General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Bone Cement Implantation Syndrome (BCIS) has the following features:
a. Reduced arterial oxygenation.
b. A combination of: hypoxia, hypotension, arrhythmias, and loss of consciousness.
c. Grade 3 (severe) BCIS is defined as cardiovascular collapse requiring CPR.
d. Only occurs in cemented hemiarthroplasties.
e. Only occurs prior to limb tourniquet deflation. - The following patient characteristics are associated with an increased risk of developing BCIS:
a. Increasing age
b. Male sex
c. Previous orthopaedic surgery or instrumentation
d. Diuretic therapy
e. Beta-blocker therapy - Key steps for managing patients at risk of Bone Cement Implantation Syndrome include:
a. Communicate with surgeons, and consider using non-cemented procedures in high-risk patients.
b. Increase inspired oxygen concentration at the time of cementation to help resuscitation if needed.
c. Monitor for BCIS only after the cement is applied.
d. Use nitrous oxide to reduce the risk of air embolism.
e. Ensure the surgeons inject cement in an antegrade fashion.
Key Points
- Bone Cement Implantation Syndrome (BCIS) likely involves pressurised bone cement to cause pulmonary emboli.
- 20% of cemented hemiarthroplasties have BCIS, with around 1% of these resulting in cardiovascular collapse requiring CPR.
- Managing BCIS requires a teambased approach, with a focus on preparation, vigilance, and prompt treatment.
INTRODUCTION
Bone Cement Implantation Syndrome (BCIS) is a potentially fatal complication of orthopaedic surgery, involving pressurised bone cement. BCIS is characterised by hypoxia and/or hypotension (with potential loss of consciousness), occurring around the time of bone cementation.1 The incidence of BCIS in relevant orthopaedic procedures is approximately 20%, and the incidence of a severe reaction resulting in cardiovascular collapse within this group is 0.5-1.7%.2 The ability to predict, recognise, and manage BCIS in the perioperative setting is important for every anaesthesia provider.
CLINICAL FEATURES AND CLASSIFICATION
Physiologically, Bone Cement Implantation Syndrome results in reduced arterial oxygenation, characterised by a combination of clinical features (Figure 1).
The cardiovascular changes present can be variable, but the most common are: a reduction in mean arterial pressure, stroke volume, cardiac output, as well as an increase in pulmonary vascular resistance, resulting in reduced rightventricular ejection fraction.3
BCIS has a wide spectrum of severity. A majority of affected patients develop non-fulminant BCIS. Non-fulminant BCIS is characterised by a significant, yet transient, reduction in arterial oxygen saturation and systolic blood pressure (SBP) in the peri-cementation period. A small proportion of patients will develop fulminant BCIS, with profound intraoperative cardiovascular changes, progressing to; arrhythmias, shock, or cardiac arrest.3 A 3-stage classification of BCIS severity was proposed by Donaldson et al. in 2009 (Figure 2).3

Figure 1. BCIS clinical signs and symptoms
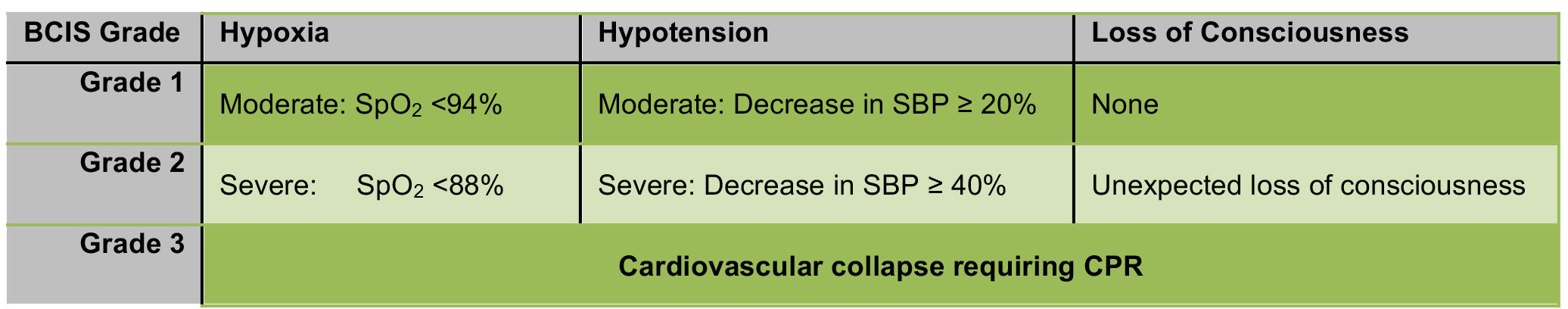
Figure 2. Classification of BCIS severity based on the degree of hypoxia, hypotension and conscious level, as per Donaldson et al.
The incidence of BCIS is highest in cemented hemiarthroplasty, but has also been observed in other cemented procedures, such as total hip replacement or knee replacement surgery.1 BCIS typically occurs at the time of bone cementation and prosthesis insertion, but may also occur during femoral reaming (before cementation), or during joint reduction and limb tourniquet deflation (after cementation).3
INCIDENCE
A large retrospective study by Olsen et al. in 2014 examined BCIS in 1080 patients undergoing cemented hemiarthroplasties after proximal femoral fractures. Using the BCIS classification system in Figure 2, a study was conducted to estimate the incidence and risk factors for BCIS, as well as its impact on mortality at 30 days and 1 year1,3. Another study published in 2014, the Anaesthesia Sprint Audit of Practice (ASAP), looked at the incidence of BCIS in over 3500 cases of cemented hemiarthroplasties.4 Their findings are summarised below (Figure 3).
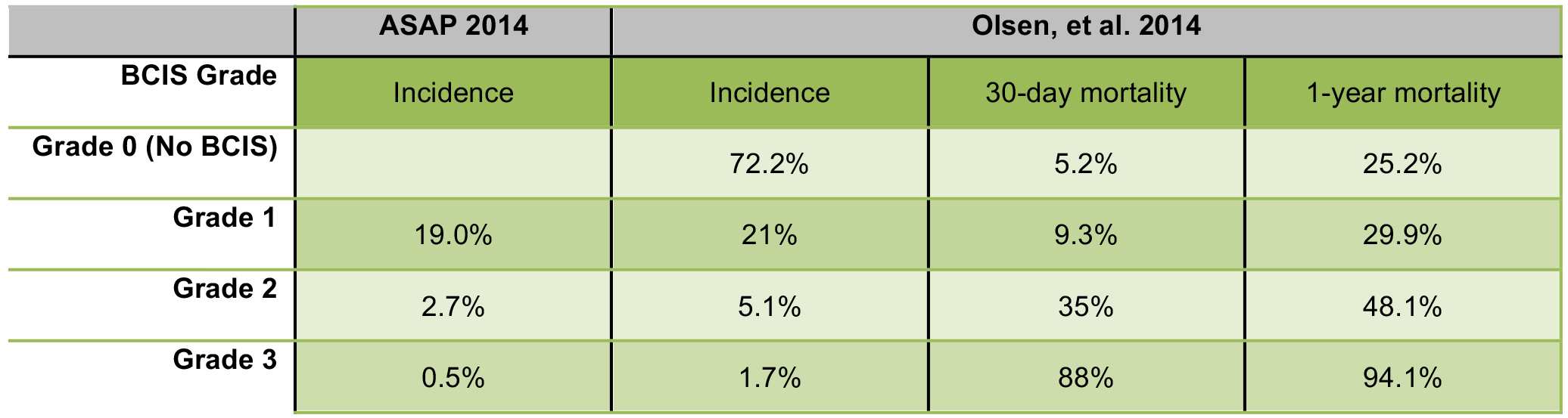
Figure 3. The incidence of BCIS in cemented hemiarthroplasties, and their respective 30-day and 1-year mortality rates.
The Olsen study showed a significantly higher mortality in patients with BCIS Grade 2 and 3, than in Grade 1. Severe BCIS (grade 3) was also a significant predictor of 30-day mortality, with an odds ratio of 16.35 (more than double the mortality of Grade 2 BCIS at 30 days, and almost double the incidence at 1 year). In patients with BCIS who died within 48 hours of surgery, 95% were found to have had severe reactions.
PATHOPHYSIOLOGY
The pathophysiology of BCIS is not fully understood, but several mechanisms have been proposed.
Embolic model
During surgical cementation and prosthesis insertion, the cement is intentionally pressurised to force it into the interstices of the bone, ideally improving bonding between the cement and bone. The cement then expands in the space between the bone and the prosthesis, further pressurising air and the medullary contents and potentially forcing them into the circulation. It has been demonstrated that these embolic contents include fat, marrow, cement, air, bone particles, and aggregates of platelets and fibrin.3
When these debris embolise, they may reach the lungs, heart, and/or coronary circulation. It is hypothesised that showers of pulmonary emboli are the cause of the characteristic hypoxia and right ventricular dysfunction of BCIS, leading to hypotension. These physiological changes from embolisation can be attributed to both the mechanical effect of the emboli and a mediator release which provokes increased pulmonary vascular tone.
Transoesophageal echocardiography has shown multiple small emboli, described as ‘snow flurries,’ visible in the heart from the start of reaming until the end of the surgical procedure. These emboli vary in size and number, with one study demonstrating emboli greater than 10mm in one-third of patients.5
Multimodal Model3 – Histamine release, hypersensitivity and complement activation
Some critics of the embolic model argue that embolisation is not always associated with haemodynamic changes, and that the degree of embolisation correlates poorly with the extent of hypotension or hypoxaemia. It is felt that the embolic model alone cannot account for all the features of BCIS, and that a combination of different factors, acting in tandem, icontributes to the full clinical picture seen in BCIS.
Anaphylaxis and BCIS share many features, notably a significant increase in plasma histamine concentration. Surgeons in frequent contact with the common cement used, Methyl Methacrylate (MMA), have been found to have an increase in blood levels of C3a and C5a. These are anaphylactoid complements and are potent mediators of vasoconstriction and bronchoconstriction. This condition has also been identified in experimental animal models, with similar results. Collectively, it is felt these mediators yield an increase in pulmonary vascular resistance, causing ventilation/perfusion disturbances, hypoxia, right ventricular failure, and cardiogenic shock (the characteristic features of BCIS).
MANAGEMENT
The Association of Anaesthetists of Great Britain and Ireland (AAGBI) published a 2015 guideline recommending a three-stage protocol for minimising the incidence and managing the occurrence of BCIS in cemented arthroplasty of hip fractures.6 :
1. Identification of patients at high risk of cardiorespiratory compromise:
- Increasing age
- Significant cardiopulmonary disease
- Diuretics
- Male sex
A 2009 National Patient Safety Agency (NPSA) Rapid Response Report highlighted that patients with poor cardiorespiratory reserve were at increased risk of perioperative morbidity from the effects of BCIS.7 Olsen et al. identified the following risk factors for severe BCIS: Grade 2 or 3 (Figure 4).

Figure 4. Odds ratio for severe BCIS, based on risk factor
Although a medical history of CHF or chronic atrial fibrillation was not itself an independent risk factor for BCIS, the use of diuretics or warfarin was identified as a risk factor for severe BCIS. It is postulated that patients with CHF, especially if associated with chronic atrial fibrillation, who require treatment with diuretics and warfarin, may have pre-exisiting pulmonary venous hypertension due to increased left-sided filling pressures.1
Factors NOT predictive of severe BCIS include:
- Arteriosclerosis
- Angina pectoris
- Congestive heart failure
- Beta-blockers
- Angiotensin-converting enzyme inhibitors.
Conditions which may potentially increase the likelihood of BCIS include osteoporosis, bone metastasis, and concomitant hip fractures. These conditions may be associated with increased or abnormal vascular channels, through which marrow contents can more readily migrate into the circulation, resulting in emboli.
2. Preparation of team(s) and identification of roles in the event of a severe reaction:
- Pre-operative multidisciplinary discussion and planning
- Pre-list briefing and WHO Safe Surgery checklist ‘time-out’
Effective communication between anaesthetists, surgeons, and ortho-geriatricians is intended to increase awareness of the potential for BCIS, enhance perioperative vigilance, reduce risk, and optimise responses should BCIS occur. It has been suggested that the step of cementation be discussed as part of the World Healthcare Organisation Safer Surgical checklist in the relevant operative procedures. A novel ‘Cement Curfew’ protocol (Figure 5) has been developed and implemented at some institutions8, which involves all members of the operating theatre team assuming specific roles and focus around the time of prosthesis insertion.

Figure 5. Cement Curfew drill, implemented by University Hospitals Coventry and Warwickshire in every trauma list and before every cemented hemiarthroplasty.
For cases identified as “high-risk”, there should be a discussion between the surgeon and anaesthetist regarding the potential benefit of uncemented versus cemented arthroplasty. The 2014 NICE Guidelines for hip fracture management in adults suggests that cemented implants are preferable to uncemented implants in patients undergoing arthroplasty, as it results in increased postoperative pain-free mobility and reduces the risk of re-operation. However, since Olsen’s 2014 BCIS study, the benefit of uncemented implants in patients with certain comorbidities is again under review.9
*Updated NICE guidelines were not yet available at the time of this publication.
3. Intraoperative roles:
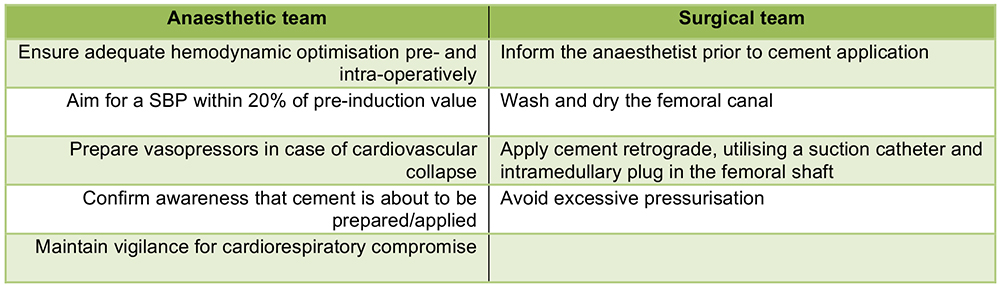
Figure 6. Key intraoperative roles for managing potential BCIS
Ensuring adequate optimisation, pre- and intra-operatively, can be done by increasing the inspired oxygen concentration in all patients at the time of cementation, avoiding intravascular volume depletion, and increasing blood pressure prior to periods of increased BCIS risk (especially during cementing). The routine use of vasopressors (even low-dose epinephrine/adrenaline infusions) during cementing is now a component of some institutions’ intraoperative protocol.
Vigilance
From the time the femoral head is removed up to the point of femoral canal instrumentation, there should be vigilance for signs of cardiorespiratory compromise. Monitor the arterial line trace or increase the frequency of the non-invasive automated blood pressure cycle during the cementing stage. Central venous pressure (CVP) monitoring can also aid in volume optimisation and inotrope administration, but may correlate poorly with changes in pulmonary arterial pressure in BCIS. The use of an intraoperative pulmonary artery catheter or trans-oesophageal echocardiography has been suggested for haemodynamic monitoring in high-risk patients, but this is not feasible in many practice settings.
The anaesthetic technique should be tailored to each individual patient and surgery. There have been no large studies on the effect of anaesthetic technique and the incidence of BCIS. However, there are surgical techniques that have been identified as reducing the incidence of BCIS (Figure 6).
Alterations in surgical technique consist of changes to minimise the likelihood of cement embolisation, including consideration of using uncemented prosthesis in high-risk patients. A recent experimental study using sheep models also explored the role of inferior vena cava (IVC) filters in preventing pulmonary embolisation and subsequent BCIS. The clinical impact of an IVC filter is yet to be determined.10 As anaesthetic management of BCIS is mainly supportive, once the decision has been made to proceed with the operation, surgical modifications are the only alterations which will affect occurrence of BCIS.

Figure 7. Surgical techniques to minimise the risk of BCIS
TREATMENT OF BCIS
Under general anaesthesia, a significant drop in systolic pressure may herald cardiovascular collapse, whilst a sudden drop in end-tidal pCO2 may indicate right heart failure leading to catastrophic reduction in cardiac output. In an awake patient under a regional anaesthetic, early signs of BCIS may include dyspnoea and/or an altered sensorium.
There is an absence of clinical trials comparing different management approaches to BCIS, so current recommendations are empiric and guided by case reports as well as basic physiological principles (Figure 7).
Cardiovascular collapse, in the context of BCIS, should be viewed and treated as right ventricular (RV) failure. In general, early and aggressive resuscitation is the cornerstone of treating BCIS. Administration of 100% inspired oxygen is a first-line therapy, with airway control dictated by clinical necessity. Invasive haemodynamic monitoring (if not already in place), should be established. In cases of severe BCIS (when the patient has arrested, or in a peri-arrest condition), standard advanced cardiopulmonary life support (ACLS) algorithms and procedures should be followed. Fluid resuscitation to maintain RV preload, and inotropes to support ventricular contractility are recommended. Vasopressors (such as phenylephrine and noradrenaline) primarily cause peripheral vasoconstriction, increase aortic blood pressure, which in turn supports coronary artery blood flow, and thus improve myocardial perfusion and contractility. Use of vasopressors and inotropes should be continued into the postoperative period as necessary, under the management of the intensive care unit (ICU).

Figure 8. Key recommendations for initial response to BCIS11
BCIS is a time-limited phenomenon; with human and animal studies strongly suggesting that pulmonary artery pressure normalises within 24 hours.12 Even with large embolic loads, healthy hearts may recover in seconds to minutes. The underlying mechanism – acute pulmonary hypertension and secondary right ventricular failure – should be considered reversible. Aggressive stabilisation and supportive therapy are the cornerstones in managing BCIS.
For patients who have not met the criteria for severe BCIS but who have a suspicious clinical picture, they should be monitored closely in a high-dependency unit for at least the first 24 hours after the operation.
SUMMARY
- BCIS is a potentially fatal complication of orthopaedic surgery.
- Pulmonary embolisation from intramedullary contents is the likely aetiology of BCIS.
- Rapid hypoxia, hypotension and loss of consciousness are the key signs.
- Good management of BCIS includes: patient risk stratification, intraoperative vigilance, good team communication, and prompt resuscitation.
ANSWERS TO QUESTIONS
- Bone Cement Implantation Syndrome (BCIS) has the following features:
a. True. This is caused by pulmonary emboli created from intramedullary pressure during cementation.
b. True.
c. True.
d. False. BCIS has also been described in other cemented procedures, including hip and knee replacement
e. False. Although BCIS typically occurs during cementation, it can also occur before or after cementation; during femoral reaming, prosthesis insertion, joint reduction, or tourniquet deflation. - The following patient characteristics are associated with an increased risk of developing BCIS:
a. True.
b. True.
c. False. An un-instrumented femur has more potentially embolic material, whereas a previously instrumented femur’s inner surface is more sclerotic and hence less permeable.
d. True.
e. False. - Key steps for managing patients at risk of Bone Cement Implantation Syndrome:
a. True.
b. True.
c. False. Vigilance should be enforced earlier on, when the femoral head is removed and the surgeon communicates their intent to instrument the femoral canal.
d. False. Nitrous oxide increases the risk of air embolism.
e. False. Retrograde cement injection in the femoral canal is one method for minimising the incidence of BCIS.
REFERENCES AND FURTHER READING
- Olsen F, Kotyra M, Houltz E et al. Bone cement implantation syndrome in cemented hemiarthroplasty for femoral neck fracture: incidence, risk factors, and effect on outcome. Br J Anaesth. 2014;113:800-806
- Griffiths R, Parker M. Bone cement implantation syndrome and proximal femoral fracture. Br J Anaesth. 2015;114:6-7
- Donaldson AJ, Thomson HE, Harper NJ et al. Bone cement implantation syndrome. Br J Anaesth. 2009;102:12-22
- National Hip Fracture Database. Anaesthesia Sprint Audit of Practice (ASAP). https://www.aagbi.org/sites/default/files/NHFD%20anaesthestic%20report.pdf (accessed on 08/08/2016)
- Lafont ND, Kalonji MK, Barre J et al. Clinical features and echocardiography of embolism during cemented hip arthroplasty. Can J Anesth. 1997;44:112-117
- Association of Anaesthetists of Great Britain and Ireland, Membership of the Working Party, Griffiths R, White S, Moppett M et al. Safety guideline: reducing the risk from cemented hemiarthroplasty for hip fracture 2015. Anaesthesia. 2015;70:623-626
- National Patient Safety Agency. Mitigating surgical risk in patients undergoing hip arthroplasty for fractures of the proximal femur. http://www.nrls.npsa.nhs.uk/resources/type/alerts/?entryid45=59867 (accessed on 08/08/2016)
- Scrase A, Horwood G, Sandys S. Coventry “Cement Curfew”: team training for crisis. Anaesthesia News 2014;327:8-9
- National Institute for Health and Care Excellence (NICE). Surveillance report – Hip fracture (2011) NICE guideline CG124. https://www.nice.org.uk/guidance/cg124 (accessed on 08/08/2016)
- Guo W, Zheng Q, Li B et al. An Experimental Study to Determine the Role of Inferior Vena Cava Filter in Preventing Bone Cement Implantation Syndrome. Iran J Radiol. 2015 Jul 22;12(3)
- Association of Anaesthetists of Great Britain and Ireland, Membership of the Working Party, Griffiths R, Alper J, Beckingsale A et al. Management of proximal femoral fractures 2011. Anaesthesia. 2012;67:85-98
- Byrick, R.J. Cement implantation syndrome: a time limited embolic phenomenon. Can J Anaesth (1997) 44: 107.
- Patterson BM, Healey JH, Cornell CN et al. Cardiac arrest during hip arthroplasty with a cemented long-stem component. A report of seven cases. J Bone Joint Surg Am. 1991;73:271



