Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Which of these features aid in the diagnosis of cardiac tamponade?
a. Pulsus paradoxus
b. Enlarged heart on chest radiograph
c. Akinesis of left ventricular wall on echocardiography
d. Low amplitude ECG voltage
e. Quiet heart sounds - The following are true regarding pericardial fluid:
a. Its presence always confirms the diagnosis of tamponade
b. The normal volume of pericardial fluid is 15-20ml
c. Echocardiographic estimation of pericardial fluid volume strongly predicts clinical haemodynamic compromise
d. It has an exponential pressure volume relationship
e. Pericardiocentesis is only diagnostic in the management of cardiac tamponade - The following are true regarding the management of cardiac tamponade:
a. Pericardiocentesis is indicated in all forms of cardiac tamponade
b. Treatment involves careful fluid resuscitation and inotropes
c. Echocardiographic evidence of chamber collapse predicts a positive fluid responsiveness
d. Emergency drainage is indicated in tamponade with incipient cardiac arrest
e. Once the pericardial effusion is drained it may be necessary to wean infusions of inotropes and vasopressors
INTRODUCTION
Cardiac tamponade is a life-threatening emergency. Claudius Galen (131-201 AD) first described pericardial effusions in gladiators suffering from stab wounds to the chest and the English physician, Richard Lowe (1669) first described its physiology. It took a further two hundred years for the term “cardiac tamponade” to be coined by the German surgeon Edmund Rose. Today cardiac tamponade is recognised as an essential diagnosis to exclude during cardiac arrest in international advanced life support algorithms. With the growth of acute bedside echocardiography tamponade diagnosis is likely to increase and thus knowledge of its management is a fundamental skill for any acute care physician.
DEFINITIONS
Cardiac tamponade is defined as an accumulation of fluid in the pericardial sac, creating an increased pressure within the pericardial space that impairs the ability of the heart to fill and to pump. As the pump function of the heart becomes impaired then there is a fall in cardiac output and systemic perfusion leading to life-threatening organ dysfunction. Clot or tumour compressing the pericardium may also have the same effect. Importantly the presence of abnormally large volumes of fluid within the pericardial sac does not always necessitate the development of tamponade. This is because slowly accumulating fluid allows physiological compensatory mechanisms to limit the rise in pericardial pressure and therefore avoid cardiac pump failure. Consequently a pericardial effusion should be distinguished from tamponade as an anatomical diagnosis, describing the presence of abnormal fluid within the pericardial sac with no haemodynamic compromise. In contrast cardiac tamponade is a physiological diagnosis in which the pericardial effusion must be accompanied by evidence of obstructive shock.
PATHOPHYSIOLOGY AND ANATOMY OF TAMPONADE
Physiology of the pericardial sac
The pericardial sac consists of 2 layers of tough, fibrous tissues surrounding and protecting the heart. An inner visceral layer is separated from the parietal pericardium by a small amount of lubricating pericardial fluid. Usually a volume of around 15-50mls of pericardial fluid is present, being produced by visceral mesothelium cells and drained from the pericardium by the lymphatic system into the mediastinum and right side of the heart. The pericardial layers envelope the heart, fusing around the points where the great vessels exit into the mediastinum. The visceral pericardium is continuous on the inner side with the epicardium. The pericardial sac layers and fluid function to buffer the heart from external impact, to reduce resistance during motion of heart and to provide a barrier against infection traversing from surrounding structures, particularly the lungs.
Pressure-volume relationship
Fluid within the pericardial space exhibits a pressure-volume relationship, which is varied depending upon factors that influence the ability of the pericardium to compensate for volume changes. When plotted the pressure-volume relationship exhibits 2 distinct phases, as illustrated in Figure 1. If fluid accumulates gradually then during the first phase an increase in volume of the pericardial fluid causes only a small increase in pericardial pressure (Plot A, Figure 1). This is because the normal pericardial membranes can stretch to accommodate a larger volume of fluid. During the second phase the reserve volume, below which pressure changes are minimised by the compliance of the pericardial membranes, is exceeded and pericardial pressure begins to rise more rapidly.
In addition a number of factors influence the rate of pressure increase within the pericardial space. Most important is the duration of time over which fluid accumulates. A rapid increase in fluid volume, typically due to bleeding caused by penetrating thoracic trauma or following injury sustained during cardiac surgery, leaves little time for the pericardial membranes to stretch and the resultant pericardial pressure increase will be rapid (Plot B, Figure 1). In the acute situation as little as 150ml of fluid may result in cardiac tamponade. In marked contrast the pericardial membranes can stretch significantly if the build up of fluid is sufficiently chronic, with the pericardial sac volume reaching 2000ml in extreme cases. It is even possible for the pericardial sac to contain these large volumes without causing significant tamponade, provided that enough stretch of the pericardial membranes can occur.
Compliance of the pericardial membranes can be reduced in rare disease states, such as pericardial mesothelioma or scarred following repeated cardiac surgery. In such instances the capacity of the pericardial sac to compensate for enlarging effusions is diminished. The nature of the fluid forming the pericardial effusion is also important. Unlike transudates blood will clot within the pericardial space, transmitting pressure changes more readily, particular onto the localised areas of the heart underlying the clot and contributing to cardiac chamber collapse. Chronic effusions can also become fibrosed and more viscous, causing greater respective pressure increases.
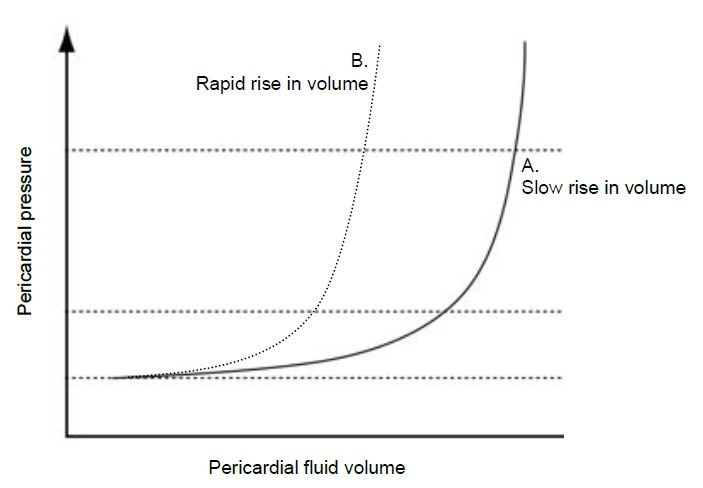
Figure 1. Pericardial fluid pressure-volume relationship (Odor & Bailey, 2013)
Stages of cardiac tamponade
Development of cardiac tamponade can be divided into early and late stages, with corresponding differences in the degree of haemodynamic compromise and the extent to which compensatory mechanisms are able to preserve cardiac output. Tamponade will only be maintained if a sustained increase in pericardial pressure is generated. Requirements for sustained pericardial hypertension are an effusion volume that exceeds the pericardial reserve volume and for the rate of fluid accumulation to exceed the rate of pericardial stretch.
Early stages
During the early stages of cardiac tamponade, fluid fills the pericardial reserve volume and the pericardial pressure begins to rise. Filling pressures within the right side of the heart are lower than within the left side of the heart and are the first to be equaled then exceeded by a rising pericardial pressure. As this occurs the cardiac chamber filling pressure gradient is reduced and there is a massive reduction in venous return to the heart. The right atrium and right ventricle become compressed causing impaired diastolic filling, since right sided pressure are lowest in diastole. As less venous blood returns to the right atrium the central venous pressure rises. Impaired right ventricular filling results in an under-loaded ventricle, which operates at the low end of the Frank-Starling curve. Underfilling and limited contractility result in a low stroke volume.
Late stages
As pericardial pressure rises sufficiently to overcome the left atrial and higher left ventricular filling pressures then a dramatic decrease in cardiac output occurs. Although a reduction in coronary blood flow occurs, in tamponade this is accompanied by limited cardiac work and the myocardium is usually not ischaemic. A cycle of exhausted physiological compensation measures follows, with increased fluid retention, decreased cardiac output and venous return leading to a state of obstructive shock and eventually cardiac arrest.
Physiological compensatory mechanisms
The most important physiological compensatory mechanism is an increase in sympathetic tone. Tachycardia and sympathetic-mediated vasoconstriction cause an increase in systemic vascular resistance in a bid to maintain mean arterial pressure. The renin-angiotension system is activated causing increased fluid retention. Elevated central venous pressures improves diastolic filling against intrapericardial pressure, but this has a limited effect. There is no change in atrial naturetic peptide levels as the heart doesn’t stretch.
CAUSES
Causes of cardiac tamponade can be divided into surgical and medical causes, typically corresponding to acute and chronic onsets respectively. The causes of cardiac tamponade are summarised in Table 1.


Table 1. Causes of cardiac tamponade
CLINICAL FEATURES
Recognition
Cardiac tamponade can present in a variety of forms, from insidious and non-specific to acute and readily apparent. Key to prompt recognition of cardiac tamponade is maintaining a high degree of suspicion in patients presenting with early signs and instigating appropriate investigations. Clinical features of cardiac tamponade vary with duration of onset and underlying cause. The most common signs of dyspnoea, tachycardia and hypotension are poor at discriminating tamponade from other causes of shock. Additional history and examination may indicate an underlying cause, including recent pacemaker lead insertion; chest pain reflecting myocardial infarction or pericarditis; a history of renal failure or tuberculosis leading to ureaemic and inflammatory pericardial effusions respectively.
HIV-related pericardial effusion should be considered if there is a history of intravenous drug abuse or multiple opportunistic infections. Recent cardiac surgery, coronary intervention or thoracic trauma are all important causes of tamponade. History of these events in conjunction with clinical signs mandates urgent investigation. The following clinical signs are particularly important in diagnosis of cardiac tamponade.
Beck’s Triad
Claude Beck, who later became professor of cardiovascular surgery at Case Western Reserve University in USA, first documented the three classical signs of acute cardiac tamponade in 1935. His triad consists of hypotension, elevated jugular venous pressure and muffled heart sounds. Although pathognomonic, these signs are only collectively present in a small number of patients presenting with cardiac tamponade. Moreover the triad is more relevant in surgical patients developing rapid onset tamponade in rather than the slower onset found in most medical causes of tamponade, in whom the findings of Beck’s triad may not be present at all.
Pulsus paradoxus
A more common finding aiding diagnosis of cardiac tamponade is the phenomenon of pulsus paradoxus. First described in 1873 by the German physician Adolf Kussmaul, pulsus paradoxus is an exaggerated fall in systemic arterial blood pressure during the inspiratory phase of spontaneous ventilation. This phenomenon occurs due to negative intrapleural pressures increasing venous return to the right side of the heart, resulting in a bulging of the interventricular septum towards the left heart chambers. A minor respiratory variation in venous return to the heart occurs in the normal heart, but this effect is exaggerated in cardiac tamponade. The inelastic pericardium in tamponade constrains ventricular movement and causes more dramatic bulging of the right ventricle towards the left. The dysfunction of the left ventricle secondary to disorder of the right is termed ventricular interdependence.
Pulsus paradoxus of >10mmHg between inspiratory and expiratory systolic blood pressure in a spontaneously ventilating patient has a high sensitivity for detection of cardiac tamponade. The extent of the ventilatory pressure variation may also help predict the degree of cardiovascular compromise in a patient. Note that in a mechanically ventilated patient intrapleural pressures become positive and the pattern of pulsus paradoxus is reversed. i.e systolic blood pressure is higher during inspiration and lower during expiration.
Reverse pulsus paradoxus is observed to a certain degree in all sedated and mechanically ventilation patients with cyclical variation in pulse pressure used as an indicator of the fluid responsiveness of a patient. Right ventricular afterload increases during positive pressure inspiration because the increase in alveolar pressure is greater than the increase in pleural pressure, hence the pulmonary capillaries are compressed and right ventricular ejection is impeded. However left ventricular preload increases during mechanical inspiration, since pulmonary blood is squeezed towards the left side of the heart. Increased left ventricular preload combined with augmented systolic emptying due to the inspiratory positive pressure results in an increased left ventricular stroke volume and systolic blood pressure during inspiration. The long pulmonary transit time of blood (approximately 2 seconds) means that the decrease in right ventricular output only causes a fall in blood pressure a few seconds later, during the expiratory phase.
Cardiovascular compromise can be worsened by mechanical ventilation in cardiac tamponade, particularly by high levels of positive end expiratory pressure causing persistent impaired venous return to the right side of the heart. Also doppler studies of respiratory alterations in cardiac blood flow can be inaccurate when diagnosing cardiac tamponade in a mechanically ventilated patient.
Absence of pulsus paradoxus should not be used to exclude tamponade as a diagnosis. Nor is tamponade the only cause for exaggerated pulsus paradoxus. Severe asthma, pulmonary embolism and tension pneumothorax can all worsen pulsus paradoxus. In patients with aortic regurgitation the left ventricle can be filled from the aorta during inspiration. Therefore if an aortic dissection produces both aortic regurgitation and cardiac tamponade, pulsus paradoxus may be abolished. Likewise shunting in atrial septal defects may balance changes in venous return during inspiration with similar effects. More commonly physiological compensatory mechanisms can act to maintain normal systemic blood pressure during the respiratory cycle, illustrating that it is important to consider a multitude of trends and clinical features whilst diagnosing cardiac tamponade.
INVESTIGATIONS
Investigations of cardiac tamponade are largely non-invasive and can be performed at the bedside.
Simple investigations
Features of tamponade present on chest radiograph and ECG are relatively non-specific and do not distinguish between pericardial effusion and tamponade. Fluid in the pericardial sac can reduce electrical conduction through the space resulting in smaller QRS complexes on ECG. Cyclical variations in QRS amplitude is more specific to tamponade and is known as electrical alternans. Sinus tachycardia is the most common rhythm in tamponade but atrial arrhythmias provoked by changes in atrial pressure may also be detected.
Chest radiograph may reveal an enlarged cardiac silhouette with a globular heart shape, reflecting expansion of the pericardial sac with surrounding fluid. This appearance is more common in chronic causes of tamponade, in which fluid accumulates gradually. Acute tamponade may occur without any change in cardiac size on chest radiograph.
Echocardiography
Echocardiography is the single most important investigation for cardiac tamponade. A continuous pericardial effusion produces a classical pattern, recognised as a “swinging heart”. The heart oscillates from side to side within the pericardium, most impressively seen with large effusions, also changing the anatomical relationship between the heart and ECG electrodes with a resultant effect on the QRS complex (the cause of electrical alternans).
Pericardial fluid can be visualised in a number of views, including parasternal long axis, parasternal short axis, apical 4-chamber and subcostal. The European Society of Cardiology (ESC) recommends the searching of the following features to aid in the diagnosis of tamponade:
- Diastolic collapse of the anterior right ventriclar free wall, right atrium, left atrium. Very rarely left ventricular collapse may also be seen.
- Increased left ventricular diastolic wall thickness, described as “pseudohypertrophy”.
- Inferior vena cava dilatation, without respiratory variation in size, suggesting raised right atrial pressure.
The ESC also recommends the use of M-mode echocardiography (motion-mode, in which a vertical axis from the ultrasound probe is graphically plotted against time on the x-axis, allowed representation of moving structures during the cardiac cycle) to identify large respiratory fluctuations in mitral and tricuspid flows.
Poor haemodynamic tolerance to an effusion are suggested by an inspiratory rise of greater than 40- 50% in right heart flows, or a decrease of more than 25-40% in left heart flows with an inverted E/A ratio across the mitral and tricuspid valves. The E/A ratio is the ratio of early diastolic ventricular filling velocity to the higher late diastolic blood velocity caused by atrial contraction. The quantity of effusion present can be estimated by using the plarimetry function (area estimation) commonly found on most echocardiograph machines but is not necessarily a predictor of haemodynamic compromise.
Transthoracic echocardiography (TTE) demonstrates tamponade by right atrial collapse with a sensitivity of 50-100% and specificity 33-100%. Right ventricular collapse has a sensitivity from 48- 100% whereas its specificity ranges from 72-100%. Conversely post cardiac surgery TTE is more limited with a negative predictive value of 41%. Transoesophageal imaging provides better views of the posterior aspects of the pericardium and therefore essential in the diagnosis of a retroatrial haematoma, a possible complication following cardiac surgery.
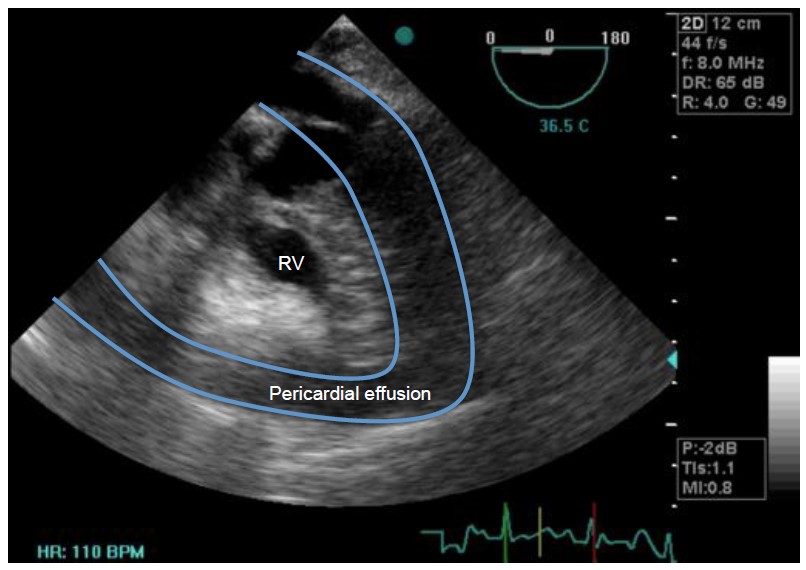
Figure 2. Transoesophageal echocardiographic image showing right ventricular collapse secondary to a pericardial effusion. This image demonstrates evidence of cardiac tamponade. (Odor & Bailey, 2013)
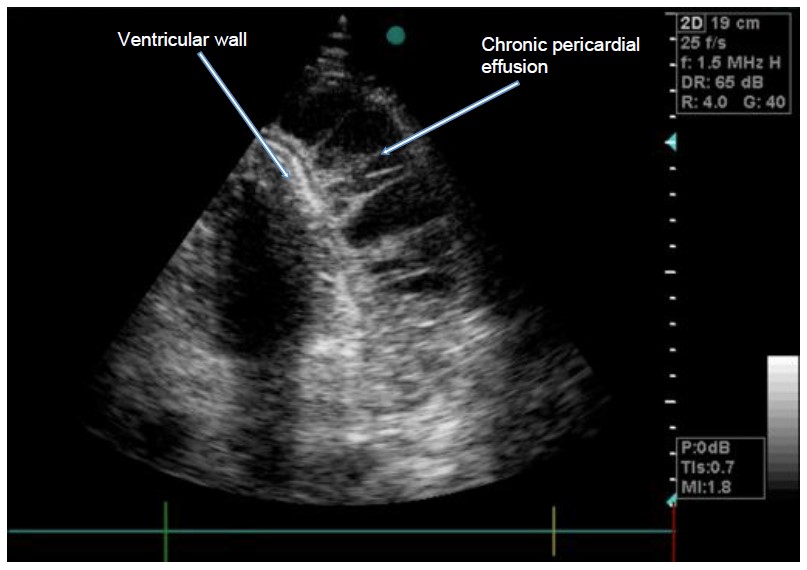
Figure 3. Transoesophageal echocardiographic image showing fibrous strands within a
chronic pericardial effusion. (Odor & Bailey, 2013)
DIFFERENTIAL DIAGNOSIS
Cardiac tamponade should be distinguished from other causes of low cardiac output failure, such as cardiogenic shock, in which primary myocardial dysfunction means the heart is unable to produce an adequate cardiac output to maintain systemic perfusion. The most common cause of cardiogenic shock is massive myocardial infarction.
Constrictive pericarditis caused by thickened pericardium impairing ventricular diastolic function may mimic tamponade in the absence of an effusion. Distinguishing between tamponade and constrictive pericarditis can be difficult, but the underlying pathological effects on cardiac filling are distinct. In tamponade the pericardial pressure is increased throughout the cardiac cycle. In contrast the myocardium is not compressed in constrictive pericarditis until the heart expands to fill the pericardium during mid to late diastole. Therefore the normal bimodal pattern of venous return to the heart is maintained in constrictive pericarditis, since there is no compression during systole, whereas venous return becomes unimodal and confined to systole in tamponade. Skilled echocardiography can help distinguish these different presentations.
Other differentials include large pulmonary emboli and tension pneumothorax. Rarely, a tension pneumopericardium can simulate acute cardiac tamponade, but with a characteristic mill-wheel murmur. This condition can be seen after penetrating chest wall injury, ruptured oesophagus and bronchopericardial fistula.
MANAGEMENT
Management of pericardial effusion can be divided into 2 groups: the pre-tamponade patients who are haemodynamically stable, and those with tamponade who are not stable. Unstable patients necessitate urgent intervention. Since pressure caused by fluid within the pericardial sac is the underlying problem, definitive treatment is drainage of the pericardial fluid thereby alleviating myocardial compromise. A number of approaches are available to drain pericardial fluid. In a life-threatening situation the swiftest and safest approach should be selected. Assistance from cardiothoracic surgical teams should be sought, but may not always be available; hence tamponade drainage should be within the capabilities of those in all acute medical care specialities.
Haemodynamic goals pre-drainage
Resuscitation of tamponade patients prior to pericardial fluid drainage should follow the basic principles of airway, breathing and circulation, taking into account the specific pathophysiology of cardiac tamponade. Oxygen should be administered to the patient. Intubation and mechanical ventilation should be avoided unless strictly necessary, as this will tend to exacerbate cardiac failure in tamponade. Ventilated patients developing tamponade in an intensive care environment should have positive end expiratory pressure minimised to avoid limiting venous return. Invasive haemodynamic monitoring should be instigated, allowing continuous blood pressure and central venous pressure measurements to be made.
Role of fluids
Successful volume expansion primarily depends on the outcome measures defining it (i.e. cardiac index, end organ perfusion or patient symptom relief), the type of tamponade and the overall fluid status of the patient. The effects of hypovolaemia are particularly pronounced in tamponade. A single fluid challenge is likely to be beneficial especially in the setting of hypotension (<100mmHg). Excess fluid administration risks worsening ventricular interdependence on the patient and decreasing their cardiac output. The use of fluid as a bridging management is important in those with a poor preload and a single fluid challenge is unlikely to cause harm. Subsequent fluid bolus need to be carefully assessed with the knowledge that they are unlikely to be of benefit.
Role of inotropes/vasopressors
The haemodynamic goals in tamponade are to increase cardiac output by augmented chronotropy, to decrease afterload and decrease right atrial pressures. Isoprenaline, dopamine and dobutamine are all appropriate first choice inotropes and have been shown to increase cardiac output in tamponade. They all increase the metabolic requirements of the myocardium and decrease its perfusion time and therefore risk myocardial ischaemia. Small studies have shown vasopressors such as noradrenaline improve mean arterial pressure with minimal myocardial strain and no change in cardiac index. The clinician’s choice of inotrope and vasopressor may therefore be best guided by their drug familiarity, and the balance of risks versus benefits for each drug.
Drainage
Definitive treatment of tamponade is only achieved through drainage of the pericardial sac. Techniques for drainage can be classified as pericardiocentesis and surgical. Rapid resolution of the low cardiac output state occurs following release of pericardial pressure and as such the anaesthetist caring for the patient should be prepared to quickly titrate down inotropes or vasopressors to avoid excessive blood pressures which can worsen any bleeding.
Pericardiocentesis
Pericardiocentesis involves placement of a catheter percutaneously into the pericardial sac in order to externally drain the effusion. The procedure can be performed using anatomical landmarks alone, or under echocardiographic or fluoroscopic guidance. Pericardiocentesis is contraindicated in aortic dissection and relatively contraindicated in severe coagulopathy. A Seldinger technique is normally employed with a needle inserted between the xiphoid and left costal margin, angled at a 15-degree angle under the costal margin then slowly advanced towards the tip of the left scapula. A J-shaped guide can be threaded and a pigtail-catheter railroaded into place if required. ECG monitoring is required as instrumentation of the heart can provoke ectopic beats or ventricular arrhythmias.
Pericardiocentesis has both therapeutic and diagnostic applications. Chronic pericardial effusions can be sampled using this technique to allow histological and biochemical analysis in suspected malignant or inflammatory causes.
Risks of pericardiocentesis are puncture and rupture of the myocardium or coronary vessels. This may cause arrhythmias or myocardial infarction. The stomach, lungs or liver may also be punctured. Infection of the pericardial membranes may occur subsequently. These risks are rare, occurring in less than 5% of cases, and as a potentially life-saving procedure it is important to be familiar with the technique of pericardiocentesis.
Surgical drainage
Emergency sternotomy is indicated in tamponade with incipient cardiac arrest. This may need to be performed in the emergency department or intensive care unit if there is insufficient time or the patient is too unstable for transfer to the operating theatre. Emergency thoracotomy equipment should be available in these locations. A small sub-xiphoid incision is made, which may relieve some of the pericardial pressure prior to direct visualisation and incision of the parietal pericardium. Drainage of fluid and control of bleeding within the pericardium can then occur. Cardiopulmonary bypass equipment and a trained perfusionist should ideally be present.
Other surgical approaches to pericardial drainage are available, but there are no prospective randomised controlled trials comparing outcomes from each. A video-assisted thoracoscopic approach is less invasive, creating a drainage window between pericardium and pleura. This has been reported to have longer operating time and higher peri-operative morbidity. Percutaneous balloon pericardiotomy can be performed under local anaesthesia in the cardiac catheterisation laboratory and involves insertion of a valvuloplasty balloon into the parietal pericardium via a subxiphoid approach under radiological guidance. This latter procedure is more suitable for patients with medical tamponade who are unfit for surgery and may be suffering from malignant effusions.
Post-operative care of these patients is often most suitable in a high-dependency unit or intensive care, as recurrence of tamponade may occur and organ support, including inotropes for the stunned myocardium, may be required.
SUMMARY
- Cardiac tamponade is a life-threatening emergency and an important cause of obstructive shock.
- Tamponade can occur as a complication of a number of medical conditions, as well as in trauma or cardiac surgery patients.
- Prompt echocardiographic confirmation of tamponade should be performed when the diagnosis is suspected.
- Echocardiographic signs of right-sided diastolic chamber collapse and ventricular interdependence are highly suggestive of cardiac tamponade.
- Treatment involves careful fluid resuscitation and inotropes, but this is not a substitute for definitive drainage via either percutaneous or open surgical techniques.
ANSWERS TO QUESTIONS
- TFFTT
A high clinical suspicion is essential, especially in combination with subtle clinical findings such as quiet heart sounds, pulses paradoxus and low voltage electrocardiograms. Chest radiographs are rarely of use in its acute diagnosis. - FTFTF
Fluid within the pericardial space will not cause tamponade if it remains at a low pressure as demonstrated by the pressure volume relationship graph. In health the volume of fluid is approximately 15ml with acute volumes of 150ml impairing cardiac function. Pericardiocentesis is therapeutic as well as diagnostic. - FTFTT
Pericardiocentesis should be used with caution in cardiac tamponade secondary to aortic dissection. Fluids and inotropes act as bridging tools to definitive management and anaethestists must be aware they should be weaned rapidly once the tamponade is relieved.
REFERENCES and FURTHER READING
- Spodick DH. The hairy hearts of hoary heroes from antiquity through the twentieth century. In: Fowler NO (ed) The pericardium in health and disease. Futura Publishing Company, Mount Kisco, NY 1985, pp 1-17
- Rose E. Herztamponade. Dtsch Z Chir 1884; 13: 329-410
- L Bodson, K Bouferrache, A Vieillard-Baron. Cardiac tamponade. Curr opin crit care 2011; 17: 416-424
- W Little, G Freeman. Pericardial disease. Circulation 2006; 113: 1622-1632
- Akkerhuis J.M, Hersbach F.M.R.J. Swinging Heart. N Engl J M 2004; 351:e1
- European Society of Cardiology. Guidelines on the diagnosis and management of pericardial diseases. Eur Heart J 2004; 25: 587-610
- Imren Y, Tasoglu I, Oktar G, Benson A, Naseem T, Cheema F, et al. The Importance of transoesophageal echocardiography in the diagnosis of pericardial tamponade after cardiac surgery. J Card Surg 2008; 23: 450-3!



