solutes, and solutions
- A solvent is a liquid, solid, or gas into which another solid, liquid, or gaseous substance can dissolve (the solute). This results in a solution which is in the same physical state as the solvent. A good example would be dissolving salt in water; the result still looks and behaves like water!
- To form a solution the solvent and solute undergo a reaction with breaking and forming of physical bonds. In this way the solute becomes completely incorporated with the solvent.
- A mixture is where one substance is added to another but no physical or chemical reaction takes place and the two remain separate.
- It is possible to have a fluid which is both a solution and also a mixture. To illustrate this consider the sea and the beach. The sea water is a solution as it is water (solvent) with salt (solute) dissolved in it. The two have undergone a reaction and combined in one new substance. At the shore the sea water disturbs the sand and some of the sand becomes mixed with the water. However the water and the sand are not chemically combined and clearly exist as separate substances, i.e. a mixture. Left undisturbed the sand would easily separate from the water and sink to the bottom, whereas the salt remains dissolved.
- Fluid within the human body is complex as it is both a solution as well as a mixture at the same time. The solvent is water, the solutes are numerous and include electrolytes e.g. sodium (Na+), potassium (K+), and chloride (Cl–) ions, solute molecules e.g. sugar and urea, and also gases e.g. oxygen (O2) and carbon dioxide (CO2). Intracellular and extracellular fluid contains proteins, while the intravascular fluid contains proteins, fats, and blood cells making these fluids mixtures as well as solutions.
Osmolarity and Osmolality
- These are ways of quantifying how much of a solute is dissolved in a solution i.e. the solute concentration of a solution.
- Osmolarity is the number of osmoles of solute particles per unit volume of solution and has units osmoles/litre. This is a very high concentration and in the body we use the milliosmole (i.e. one thousandth of an osmole).
- Osmolality is the number of osmoles of solute particles per unit weight of solvent and has units osmoles/kilogram. Again in the body we use milliosmoles.
- The two terms refer to similar concepts, the main difference being when temperature changes; volumes will change, but mass remains the same. Under most physiological conditions temperature is fairly constant and the two are very similar however, osmolality is the preferred term.
- One mole of any substance contains a constant number of molecules (or atoms) equivalent to Avagadro’s number, but not necessarily the same mass. It is the number of atoms or molecules, rather than overall mass, which is important when considering solutes, and hence measurement in moles allows better comparisons to be made. The unit osmoles is used to quantify the number of solute particles in a solution. It is related to the number of moles of solute and how much the solute dissociates in solution. This can be illustrated by considering compounds which do and do not dissociate:
- Ionic compounds dissociate e.g. sodium chloride (NaCl) dissociates into Na+ ions and Cl– ions which can be considered as separate solute particles, each of which influences the osmolality. Hence when 1 mole of NaCl is added to 1 Kg of water 2 osmoles of solute particles are formed and a solution with a concentration of 2 osmol/kg H2O is produced.
- Non-ionic compounds e.g. glucose do not dissociate and so 1 mole of glucose added to 1 Kg of water will produce 1 osmole of solute particles and a solution with concentration 1 osmol/kg H2O.
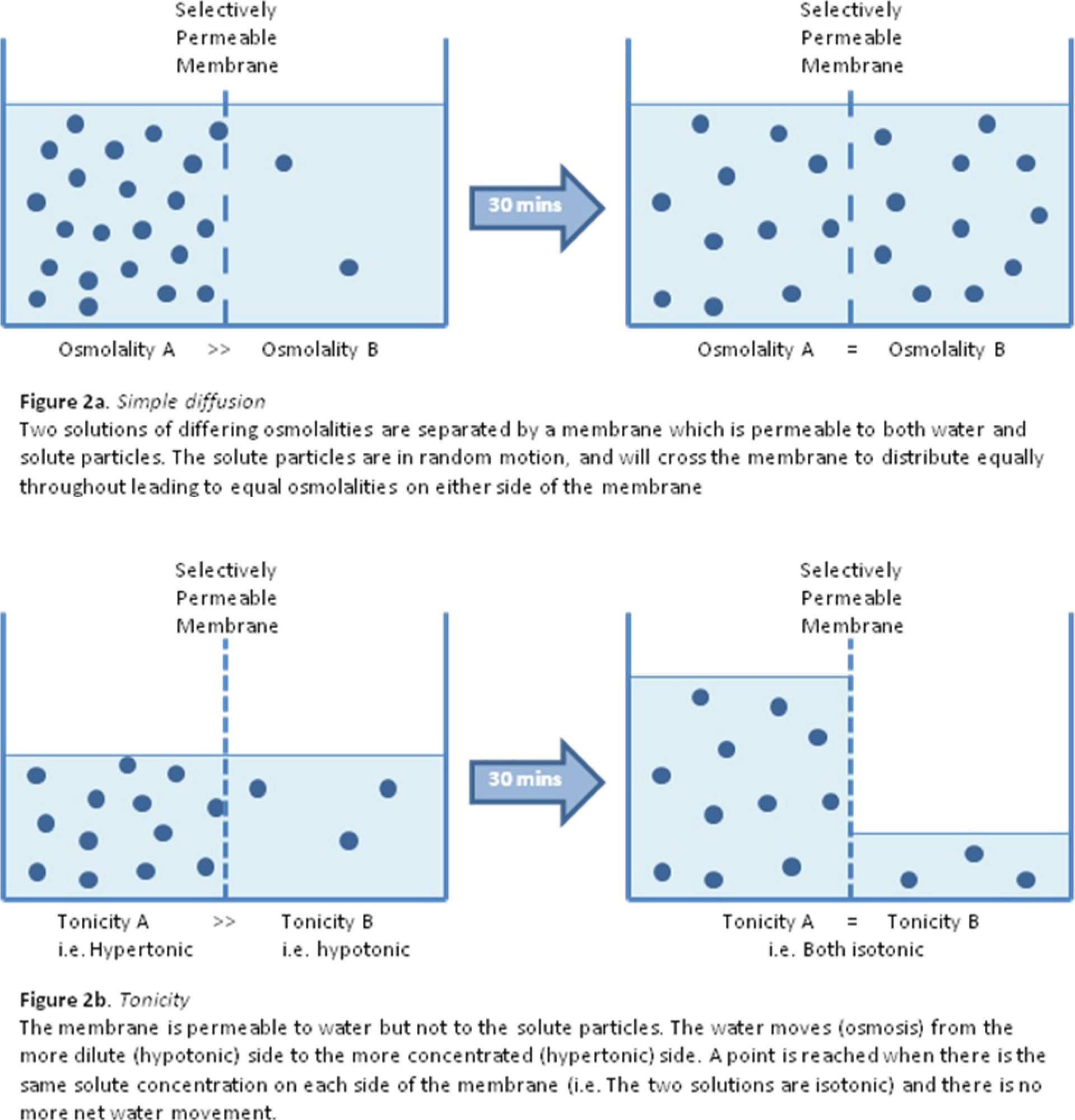
- Solute particles in solution have energy which means they have random motion, and will tend to distribute (diffuse) evenly throughout any solution if there is nothing to prevent this. (see figure 2a)
- It is possible to estimate the osmolality of plasma by summing the major plasma solute particles i.e. sodium ions, potassium ions, chloride ions, glucose, and urea. The serum sodium and potassium ion concentrations are doubled to account for the dissociation of sodium chloride and potassium chloride that occurs in solution:
- Plasma osmolality
- = 2 (Na + K) + glucose + urea
- = 2 (137 + 4.0) + 5.0 + 4
- = 291 mosmol/kg H20
- Plasma osmolality
Tonicity
- This is similar to, but not the same as, osmolality.
- Tonicity is a way of describing the relative solute concentrations of two solutions which are separated by a selectively-permeable membrane (often called a semi-permeable membrane). For example intracellular and extra-cellular fluid separated by the cell membrane.
- Whereas osmolality is determined by the total number of solute particles within a solution, tonicity is only influenced by those solute particles which are not able to cross the membrane separating two solutions.
- From this it follows that there are two key determinants of tonicity – the solute particles and the properties of the membrane involved. Strictly speaking tonicity should always be described with reference to a particular membrane e.g. the cell membrane.
- The tonicity of two solutions separated by a membrane can be described in relative terms to each other as:
- Hypertonic (contains a higher concentration of solute on one side of the membrane)
- Isotonic (contains the same concentration of solute on both sides of the membrane)
- Hypotonic (contains a lower concentration of solute on one side of the membrane)
- Frequently, in clinical practice the tonicity of fluid administered intravenously is described relative to the tonicity of the internal environment of the red blood cell and with reference to the red blood cell membrane. This is because administration of hypotonic or hypertonic solutions intravenously will lead to water movement into or out of the red blood cell causing damage to the cells. (see figure 2b)
Osmosis
- This is the passive movement of water from a dilute solution (i.e. relatively hypotonic) to a more concentrated solution (i.e. relatively hypertonic) across a selectively permeable membrane. (see figure 2b)
- The membrane is described as selectively permeable as it allows free passage of water molecules from one side to the other but does not allow any solute particles to cross.
- The fact it is a passive process means it is not dependent on energy produced from aerobic or anaerobic metabolism.
HOW DOES WATER MOVE BETWEEN FLUID COMPARTMENTS?
The intravascular, interstitial, and intracellular fluid compartments are separated from each other by selectivelypermeable membranes. The capillary wall separates the intravascular and interstitial fluid while the cell membrane separates the intracellular and extracellular fluids. These two membranes have different properties.
- Capillary wall – Movement of water and solutes across this membrane is largely passive as a result of either filtration or simple diffusion.
- Filtration results from the balance of Starling forces acting to drive water out of the capillary into the interstitium and in the process taking dissolved solutes with it.
- Diffusion is simply the movement of solutes from an area of high concentration to an area of lower concentration.
- The water crosses the capillary wall via the gaps between cells or directly through the cell membrane.
- Cell membrane – most solute and water movement across this membrane is via trans-membrane proteins or ion channels which can act to provide different permeability to different solutes.
- In a steady state the extracellular and intracellular fluids are isosmotic.
- For water to move between them there must be a difference in the tonicity between the two sides of the cell membrane.
- Any difference in extracellular or intracellular tonicity is due a change in the solute concentration in either fluid compartment. Water then moves by osmosis from the area of relative hypotonicity to the area of hypertonicity.
- The cell membrane is effectively impermeable to Na+ ions, and so it is the extracellular fluid Na+ concentration that effectively governs the distribution of water between ECF and ICF
- Extracellular fluid Na+ concentration is controlled by the kidneys under various neuroendocrine controls.
- Any net movement of fluid between two compartments can be in either direction depending on the balance of forces acting.
WHAT IS THE ‘NORMAL’ DAILY INTAKE AND OUTPUT OF FLUID AND ELECTROLYTES?
To ensure optimal function of the body the overall amount of body water must remain at the correct volume (as determined by a person’s age, weight, sex etc.). Just as vital, is that it is appropriately distributed throughout the body to ensure the correct volume of water is contained within all the fluid compartments at all times. This process whereby the body tries to ensure the correct fluid balance is maintained is called homeostasis.
In health, as part of the body’s normal function, water (and electrolytes) are continually being lost. These need to be replaced to maintain equilibrium (Table 2). There are times when these losses are dramatically increased e.g. diarrhoea, burns, or when fluid can be incorrectly distributed resulting in too much or too little in some of the body’s fluid compartments e.g. post-operatively, sepsis.
When the body is not under any stressors, the daily water output for a 75 kilogram man will average 2.5 litres and so an input of 2.5 litres is required. Some of the fluid loss is ‘sensible’ meaning that it is easily seen and measured e.g. urine output and loses from the gastrointestinal tract, while other fluid loses are ‘insensible’ meaning they are not seen and not easy to quantify e.g. sweat, and water vapour in exhaled gases.
These fluids all contain varying amounts of electrolytes. In order to decide on appropriate fluid replacement it is important to appreciate what quantity of electrolytes is lost as part of this fluid, on average, in a 24 hour period.
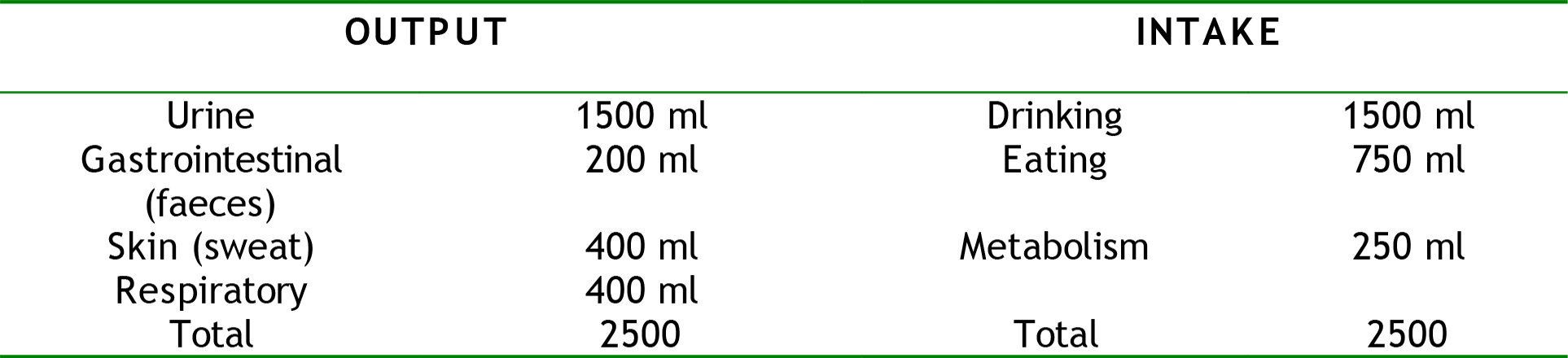
Table 2. Normal average adult water intake and output in a 24hr period
Sweat
The rate of sweating is controlled by the hypothalamus in response to changes in skin and core body temperature. It can be an important source of fluid loss as the rate of sweating can vary between 100 and 8000mls/day. The actual electrolyte content of sweat varies depending on emotional state, diet, exercise, acclimatisation, and various hereditary factors such as cystic fibrosis. The main electrolyte lost in sweat is sodium and can vary from 5mmol/day up to 350mmol/day.
Urine
Just as for any solution, the urine concentration is described in terms of its osmolality. There is no one ‘normal’ value for urine volume, composition, or concentration. The volume and composition of urine produced by the kidneys can vary hugely depending on the volume and composition of fluid in the IVF compartment as well as quality of kidney function. IVF fluid composition is itself a result of numerous factors e.g. recent fluid intake (drinks and food), body and environmental temperature, excessive fluid losses from the body.
For a normally hydrated patient with normal kidney function there is an obligatory volume of urine produced and electrolyte loss in a 24 hour period. The obligatory loss is a result of the daily solute intake and waste products of the body’s metabolism e.g. urea, creatinine and drug metabolites (which will tend to increase ECF tonicity), and need to be excreted, and the maximum concentration of urine that the kidneys can produce. Put another way – what is the smallest volume of urine the kidney needs to produce to excrete the excess solutes within the body? The urine concentrating ability of the kidney changes with age; children and the elderly cannot produce urine of as high concentration, and hence will lose more water to excrete a given solute load.
For healthy patients with normal renal function urine osmolality can range from 50-100 mosmol/kg H2O up to around 1200mosmol/kg H2O
In a 24 hour period, the urine excretion of these products is as follows:
- Sodium 50-200 mmol
- Potassium 20-60 mmol
- Urea 330-500 mmol
- Creatinine 9-16 mmol
- Protein <0.1g
Alterations in the intravascular fluid volume and osmolality affects the kidney’s glomerular filtration rate (GFR) and hence the amount of sodium chloride in the filtrate reaching the distal tubule of the kidney. This is detected by an area called the ‘juxtaglomerular apparatus’ which is responsible for producing and secreting the hormone renin which controls the renin-angiotensin-aldosterone system (RAAS).
At times of hypovolaemia (low intravascular fluid volume) or high osmolality (dehydration) more sodium chloride is delivered to the juxtaglomerular apparatus in the filtrate stimulating an increase in renin production thereby activating the RAAS. One of the actions of the RAAS is to increase sodium and water reabsorption in the proximal tubule (by the action of angiotensin 2) and the in the collecting ducts (by the action of aldosterone and anti-diuretic hormone (ADH)). This will tend to return IVF fluid volume and osmolality towards normal.
Gastrointestinal Tract
During a 24-hour period large volumes of fluid and electrolytes are secreted into and reabsorbed from the gut (Table 3). In a normally functioning gut around 9000ml of fluid enters the gut lumen as a result of oral intake, saliva, gastric juices, and small intestine secretions. The vast majority (8500ml) is reabsorbed in the jejunum and ileum, and as a result only 500ml enters the colon, of which 250ml is reabsorbed. Due to this recycling mechanism, the volume of fluid contained within the lumen of the gut at any one time is actually only a very small volume, and is considered part of the transcellular fluid.
If the recycling mechanism of the gut tract is disturbed e.g. persistent vomiting, small bowel obstruction, post operative ileus, the large volumes of fluid and the electrolytes secreted proximally will not be reabsorbed more distally. This results in excess loss of extracellular fluid along with large amounts of electrolytes.
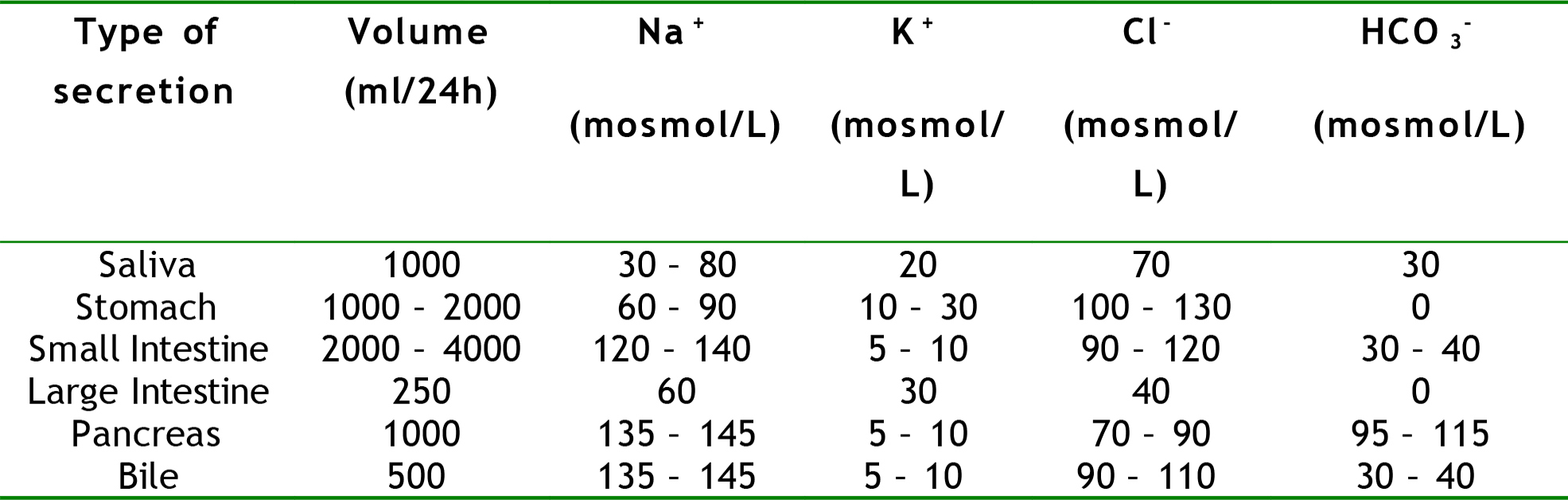
Table 3. Composition and daily volume produced of the various different gastrointestinal secretions
Respiratory Tract
Exhaled gases contain water vapour, but not solutes. Typically 400ml of pure water is lost this way in a 24-hour period. This loss is increased with increasing minute ventilation, or by breathing dry gases e.g. non-humidified oxygen. These losses are usually not clinically significant in isolation but it is good practice to minimise them where possible e.g. by using warmed humidified oxygen.
Evaporative Losses
In addition to sweating, about 400ml of fluid is also lost from the skin by evaporation. In contrast to sweat this evaporative loss is pure water, with no electrolyte content and ordinarily is of little clinical significance.
However, there are circumstances which give rise to a significant increase in evaporative losses for other reasons. An example of this is during surgery on open body cavities e.g. the chest or abdomen, where visceral surfaces and membranes are exposed to the air and subject to heat from the theatre lights, sometimes for several hours. In these circumstances it is important to anticipate this loss and plan for adequate replacement.
Total daily electrolyte loss
In a normal healthy person it is possible to combine all of the losses described above in order to estimate the daily electrolyte loss and hence the amount that needs replacing (Table 4).

Table 4. The average total daily adult electrolyte loss, and hence requirement
SUMMARY
It is vital to maintain the optimal function of every individual cell and organ, as well as the body as a whole, that the right volume of fluid with the right composition is contained within the right compartment. It is important to have a good knowledge and understanding of the composition and volume of fluid in the various compartments, and the body’s regulatory mechanisms, in order to appreciate patients’ fluid shifts, losses, and replacement requirements in acute illness and the peri-operative period.



