General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Which of the following statements is correct?
- Blood groups are determined by antibodies carried in the plasma
- Blood group A- can be transfused into a person with group AB+
- The most common blood group type is O+
- Anti A and Anti B antibodies are present in blood group O foetal blood
- With regard to transfusion compatibility;
- Group AB+ can be considered to be a ‘universal recipient’
- A+ blood can be given to an A- recipient
- A- blood can be given to an A+ recipient
- In an emergency O+ blood can be used
- Recommended routine testing of donated blood includes the following;
- HIV infection
- Hepatitis C antibody
- A full coagulation screen
- Syphylis serology
- Typical values found in blood stored for 35 days include the following;
- pH 7.0
- Potassium level 27mmol/l
- Haematocrit approximately 60% – 70%
- Platelets 50 x 109
INTRODUCTION
As long ago as 1666 Richard Lower performed the first transfusion in animals, but it is only in the last century since Karl Landsteiner discovered blood groups (1900) that blood transfusion has become a conventional therapeutic intervention in humans. In modern medical practice transfusion of blood products has become commonplace, and in 2004, 3.4 million blood components were issued in the UK. The most common indications for transfusion are anaemia (23%), orthopaedic surgery (14%), haematological disease (15%), gastro intestinal bleeding (11%), general surgery (10%), cardio thoracic surgery (6%), obstetrics & gynaecology (6%), vascular surgery (5%) and urology (3%). To make transfusion practice as safe as possible requires an understanding of the basic immuno-physiology of blood and this article, which is the first of a series, sets out to describe blood groups, blood typing, and the basics of donation and storage of blood. A brief overview will also be given of blood components, and the problems associated with blood storage will be discussed (the ‘lesion’ of stored blood).
BLOOD GROUPS
Blood groups are inherited and are determined by the antigens carried on the surface of red blood cells. There are as many as 30 different blood groups, but the main ones are the ABO and rhesus groups, and these will be discussed further.
The ABO system
ABO is the most important system, and is named for the A and B antigens carried on the surface of the red blood cells (Fig 1). Persons with blood group A have the A antigen, B have the B antigen, and AB both. Group O has neither. The ABO system has associated anti-A and anti-B antibodies that are found in the plasma. Since blood group A has the A antigen, it recognises the B antigen as foreign and forms anti-B antibodies. Similarly, blood group B recognises the A antigen as foreign and can make anti-A antibodies. Group AB has both the A and B antigens so this group makes no antibodies. Group O has neither A nor B antigen so can form both anti-A and anti-B antibodies if exposed to these antigens. Human beings develop plasma antibodies against the A and B antigens very early on in life.
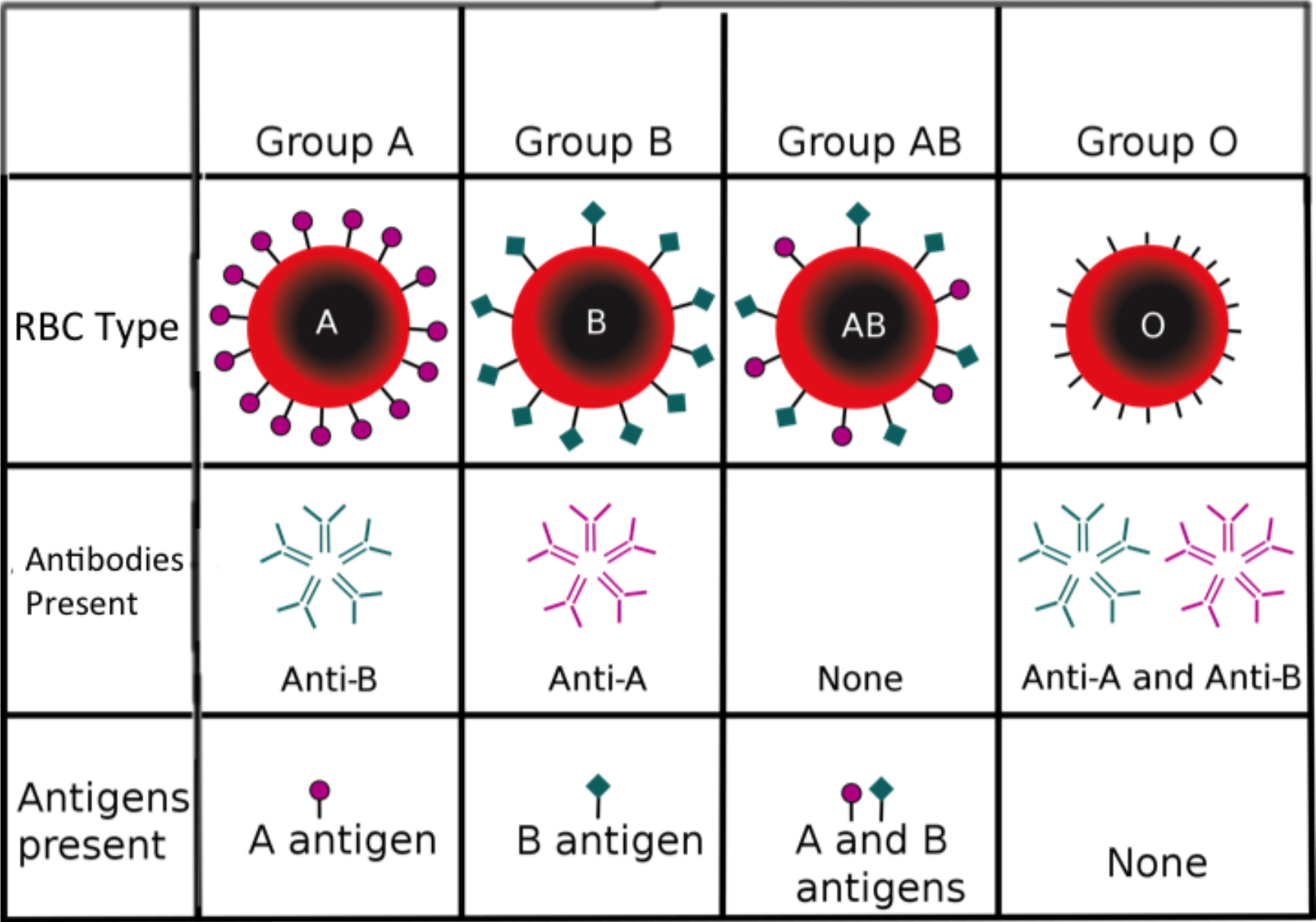
Figure 1.The four blood groups in the ABO system, with their associated red cell antigens and plasma antibodies.
The Rhesus system
The second most important blood group system is the ‘Rhesus Group’ named for its similarity to blood groups in rhesus monkeys. A blood type is most frequently defined both by the ABO and rhesus (RhD) systems, where the letter stands for the ABO type, and the sign indicates the rhesus status. If the D antigen is present the person is said to be positive (+), and if absent is negative (-). In the UK approximately 85% of people are positive and 15% negative (table 1). The rhesus blood group is particularly important in the context of pregnancy because a D positive baby can cause a D negative mother to develop antibodies, which can cross the placenta to affect future D positive babies. In the Far East the D negative group is less common and so there are fewer problems associated with pregnancy and the D blood group. The prevalence of each of the blood groups present in the population varies in different parts of the world (table 1).
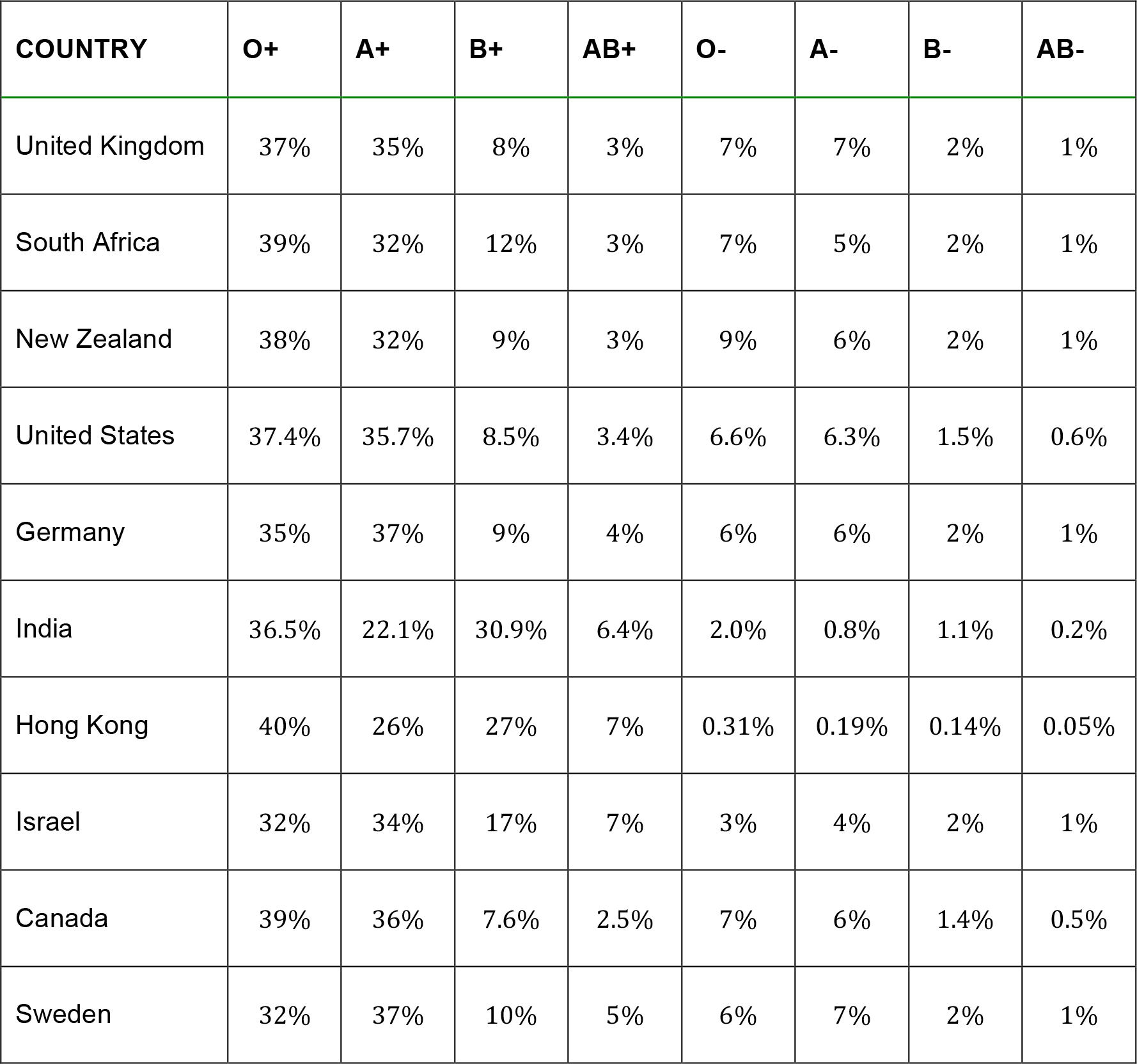
Table 1.Prevalence of blood types by country.
CROSS-MATCHING
Blood grouping characterises the blood by the ABO and rhesus systems, whereas cross-matching refers to the testing for compatibility between donor and recipient blood before transfusion occurs. The compatibility is determined not only by the main groups of the ABO and rhesus systems, but also all the minor systems (eg Kell). The two methods for cross-matching include electronic and serological.
Electronic cross-matching relies on computer analysis of tests done on the donor unit of blood and blood samples from the intended recipient. Information includes ABO and Rh typing of donor and recipient, as well as an antibody screen of recipient (patient) blood. If the patient serum has a negative screen, it means that there are no detectable atypical antibodies against red blood cells of the donor unit. Based on this data the computer will indicate that the unit of blood is safe to transfuse.
Serological cross-matching requires that red blood cells from the donor blood are suspended in serum from the patient (recipient). If the serum contains antibodies against antigens on the donor red cells they will cause agglutination and the test will be said to be positive, indicating that there is incompatibility. If no agglutination occurs then the blood is safe to transfuse.
The testing described above is called a major cross-match, where donor red cells are tested against recipient serum. In contrast, a minor cross-match tests recipient red cells against antibodies that may be present in the donor serum, but this is no longer a requirement since the very small amount of serum (and antibodies) remaining in a unit of packed cells is so small as to have little clinical effect.
The full cross-matching process takes approximately 30-60 minutes, which may be too long in urgent or emergency cases. In urgent cases the laboratory can issue un-crossmatched, group-specific blood (e.g. A+ blood for an A+ patient), or in an emergency where the blood type of the recipient is unknown, O-negative blood. Since O-negative blood has no antigens it is considered to be the ‘universal donor’ blood group (table 2 and figure 2). Even though there are risks associated with giving un-crossmatched blood, the risks of exsanguination are considered to be greater.
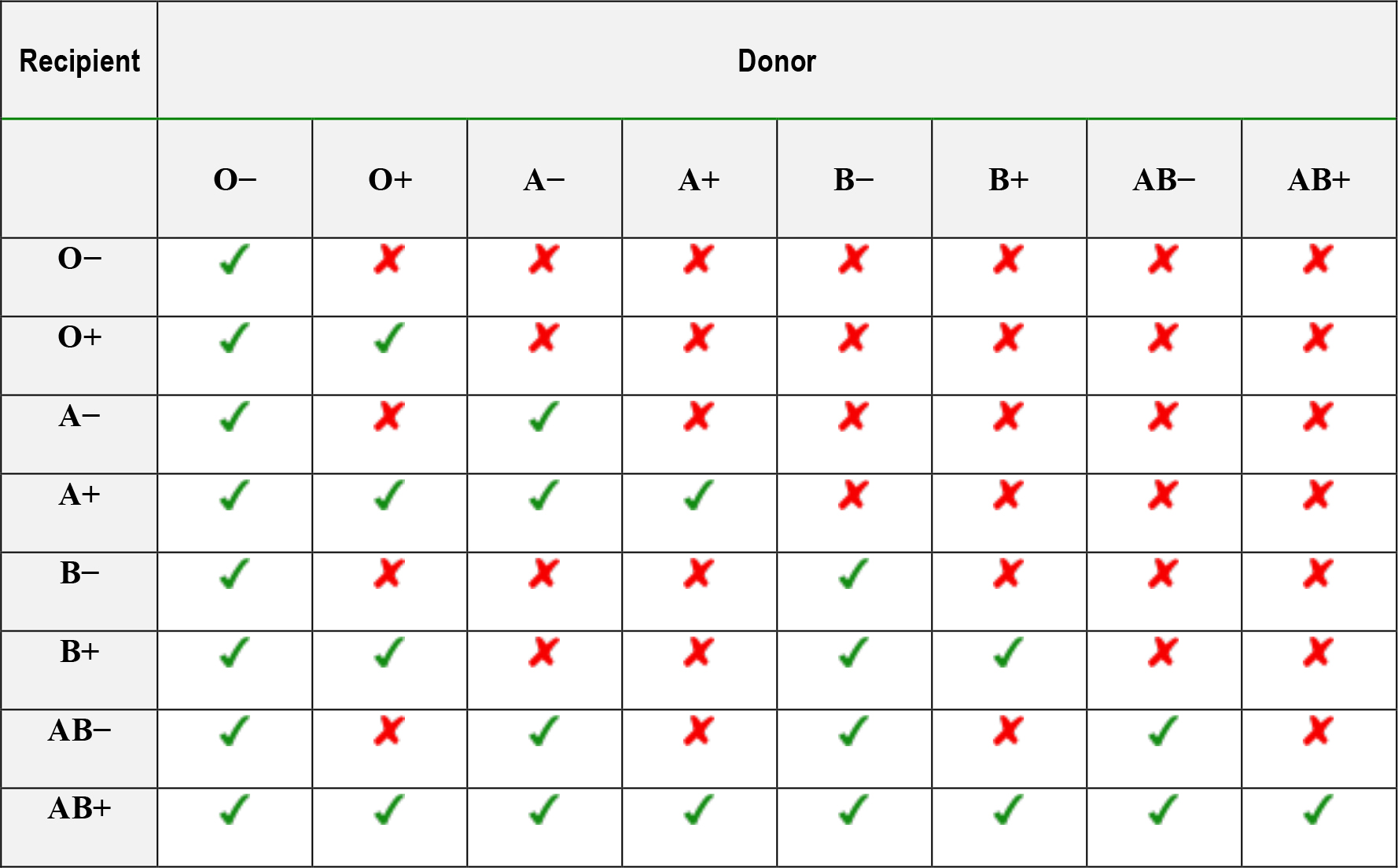
Table 2.Compatibility table for the transfusion of red blood cells.
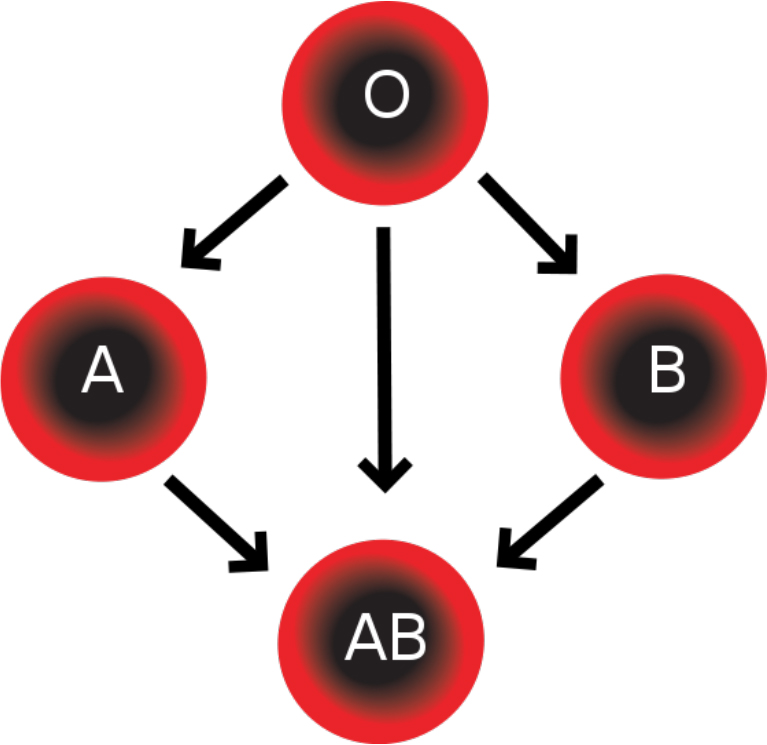
Figure 2.Group O (universal donor) can be given to any recipient, while other safe donor transfusions are indicated by the arrows.
DONATION, TESTING AND STORAGE
There are risks associated with both donating and receiving blood, but these can be minimised by good practice. Donors have their current health assessed by the Donor Health Check (DHC), which includes a history and examination (temperature, pulse and blood pressure) to make sure that donating will not be a hazard to their health. Haematocrit or haemoglobin level is also assessed since risk of anaemia is the most common reason for ineligibility. To ensure recipient safety the donor is also screened for health risks that might make the donation unsafe. Donation may be turned down for homosexual men owing to HIV risk, and persons on certain medications. Donors are examined for risk and signs of transmissible diseases such as HIV, malaria, viral hepatitis and variant Creutzfeldt-Jacob Disease (vCJD).
The blood that is collected is identified for blood type (ABO and Rh), but is also tested to minimise the risk of transmissible infections. The World Health Organisation recommends four core tests as a minimum, which are:
- Hepatitis B Surface Antigen
- Antibody to Hepatitis C
- Antibody to HIV, usually subtypes 1 and 2
- Serology for Syphylis
However, compliance with this standard is not universal and in 2006 there were 56 out of 124 countries surveyed that did not use these tests on all blood donations.
Many countries carry out other tests based on local needs, which include West Nile Virus, Chagas Disease, and Cytomegalovirus (CMV). The tests used are high-sensitivity screening tests which have a relatively high incidence of false positives. In the case of positivity the blood is discarded, with the exception of autologous transfusions where donors receive their own blood back. False negatives are very uncommon. No test is infallible since recent infections may be missed within the ‘window period’ when antibodies have not yet formed against the infection. However, owing to the stringency of testing and screening, the main risk from a blood transfusion is not infection but being given blood of the wrong blood group.
For patients with specific risks from transfusion, the blood may undergo further processing. Leukoreduction is the removal of white blood cells by filtration, after which the blood product is less likely to cause alloimmunization (formation of antibodies), febrile reactions, CMV infections, and platelet refractoriness. Pathogen Reduction treatment may involve the addition of riboflavin with subsequent exposure to UV light, and helps to inactivate pathogens (viruses, bacteria and parasites), as well as inactivating white blood cells to prevent graft-versus-host disease.
Storage
Short-term storage of blood relies on a combination of refrigeration and the addition of preservatives. The standard temperature used is 1°- 6°C, but the preservatives may vary. When using acid-citratedextrose (ACD), citrate-phosphate-dextrose (CPD) or citrate-phosphate-double dextrose (CP2D), the storage is limited to 21 days, but with citrate-phosphate-dextrose-adenine (CPDA1) this can be extended to 35 days.
Red blood cells can be stored for much longer periods, even up to 10 years, by freezing (cryopreservation). This requires the cells to be incubated in a glycerol solution which acts as an intracellular “antifreeze”, so protecting against damage. The units are then placed in special sterile containers and frozen to below -60°C. The exact temperature depends on the glycerol concentration used.
Blood components
Whole blood is rarely used for transfusion anymore, and only the components that are specifically required by the patient are used. The collected blood is therefore fractionated into its respective components by centrifugation to produce packed red blood cells, plasma, platelets, albumin, clotting factor concentrates, cryoprecipitate, fibrinogen concentrate, and immunoglobulins. Another process that may be used is apheresis, where only the component that is to be used is separated out and collected, and the rest returned to the donor.
- Packed Red cells: A bag of RBCs have a haematocrit of between 60-70%, and an average shelf life of 35 days if properly stored. Their function is to act as carriers of oxygen and carbon dioxide in the blood. The main indications for transfusion is the correction of anaemia or replacement in acute haemorrhage, but there is no absolute level of Hb to trigger a transfusion (see part 2 of tutorial). A single unit of red blood cells will typically increase the Hb by 1g/dl.
- White Blood cells (granulocytes): are collected by apheresis (filtration), and can only be stored for 24 hours at 20-24°C. They need to be cross-matched because they contain large numbers of RBCs, and also irradiated to remove the lymphocytes. They are indicated for life-threatening infections in neutropenic cancer patients who are unresponsive to antibiotics.
- Platelets: A unit of platelets is prepared from a single whole blood collection and contains at least 5.5 x 1010 platelets in 50 ml plasma. They are stored at 20-24°C under agitation and have a shelf life of 5 days. Each unit can raise the platelet count by 5-10 x 109. Platelets are not usually crossmatched with the recipient, but where possible ABO specific platelets should be used. The indication for use is thrombocytopenia secondary to various causes.
- Fresh Frozen Plasma: is collected as the supernatant after centrifuging a donation of whole blood. It is frozen within 8 hours to maintain the activity of factors V and VII. The main indication is deficiency of multiple coagulation factors such as that found in massive haemorrhage, DIC, liver disease, and occasionally for reversal of warfarin effect.
- Cryoprecipitate: is made by thawing frozen FFP at 1-6°C, and is indicated for von Willebrand’s Disease and severe hypofibrinaemia.
- Albumin: is available as a 5% and 25% solution. The strongest indication for use is expansion of blood volume after removing ascites fluid, but its use has largely been superseded by non-human colloidal solutions. It is tested for Hepatitis C and is virally inactivated, and so considered very safe.
- Immunoglobulin: specific immunoglobulins can be given to treat specific infections, e.g. chicken pox and Hepatitis B. They are also given for certain immune-related diseases such as Guillain-Barre syndrome. Anti-D immunoglobulin is given to rhesus negative pregnant women who carry an Rh positive baby to prevent Rh sensitisation and future haemolytic disease of the newborn.
- Coagulation factor concentrates: specific factors can be transfused individually to treat specific factor deficiencies. These include Antithrombin III, Factor VIII, Factor IX, Factor XIII, and Protein C. All of these are prepared from human plasma.
LESION OF STORED BLOOD
Despite refrigeration and the addition of preservatives, the quality of donated red blood cells deteriorate over time as predictable cellular and biochemical changes occur that result in the ‘storage lesion’ of banked blood (table 3).
Red blood cells
Red cells that are drawn at the beginning of donation are subjected to an acidic and hypotonic anticoagulant solution, which damages a small proportion of them irreversibly. Cells that survive the first 24 hours will remain viable for the rest of their expected life span, but during storage they depend on anaerobic glycolysis for energy production and viability. Their shelf life requires that at least 70% of them remain viable in the recipient’s circulation 24 hours after transfusion.
Other cellular elements
Granulocytes become non-functional after 24 hours of storage, but still retain antigenic potential to cause febrile reactions. Some lymphocytes may remain viable for several weeks. Platelet function declines to zero after only 48 hours of storage.
Oxygen affinity
Stored blood has depleted levels of 2,3 Diphosphoglycerate (DPG), which causes the oxygen dissociation curve to shift to the left, so increasing affinity of Hb for O2 (less O2 release to tissues). The 2,3 DPG levels rise rapidly following transfusion and normal oxygen affinity is usually restored within a few hours.
Coagulation
Stored blood contains an anticoagulant, commonly in the form of citrate, but this usually does not affect recipient coagulation status (see below). The labile coagulation factors V and VIII have a 50% decline in activity within the first 72 hours of storage, but all other factors remain normal.
Temperature
The optimum storage temperature of 2°- 6°C is well below normal body temperature.
Electrolytes
Potassium: During storage there is a constant leak of potassium out of cells, and levels may sometimes exceed 30mmol/l. This is usually not a problem since the potassium rapidly re-enters red cells as they begin active metabolism after transfusion. It may, however, become a problem with rapid transfusion of large amounts of stored blood, in severe renal failure, or in neonatal exchange transfusion.
Calcium: Each unit of blood contains approximately 3g citrate, which binds ionized calcium. However, the liver is able to metabolise 3g of citrate every 5 minutes, so that only when transfusion rates are greater than one unit per five minutes or in the presence of impaired liver function is there a risk of citrate toxicity and hypocalcaemia. The resultant hypocalcaemia does not have a clinically apparent effect on coagulation, but patients may exhibit transient tetany and hypotension. Calcium should only be given if there is biochemical, clinical or electrocardiographic evidence of hypocalcaemia.
Acid/Base
During storage there is a gradual accumulation of lactic acid which can result in an acid load of 30- 40mmol/l and a resultant fall in pH. However, this and the citric acid load is usually metabolised rapidly. Because the citrate is metabolised to bicarbonate there can be a paradoxical metabolic alkalosis associated with transfusion.
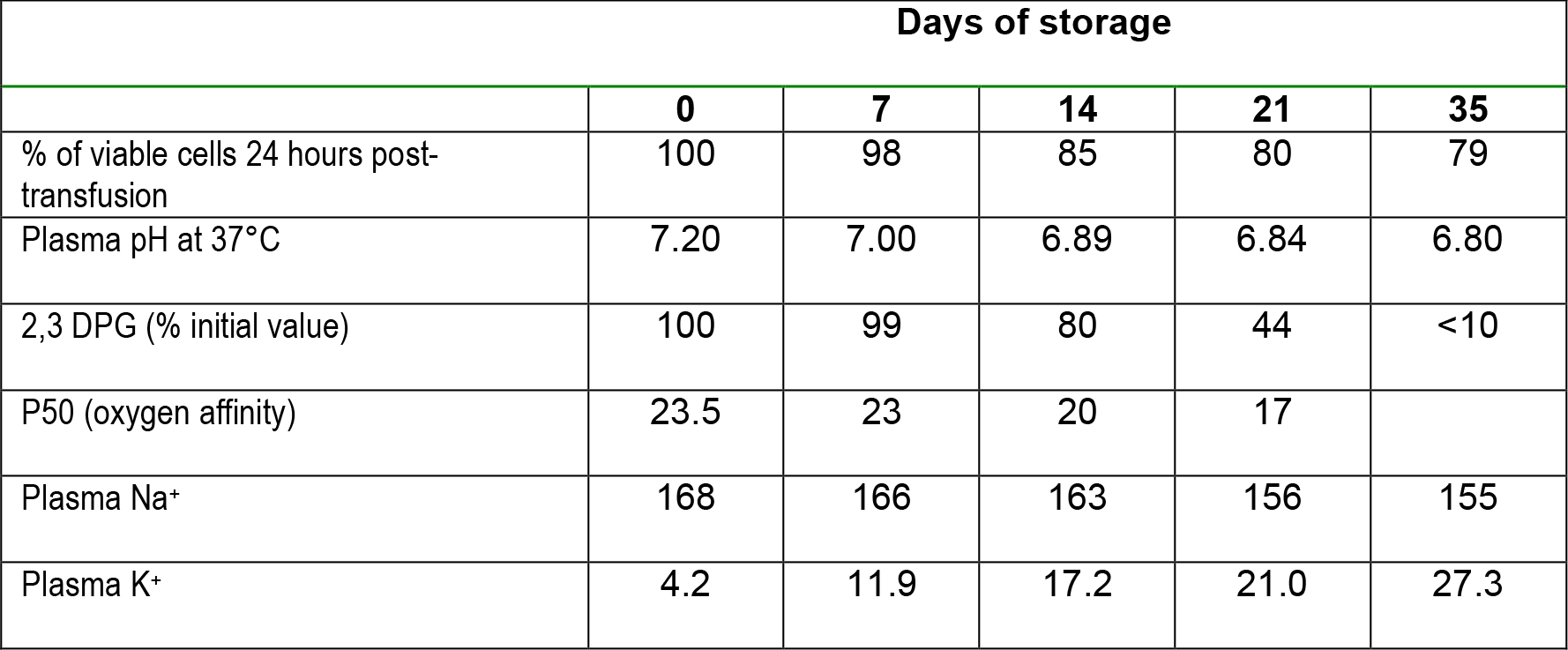
Table 3.Typical changes associated with the storage of blood.
IMPORTANT POINTS
- A basic knowledge of the major blood groups is required for the safe practice of transfusion
- Blood components should be used rationally with specific indications in mind
- The changes that occur during storage of blood makes it easier to understand the problems that can be anticipated with its use
- Use of any of the products of donated blood carries a risk that needs to be weighed against the benefits of transfusion for that patient
ANSWERS TO QUESTIONS
- F – Blood groups are determined by antigens on the red cell membrane
T – AB+ is a universal recipient
T
F – Anti A and Anti B antibodies are formed in early extra-uterine life - T
F – A- recipient may form antibodies to the D antigen
T
F – In an emergency O negative blood should be used - T
T
F – this is irrelevant since the blood will be anticoagulated
T - F – pH will be approximately 6.80
T
T
F – there are virtually no platelets present after 48 hours
WEBLINKS
- NHS Blood and Transplant Website http://www.blood.co.uk/



