Neuroanaesthesia
KEY POINTS
- The trigeminocardiac reflex (TCR) and glossopharyngeal-vagal reflex (GVR) are usually caused by the surgical stimulus of peripheral nerves during neurosurgery and they both can potentially lead to bradyarrhythmia.
- The TCR is a well-described brainstem reflex which is characterised by bradycardia, hypotension, apnoea, and gastric hypermobility.
- The Bezold-Jarisch reflex (BJR) is a cardio-inhibitory reflex that presents as symptomatic bradycardia and hypotension in the presence of hypovolemia.
- All reflexes may lead to severe haemodynamic instability and require prompt recognition and management.
INTRODUCTION
Hemodynamic alterations are seen in different neurosurgical operations and commonly relate to various brainstem reflexes. Neural communication pathways interacting between the heart and brain are responsible for the generation of various intraoperative heart rate and blood pressure disturbances. These hemodynamic disturbances range from transient episodes of bradycardia, hypotension, and hypertension to more severe haemodynamic instability including cardiac arrest.
It is important for anaesthetists providing perioperative care to neurosurgical patients to have an awareness of the potential brainstem reflexes that may occur and an understanding of the aetiologies and mechanisms that may precipitate them to provide appropriate and timely management. These brainstem reflexes include the trigeminocardiac reflex (TCR), glossopharyngeal-vagal reflex (GVR), and Bezold-Jarisch reflex (BJR).
TRIGEMINOCARDIAC REFLEX
The TCR is a well-known brainstem reflex and the most common cause of severe haemodynamic disturbance during supratentorial surgery.1 It clinically manifests as acute onset of bradycardia and hypotension during stimulation of any of the branches of the trigeminal nerve. It may be associated with asystole, gastric hypermotility, and apnoea. The typical cardiac manifestation of the TCR is bradycardia but other dysrhythmias can occur. As onset may be sudden and without warning signs, prompt recognition and therapeutic intervention are essential to ensuring a good outcome.
The TCR was first described by Schaller et al2 in 1999 in a series of patients undergoing cerebellopontine angle and brainstem surgery and was defined as a drop in heart rate to less than 60 beats/min and a drop in mean arterial blood pressure of more than 20%. This has since become an accepted definition of the TCR. However, this definition may underestimate the actual number of TCR episodes as many of them may show lesser degrees of change in heart rate or blood pressure. Alternatively, the TCR may be defined as a cardiac reflex which may or may not be associated with hemodynamic changes upon stimulation of the trigeminal nerve anywhere along its course.3
Any form of stimulation (mechanical, chemical, electrical, and/or thermal) can incite the TCR but stretch is considered the most powerful stimulus.
Reflex Arc
The sensory nerve endings of branches of the trigeminal nerve (cranial nerve V) transmit neuronal signals via the Gasserian ganglion to the sensory nucleus of the trigeminal nerve located in the floor of the fourth ventricle, forming the afferent pathway of the reflex arc. The short internuncial fibres of the reticular formation connect the afferent (trigeminal) and efferent nuclei (dorsal motor nucleus of vagus). This results in activation of cardio-inhibitory parasympathetic vagal neurons and leads to the clinical manifestations of the TCR.
Risk Factors
There is a strong correlation between severity of bradycardia and light anaesthesia and this is considered an independent risk factor for stimulation of the TCR. Other factors including hypoxia, hypercarbia, fast-acting opioids, and acidosis are thought to augment the TCR.3,4
Incidence and High-Risk Surgeries
Meuwly et al5 analysed the prevalence of the TCR in different anatomic regions during neurosurgical procedures (Table 1).

Table 1. Trigeminocardiac reflex (TCR) prevalence by surgery type.
Types of TCR
Classically, the TCR was divided into 3 subtypes depending upon the location of the triggering point in relation to the Gasserian ganglion3 (Figure 1).
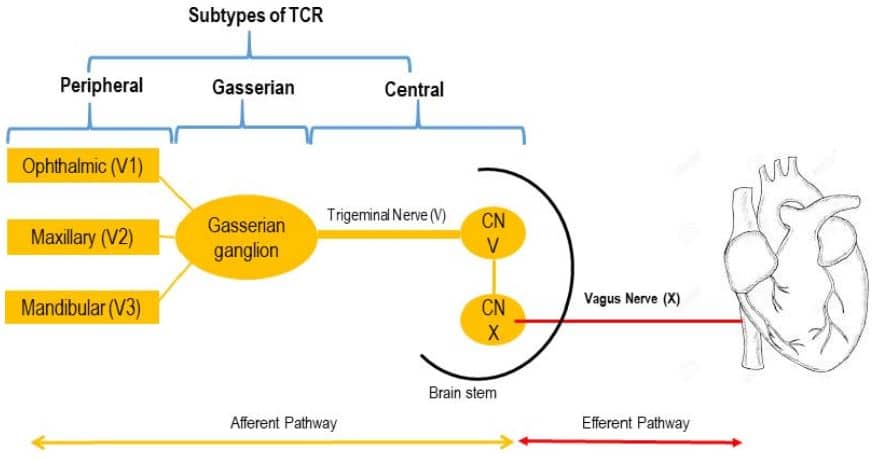
Figure 1. Reflex arc and its types.
- Central (proximal) subtype: the trigger point is intracranial and proximal to the Gasserian ganglion
- Peripheral (distal) subtype: the trigger point is the extracranial course of the trigeminal nerve
- Gasserian ganglion subtype
Recently, Leon-Ariza et al6 devised a novel classification based on morphophysiological aspects of the TCR to allow quick perioperative identification and intervention. According to this classification, type would help to identify the anatomical distribution and class would identify the behaviour of the cardiovascular variables involved with the TCR.
- Type I: stimulation of first branch (V1) of trigeminal nerve, eg oculo-cardiac reflex
- Type II: stimulation of second branch (V2) of trigeminal nerve, eg maxillo-cardiac reflex
- Type III: stimulation of third branch (V3) of trigeminal nerve, eg mandibular-cardiac reflex
- Type IV or extra-trigeminocardiac reflex: may occur due to stimulation of trigeminal nerve anastomoses originating from structures anatomically distant from brainstem structures such as stomach and lumbosacral spinal cord
Classes of TCR
- Class A: if the initial changing haemodynamic parameter is heart rate
- Class B: if the initial changing haemodynamic parameter is mean arterial blood pressure
Management
The initial steps are to inform the surgeon, immediately cease surgical stimulation and review the depth of anaesthesia. Often this alone terminates the reflex and allows restoration of hemodynamic stability. If bradycardia is severe or persistent, administer an anticholinergic such as atropine 500-600 µg (maximum 3 mg) or glycopyrrolate (4–5 µg/kg up to a maximum of 400 µg). Meanwhile, consider ceasing drugs that may cause bradycardia like remifentanil, phenytoin, and dexmedetomidine and follow the ACLS (Advanced Cardiac Life Support) algorithm for bradycardia in resistant cases. If the patient develops cardiac arrest (asystole), start cardiopulmonary resuscitation as per cardiac arrest in neurosurgical procedure and ACLS guidelines7 (Figure 2).
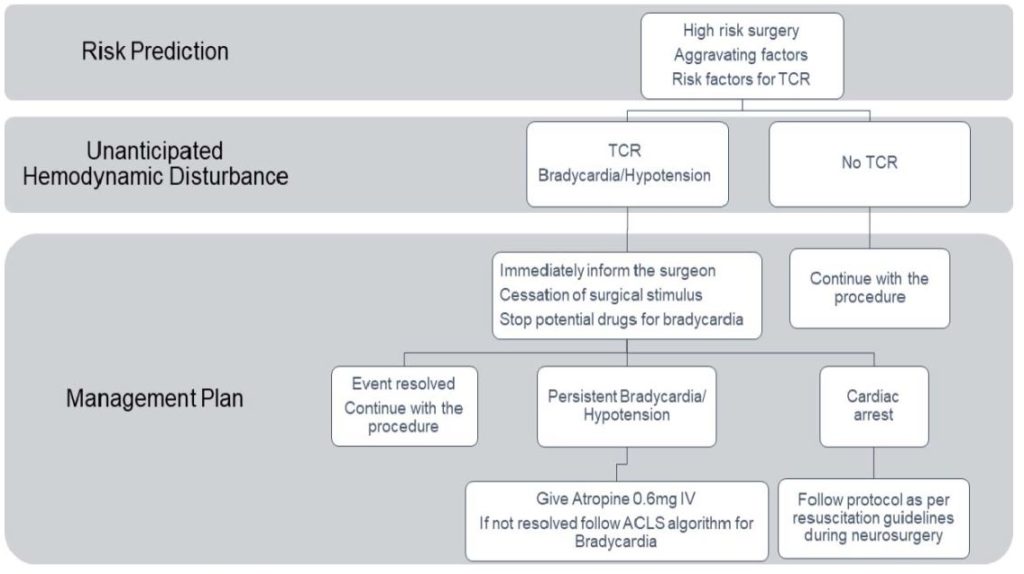
Figure 2. Trigeminocardiac reflex management algorithm.
GLOSSOPHARYNGEAL-VAGAL REFLEX
The GVR is also postulated as a mechanism for bradycardia and cardiac arrest in posterior fossa surgeries.8
Reflex Arc
Afferent fibres of the glossopharyngeal nerve transmit neuronal signals via the tractus solitarius and solitary nucleus of the midbrain to the dorsal motor nucleus of the vagus nerve, which stimulates the parasympathetic fibres that lead to bradycardia, hypotension, and sometimes asystole (Figure 3).
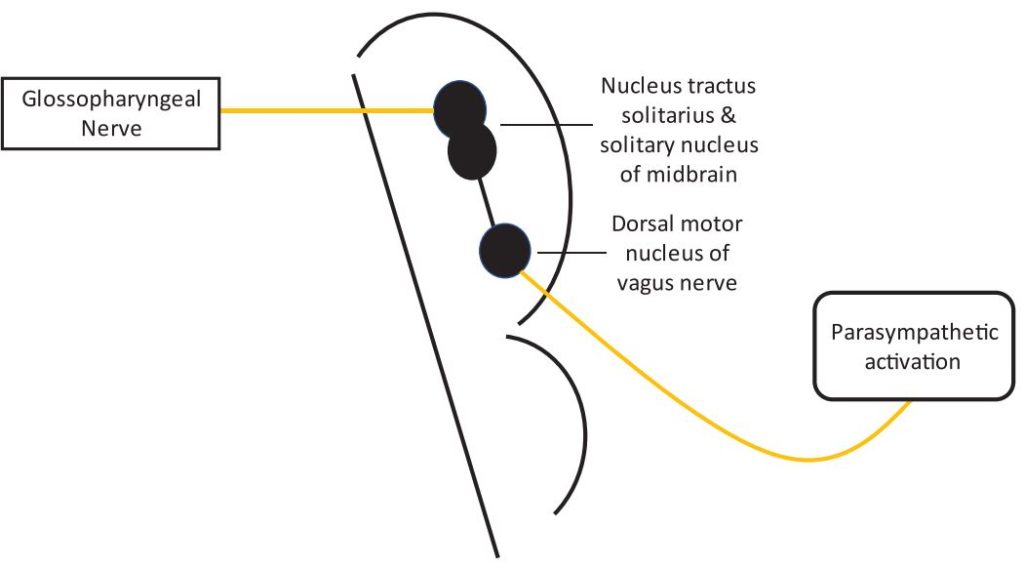
Figure 3. Glossopharyngeal-vagal reflex arc.
Treatment
The management of the GVR is similar to that of the TCR.
BEZOLD-JARISCH REFLEX
The BJR is a cardio-inhibitory reflex that presents as haemodynamic instability such as symptomatic bradycardia and hypotension in the presence of hypovolemia.9 This can mimic the picture of venous air embolism but in venous air embolism there is usually a significant drop in end-tidal CO2 as well.
Risk Factors
Neurosurgical positions such as sitting, semisitting, and supine with elevated head lead to peripheral venous pooling. Additionally, preoperative fasting and osmotic diuretics may contribute to a state of hypovolemia. BJR can also be stimulated by major haemorrhage during surgery.
Reflex Arc
An initial response to hypovolemia is activation of carotid sinus baroreceptors leading to increased heart rate, vasoconstriction, and an increase in contractility of the myocardium. However, in an underfilled hypercontractile heart, intramyocardial C fibres (pressure receptors) may be stimulated, leading to increased vagal tone and arterial vasodilatation, which can result in hypotension and bradycardia (Figure 4).
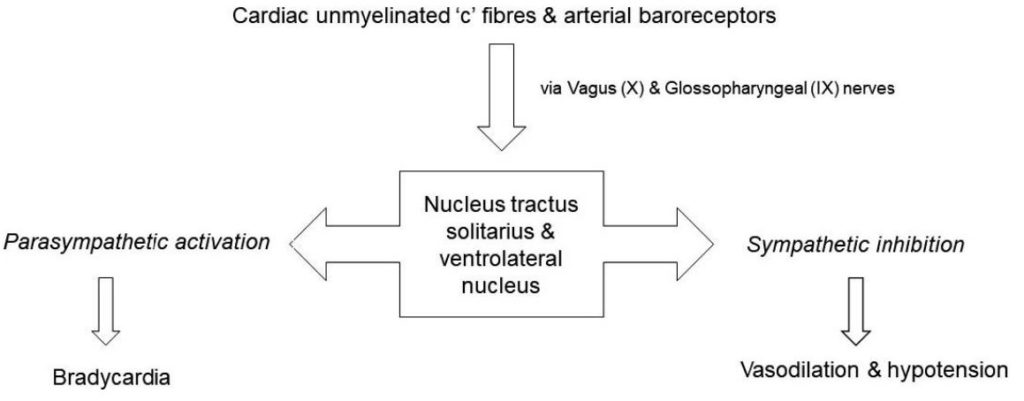
Figure 4. Bezold-Jarisch reflex arc.
Treatment
During anaesthesia, the treatment for BJR includes immediate fluid administration and early use of sympathomimetics and anticholinergics (atropine and glycopyrrolate).10 Ephedrine is the drug of choice because it counteracts both the vasodilation and slow heart rate. If deterioration to asystole occurs, epinephrine is essential. Additionally, restoration of venous return by changing the patient’s position to supine may be helpful.
OTHER CAUSES OF VAGUS NERVE STIMULATION DURING NEUROSURGERY
Increased parasympathetic outflow can be related to direct surgical manipulation: for example anterior hypothalamic stimulation during skull base surgery and direct brainstem stimulation during posterior fossa surgery.11 Likewise, in epilepsy surgery, numerous cases of bradycardia and sinus arrest have been attributed to stimulation of parts of the limbic system such as the amygdala, insular cortex, and hippocampus.12 This suggests a connection between the limbic system and autonomic nervous system but the true mechanism remains unclear.
Saline bolus irrigation of the operative field during supratentorial and infratentorial surgeries can also produce severe bradycardia and sinus arrest due to increased parasympathetic outflow via the vagus nerve.13,14
Rapid drainage of cerebrospinal fluid during ventriculoperitoneal shunt or extraventricular drain insertion can result in intracranial hypotension and reverse herniation of the brain that is occasionally associated with bradycardia and asystole. Similarly, negative-pressure epidural or subgaleal drain placement at the end of a procedure may lead to episodes of bradycardia and hypotension.15
SUMMARY
- Brainstem reflexes are commonly seen during elective neurosurgical procedures resulting in haemodynamic alterations.
- These haemodynamic perturbations are often transient in nature and resolve spontaneously without any pharmacological interventions.
- However, on some occasions, if they are not recognised and managed in an appropriate and timely manner, they can deteriorate into catastrophic cardiac complications.
REFERENCES
- Chowdhury T, Petropolis A, Cappellani RB. Cardiac emergencies in neurosurgical patients. Biomed Res Int. 2015;2015:751320.
- Schaller B, Probst R, Strebel S, et al. Trigeminocardiac reflex during surgery in cerebellopontine angle. J Neurosurg. 1999;(90):215-220.
- Chowdhury T, Mendelowith D, Golanov E, et al. Trigeminocardiac reflex: the current clinical and physiological knowledge. J Neurosurg Anesthesiol. 2015;(27):136-147.
- Meuwly C, Chowdhury T, Sandu N, et al. Anaesthetic influence on occurrence and treatment of the trigeminocardiac reflex: a systematic literature review. Medicine (Baltimore). 2015;94(18):e807.
- Meuwly C, Chowdhury T, Sandu N, et al. Meta-areas of the trigeminocardiac reflex within the skull base: a neuroanatomic ‘‘thinking’’ model. J Neurosurg Anesthesiol. 2016;(28):437-438.
- Leon-Ariza DS, Leon-Ariza JS, Nangiana, et al. Evidences in neurological surgery and a cutting edge classification of the trigeminocardiac reflex: a systematic review. World Neurosurg. 2018;(117):4-10.
- Resuscitation Council (UK). Management of cardiac arrest during neurosurgery in adults. Accessed June 02, 2021 https://www.resus.org.uk/library/publications/publication-management-cardiac-arrest-during
- Tomar GS, Mishra RK, Chaturvedi A. Glossopharyngeal vagal reflex: a matter of concern during neurosurgery. Neurol India. 2018;(66):1822-1824.
- Bilgi K, Kamath S, Sultana N. Bezold Jarisch reflex and acute cardiovascular collapse during craniotomy. Indian J Anaesth. 2017;(61):176-177.
- Kinsella SM, Tuckey JP. Perioperative bradycardia and asystole: relationship to vasovagal syncope and the BezoldJarisch reflex. Br J Anaesth. 2001;(86):859-868.
- Goyal K, Philip FA, Rath GP, et al. Asystole during posterior fossa surgery: report of two cases. Asian J Neurosurg. 2012;(7):87-89.
- Sato K, Shamoto H, Yoshimoto T. Severe bradycardia during epilepsy surgery. J Neurosurg Anesthesiol. 2001;(13):329-332.
- Kim Y, Change CH, Ham SY, et al. A brief cardiac arrest due to saline irrigation during surgery for infratentorial cerebellar tumor. Korean J Anesthesiol. 2013;(65):111-112.
- Chowdhury T, Sokhal N, Prabhakar H. Severe haemodynamic disturbances following normal saline irrigation in cerebropontine tumour surgery. Indian J Anaesth. 2012;(56):312-314.
- Karamchandani K, Chouhan RS, Bithal PK, et al. Sever bradycardia and hypotension after connecting negative pressure to the subgaleal drain during craniotomy closure. Br J Anaesth. 2006;(96):608-610.



