Intensive Care Medicine
Before reading the tutorial, please consider the questions below: answers can be found within the text.
- Define preload, contractility and afterload. What is the significance of these variables in the management of a hypotensive patient?
- What is shock and how do you categorise shock?
- A 60 year old man presents to the emergency department, hypotensive tachycardic and tachypnoiec, presumed to be secondary to pneumonia. How would you manage this case?
- How would your management change if this was secondary to a myocardial infarction?
- Describe the management of anaphylaxis. What are the advantages of using adrenaline in anaphylaxis?
- List the relative advantages and disadvantages of using different colloids or crystalloids for the resuscitation of a shocked patient.
Introduction
The aim of cardiovascular resuscitation is to improve tissue perfusion and consequently oxygen delivery in clinical situations where this has been compromised. It is important first to consider the disease process and its effect on cardiac physiology, before then deciding on a treatment strategy. Many conditions will require a specific therapy for the underlying cause; examples are antibiotics for septic shock and acute angioplasty or thrombolysis for cardiogenic shock resulting from myocardial infraction. Meanwhile the cardiovascular system can be supported using either fluid therapy, sympathomimetic drugs or a combination of both. The key to successful outcome in this setting is prompt intervention and frequent reassessment of the patient. This tutorial aims to address each of these factors in turn, using both basic principles and an example case.
Case example
A 68 year old male presents to the emergency department. He had a myocardial infarct 8 years ago, but since only gets angina on significant exertion. He is otherwise fit and well. He has not been well over the last 3 days, with a cough and fever.
Introductory Concepts
Assessment of cardiac function
When considering cardiovascular resuscitation, it is important to understand three haemodynamic variables that influence cardiac performance: preload, afterload and contractility.
Preload represents the length of the cardiac muscle fibres of the ventricular wall at enddiastole (i.e. at maximal filling, just prior to contraction). In practice muscle fibre length (or even ventricular volume) is difficult to measure and end-diastolic pressure is taken as a surrogate measure; this in turn is estimated (rather inaccurately) by central venous pressure or pulmonary artery wedge pressure. The stroke volume is the volume ejected from the ventricle during a single contraction. As the preload (i.e. the venous return to the heart) increases, the length of the cardiac muscle fibres at end-diastole increases. Therefore, the stroke volume and the force of contraction of the ventricle increases (‘moves up the Starling curve’).
Afterload is the ventricular wall stress developed during systole and relates to the mechanical resistance to shortening of the ventricular muscle fibres. In practical terms it gives an indication of how forcefully the ventricle must contract during systole to eject blood and the major contributor to this load is the systemic vascular resistance (SVR).
Contractility refers to the amount of work the heart can generate, at given levels of preload and afterload, and is estimated by the maximum rate at which the ventricle can generate a change of pressure over time. Inotropy is used to explain an increase in the work done by the heart, which is independent of heart rate, preload and afterload
Case example (cont)
You assess your patient
- Airway – he is talking, but confused.
- Breathing – his respiratory rate is 30/minute, SaO2 is 86% on air and you hear coarse crackles in the base and midzone of his left lung.
- Circulation – his pulse rate is 120/minute (sinus rhythm), blood pressure 80/40mmHg.
- He has cool peripheries with a capillary refill time of 4 seconds.
- His temperature is 39°C
What is your differential diagnosis?
What are the priorities for his initial management?
What other investigations will you perform, and what else would be useful to know?
Assessment and monitoring
Shock
Shock is defined as a syndrome in which tissue perfusion is inadequate for its metabolic requirements. Shock can be classified into:
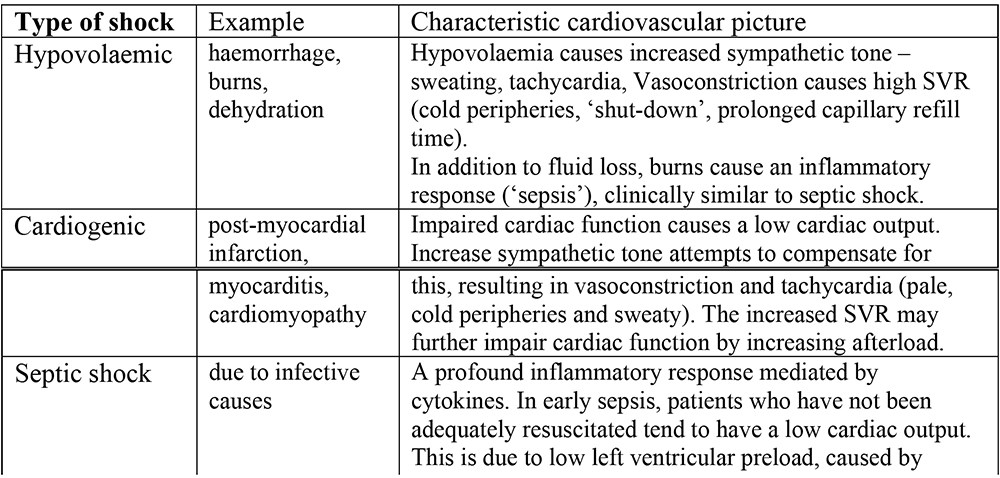
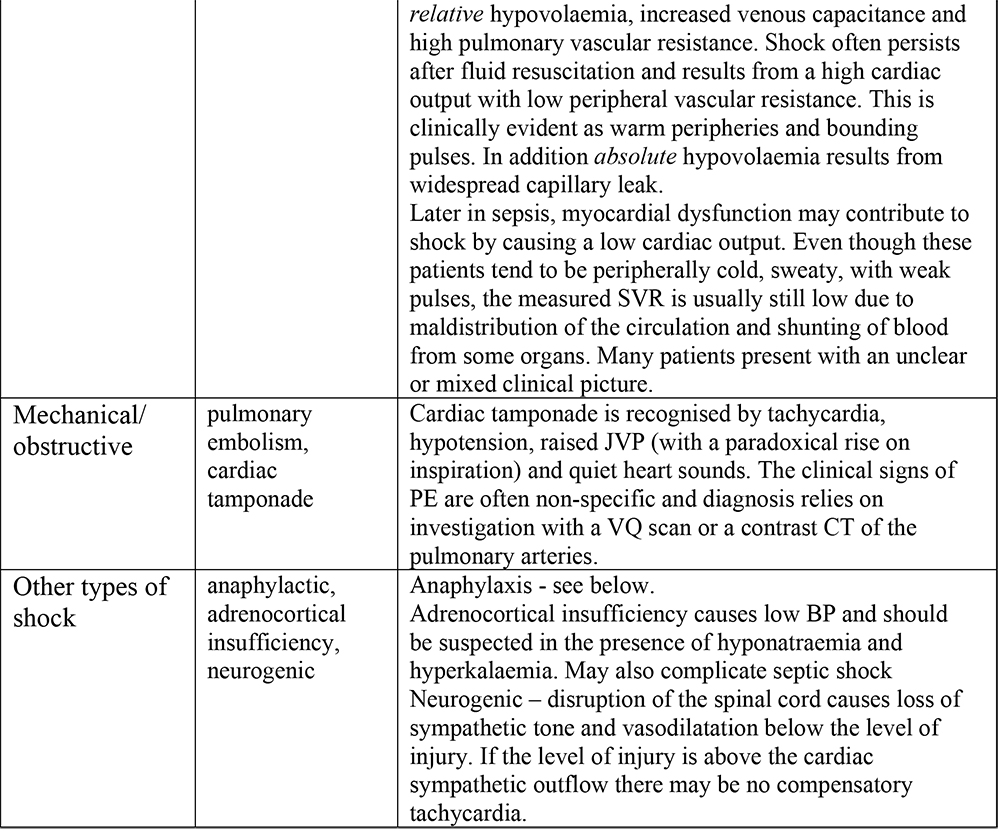
Table 1. Types of shock
Clinical assessment is the most important step in evaluating a patient’s cardiovascular status and subsequent management. When possible, take a full history and then assess the peripheral pulses, heart rate, respiratory rate, blood pressure, capillary refill time (an indicator of peripheral perfusion) and jugular venous pressure.
A patient’s ability to compensate for a haemodynamic insult is highly variable, depending on age, premorbid status and other comorbidities. An example is the rise in the diastolic pressure in early hypovolaemic shock, associated with peripheral vasoconstriction, that is usually only seen in young fit individuals. In addition to these indicators of cardiovascular status, urine output and cognitive function give an estimate of end organ perfusion.
Measurement of lactate and base deficit in arterial blood and, in particular, the trend of these variables over time gives non-specific information about a patient’s organ perfusion. The oxygen saturation in central venous blood (SCVO2) also gives a global indication of haemodynamic status and is useful in directing fluid therapy (see later).
A central venous cannula allows administration of vasoactive drugs and can be used to measure right atrial pressure (RAP). Single measurements are of limited use and the trend of RAP following administration of fluid is more reliable; some clinicians believe that a sustained rise in RAP following a fluid bolus, indicates that the patient’s venous pressure is adequate (i.e they are ‘well-filled’). In mechanically-ventilated patients stroke volume variation, which is the percentage change between the maximal and minimal stroke volumes (arterial swing), has been shown to be a reliable indicator of hypovolaemia.
A number of cardiac function monitors are now available, each of which give an indication of cardiac output, left ventricular filling, and derived variables such as systemic vascular resistance. They all have numerous advantages and drawbacks. Pulmonary artery catheters are rarely used in the UK at present, because of lack of evidence of benefit over the significant risks of harmful side effects. Oesophageal doppler, Lidco/Lidcoplus (a technique combining lithium dilution) and PiCCOplus (Thermodilution Pulse Contour Monitoring) are all minimally invasive techniques and are gaining popularity.
Case example (cont)
The respiratory clinical signs, pyrexia and hypoxia suggest that this patient has pneumonia. In addition he has cardiovascular compromise (shock); this is probably due to sepsis, but he also has a past history of ischaemic heart disease (with myocardial infarction) and his left ventricular function may be impaired. (Note that clinically he has poor peripheral perfusion, rather than the warm peripheries typically seen in early septic shock – however, he has been unwell for three days).
The clinical priorities are to give supplemental oxygen, to begin cardiovascular resuscitation and to investigate and treat the pneumonia.
- Airway/breathing
- Give high flow oxygen (10-15 l/min) through a mask with reservoir bag. Use pulse oximetry to monitor his SaO2. His confusion may be due to hypoxia, so aim to get his SaO2 above 93%.
- Circulation
- Obtain IV access (preferably 18G or larger). If this is difficult, try the long saphenous vein (anterior to the medial malleolus at the ankle) or the femoral vein. Where available, take bloods for FBC, electrolytes, creatinine or urea, glucose, coagulation screen and blood cultures.
- Monitor ECG (he may develop myocardial ischaemia) and blood pressure.
- Give IV antibiotics
- In the UK first line treatment for community acquired pneumonia would be augmentin 1.2g IV 8hourly, with a macrolide (e.g. erythromycin to cover atypical organisms).
- Further investigations and monitoring
- Chest Xray, sputum culture, urinary catheter (hourly output), arterial blood gases (where available).
- Begin fluid resuscitation
What fluid would you, use? How much would you give and how quickly?
Intravenous fluids for resuscitation
Fluid Physiology (see figure 1)
In order to understand where infused fluid goes in the body, it is important to understand the body’s fluid compartments. Water makes up approximately 60% of the total bodyweight in an adult; so a 70kg man contains approximately 42 litres of water (the total body water – TBW). Two thirds of this is intracellular (28 litres) and one third extracellular (14 litres). The extracellular component can then be further subdivided into interstitial fluid (three quarters – approximately 10.5 litres) and intravascular fluid (approximately 3.5 litres).

Figure 1. The body’s fluid compartments: total body water is 42l for a 70kg individual.
Fluid replacement
Overview
Fluid replacement should address daily maintenance requirements and additional losses. Maintenance fluid, for patients who are unable take fluid enterally, should provide at least the minimal requirements of water, sodium and potassium. Remember that water and electrolyte requirements may increase in certain disease processes such as diarrhoea and vomiting.
Types of intravenous fluid
Crystalloid
Crystalloids are relatively small molecules that dissociate into ions to form true solutions, and are therefore capable of passing through a semi-permeable membrane. Commonly used crystalloids include 0.9% saline, dextrose and Hartman’s (Ringer’s lactate) solution. The electrolyte content (mmol/l), pH and osmolarity (mmol/l) these crystalloids are shown in the following table.
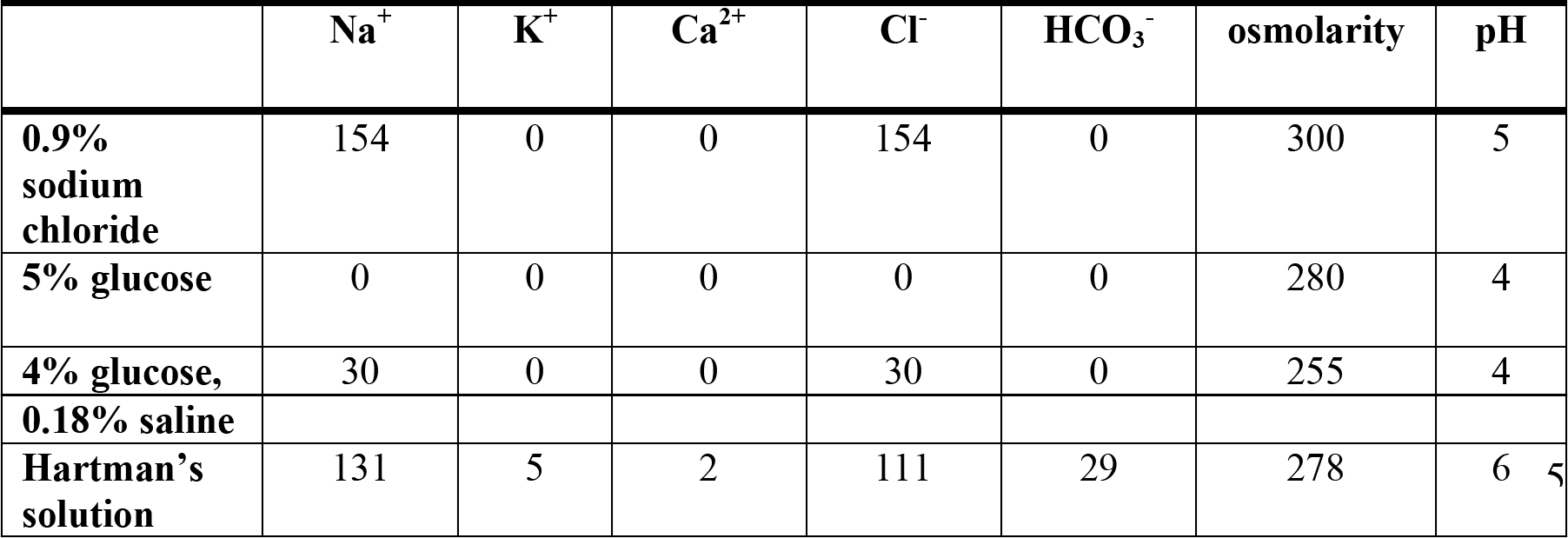
Table 2. Comparison of the constituents of different crystalloids.
On intravenous infusion, 0.9% saline and Hartman’s solution rapidly distribute into the entire extra vascular space (in blue below), leaving 25% of the infused volume in the intravascular a space, i.e. 250ml of a 1000ml fluid bolus.

Figure 2. Distribution of 0.9% saline or Hartman’s solution after infusion.
5% glucose (the optical isomer ‘dextrose’ is now rarely found) loses all of its glucose on first pass through the liver and skeletal muscles. The remaining water is distributed evenly throughout the entire TBW (shown in orange), leaving only 8.3% (one twelfth) of the original volume in the intravascular space (i.e. only 83ml of a 1000ml fluid bolus).

Figure 3. Distribution of 5% glucose after infusion
Colloids
Colloids are larger molecules that are dispersed throughout the solvent, rather than in solutions. They cannot pass through a semi-permeable membranes and consequently tend to remain in the intravascular space.
The electrolyte content (mmol/l), pH and osmolarity (mmol/l) of some commonly used colloids are shown in the table below.
Table 3. Comparison of different colloids.
Gelatins (e.g. gelofusine, haemaccel)
These colloids are polysaccharides, derived from gelatine, which in turn is derived from collagen and have an average molecular weight of 35,000 Daltons. (A Dalton or ‘atomic mass unit’ is a unit of mass equal to 1/12 the mass of carbon 12, which is assigned a mass of 12.). Their half-life in the intravascular space is approximately 3 hours and they are renally excreted. Anaphylactic reactions have been reported with an incidence of 1 in 13,000. Gelatin solutions may interfere with platelet function and coagulation via a reduction in the levels of von Willebrand factor.
Dextrans (e.g. dextran 40, dextran 70)
A dextran is a polysaccharide, derived from sucrose by the action of the bacterium Leuconostoc mesenteriodes. Dextran 40 has a molecular weight of 40,000 Daltons and dextran 70 a molecular weight of 70,000 Daltons.The intravascular half-life increases with molecule size and ranges from 15 minutes to several days. The smaller molecular weight dextrans are predominantly excreted unchanged by the kidney (accounting for up to 70% of Dextran 40), whilst the larger molecular weight dextrans are retained in the circulation for several days. Dextrans have an incidence of anaphylaxis of 1 in 4,500.
Dextrans are rarely used for fluid resuscitation, but they have found a role in thromboembolic prophylaxis via volume expansion, reduction in viscosity and lowering platelet and erythrocyte aggregation. Side-effects include renal failure (due to tubular obstruction), interference with cross matching and coagulopathy.
‘Starches’ (e.g. hydroxyethyl starch (HES), Hetastarch)
These synthetic colloids are of similar structure to glycogen, consisting of chains of glucose molecules (>90% amylopectin). They have molecules with a large range of molecular weights, with the smaller molecules (approximately 50,000 daltons) excreted by the kidney and the larger molecules slowly broken down by alpha-amylase hydrolysis of glycosidic bonds, yielding molecules small enough for renal clearance.
Anaphylactic reactions are rare with an incidence of 1 in 16,000. Slight prolongation of coagulation may occur after large infusions, and pruritis has also been reported.
Total dose should not exceed 20 ml/kg (1500 ml/day for an average male).
Human Albumin Solutions
Human albumin solutions are derived from human plasma by fractionation, and then heat sterilised to reduce the risk of infective transmission. They are presented as either 4.5% (40-50g/l) or 20% (150-200g/l) solutions in 0.9% saline. Traditionally albumin solutions were used as colloid in patients who were hypoalbuminaemic, or where high albumin loss was anticipated (e.g. burns), and in the resuscitation of children.
Choice of resuscitation fluid
Theoretically, resuscitation fluid in acute illness should remain in the intravascular space, and therefore restore circulating volume and cardiac preload. Colloids, by definition, do not cross semi-permeable membranes and so remain in the intravascular space, in theory making them more suitable resuscitation fluids.
A Cochrane database meta-analysis published in 1998 suggested that the use of albumin as a resuscitation fluid was associated with an increased absolute risk of death of 6% (one extra death for every 17 patients given albumin). The SAFE (Saline versus albumin fluid evaluation) study1 aimed to assess the safety of albumin, compared to saline, when used as a resuscitation fluid in a mixed group of 7000 intensive care patients. The study was conducted in Australasia where use of albumin is widespread. No demonstrable mortality difference between the two fluids was shown, the conclusion being that albumin is safe to use.
Conversely there is little evidence to suggest that colloids have any significant mortality benefit over crystalloids when used to a resuscitate hypovolaemic patients. Most clinicians begin with crystalloid (e.g. 1000ml warmed Hartmann’s solution) and alternate with their chosen colloid. Remember that blood is the ultimate colloid, and the ATLS (Advanced Trauma Life Support) guidelines recommend that in trauma victims, initial resuscitation should be with crystalloid followed by blood if indicated by profound anaemia and/or ongoing blood loss.
Case example (cont)
You give 1000ml 0.9% saline over 20 minutes. He is very sick so you stay at his side and then reassess him using ABC when the fluid is in. His SaO2 is 92% on oxygen, but his pulse rate and blood pressure remain the same. His capillary refill time is marginally better at 3.5 seconds. His bladder is empty. You give more fluid – 500ml gelofusine – and then reassess him.
He remains hypotensive at 85/40, with a pulse rate of 120/min. However his fingers are now warming up and better perfused (capillary refill time is 2.5seconds)
Blood gas analysis shows:
pH 7.23, PaO2 9.6kPa, PaCO2 2.9kPa, lactate 6.2, base excess -10, chloride 110mmol/l
What does this blood gas indicate?
What would be an appropriate next step?
Additional considerations for fluid resuscitation
Hyperchloraemic acidosis
One of the end points for resuscitation is an improvement in metabolic function. Resuscitation with large volumes of chloride-rich fluids (e.g. 0.9% Saline) is commonly associated with a hyperchloraemic acidosis. Note also that the pH of 0.9% ‘normal’ saline is 5. The significance of this acidosis in itself may be inconsequential, however a residual base excess of –3 to –6 may trigger unnecessary investigation and treatment. For this reason, many clinicians choose to use Hartman’s solution (with a chloride content of 111mmol/l compared to 154mmol/l) rather than 0.9% saline. Remember that the gelatins are suspended in 0.9% saline and so may cause a similar picture when infused in high volumes.
Case example (cont)
The arterial blood gas shows hypoxaemia (his chest Xray has confirmed left lower lobe pneumonia) and a partially compensated metabolic acidosis with an elevated lactate. This result is consistent with the patient’s clinical picture and suggests that he still has poor tissue perfusion and required further fluid resuscitation.
If the facilities are available it would be reasonable to insert a central venous cannula at this point. You run a venous sample from the CVC through the blood gas machine and the SCVO2 is 57%. A normal value is about 70%, so this provides further evidence that he is still not perfusing his tissues adequately.
Measuring his right atrial pressure via the central line, you give further 250ml aliquots of Hartman’s solution (you don’t want his chloride level to get any higher, so avoid 0.9% saline). From an initial of 8mmHg, the RAP rises to 13mmHg after each of three 250ml infusions of fluid. After the fourth, the RAP remains at 13mmHg and you believe that you have given him sufficient fluid resuscitation.
(Where available, use of a cardiac output monitor would be useful here. Pulmonary artery catheters are not currently popular in the UK, but insertion of an arterial line would allow use of LidCO or PiCCOplus to further assess his intravascular fluid status and his cardiac function.)
Despite this fluid resuscitation he remains hypotensive, is still confused and has only passed 15ml urine in the last 2 hours.
What would be your next strategy in his management?
Sympathomimetic agents
Definitions

General Considerations
Sympathomimetic agents should only be considered when:
- You believe the patient has been optimally resuscitated with fluid.Starting a vasoconstrictor in an unresuscitated, hypovolaemic patient the resulting vasoconstriction will increase the SVR. The blood pressure may rise, but at the cost of reducing cardiac output, tissue perfusion and oxygen delivery.
- You have considered the cause of hypotension.Consider and, if possible, exclude rapidly treatable causes such as tension pneumothorax and cardiac tamponade. Different causes of shock cause hypotension by different mechanisms, requiring different modes of treatment. For example, profound vasodilatation (reduced systemic vascular resistance) is seen in septic, neurogenic and anaphylactic shock and will respond to vasopressors, whereas the reduced myocardial contractility that characterises cardiogenic and late septic shock requires administration of an inotropic agent
Physiology of adrenoceptors
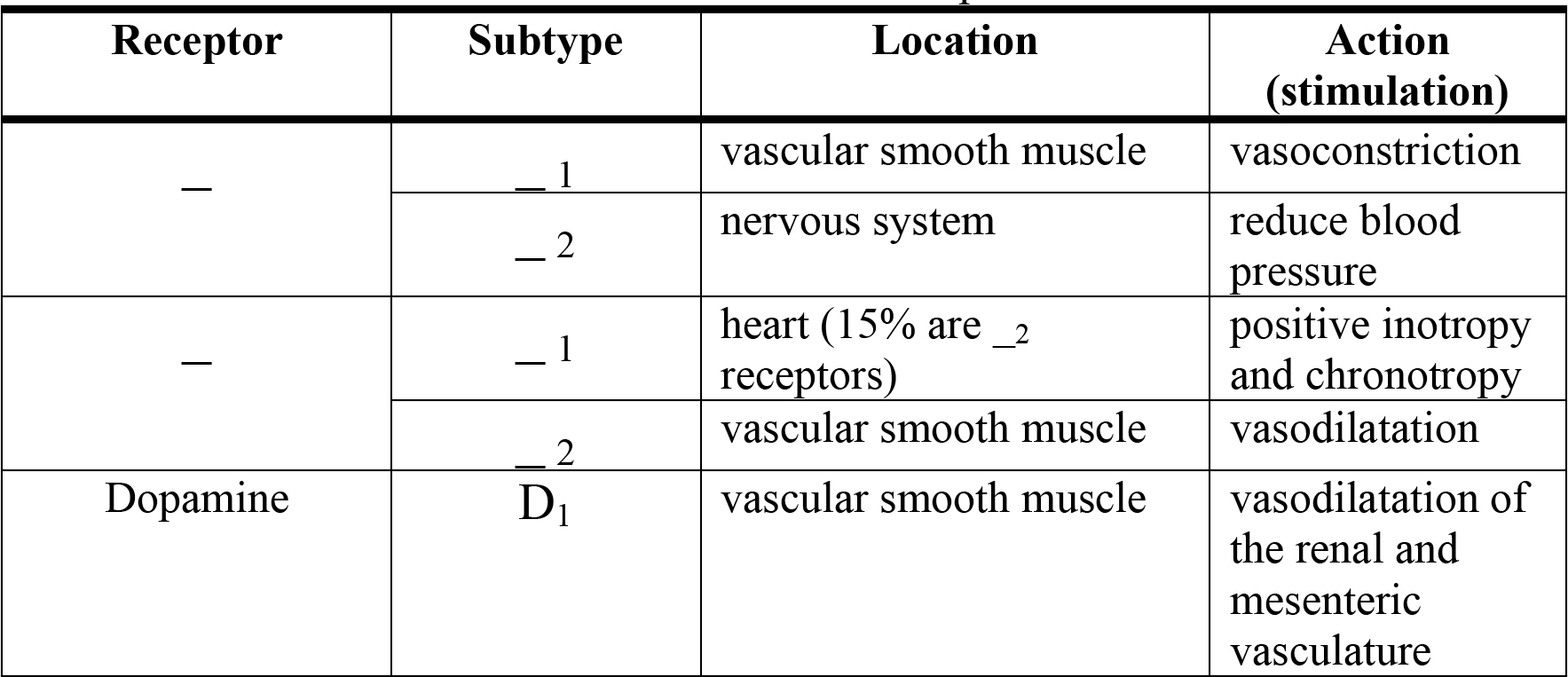
Table 4. The location and effect of different adrenoceptors.
Endogenous ‘naturally-occurring’ sympathomimetics
Adrenaline (Epinephrine)
Adrenaline exerts its effect by stimulation (agonism) of _- and _-adrenoreceptors, in a dose dependent manner. In theory, at low dose _- effects predominate causing increased cardiac output, myocardial oxygen consumption and coronary artery vasodilatation. At high dose it is proposed to have proportionally more _1 effect causing a rise in systemic vascular resistance. In practice these dose-dependent effects are less predictable. Blood glucose levels tend to rise as glucose production is enhanced and insulin release is inhibited. Adrenaline not only decreases hepatosplanchnic blood flow and oxygen exchange, but also compromises hepatoplanchnic lactate clearance. The resulting undesirable hyperlactataemia has limited use of adrenaline in many intensive care units.
Noradrenaline (Norepinephrine)
Noradrenaline acts mainly via _1 receptors, but also has significant _- effects. As a result it increases both systolic and diastolic blood pressure, causing a reflex bradycardia. Cardiac output may fall due to rising SVR and myocardial oxygen demand may increase.
Dopamine
Again the theoretical dose-dependent effects of dopamine on certain receptors are less well defined in practice. At low dose (approximately 2.5 mcg/kg/min) action at dopamine-1 receptors cause mesenteric vessel vasodilatation and a consequent diuresis. At infusions rate of less than 10 mcg/kg/min _1 effects predominate, and at rates of above 10 mcgs/kg/min it has _1 effects are seen.
Traditionally low dose dopamine was used to protect the kidney from impending acute renal failure, the proposed mechanism being improved blood supply and the maintenance of urine flow. However, this diuresis can also be attributable to improved cardiovascular parameters and a proximal tubular inhibition of sodium reabsorption and has been shown not to impact on the development of acute renal failure. Dopamine has the advantage of peripheral administration in adults and remains a first-line vasoactive agent in criticallyill children.

Table 5. Infusion doses and administration of commonly-used sympathomimetics
Exogenous synthetic sympathomimetics
Dobutamine
Dobutamine is synthetic direct-acting catecholamine, which has _1 and to a lesser extent _2 effects. It is a positive inotrope and chronotrope, causing an increase in myocardial oxygen requirement. Although cardiac output rises, blood pressure may fall due to a reduction in systemic vascular resistance (_2 effect). Arrhythmias may be precipitated. Dobutamine can be used for the management of low cardiac output states, but may need to be combined with a vasoconstrictor in the first instance to prevent further hypotension and reduction in diastolic perfusion pressure.
Isoprenaline
Isoprenaline is a synthetic agent with _1 _2 agonist activity. It’s main use is as a positive chronotrope in patients with complete heart block, prior to insertion of pacing wire.
Phosphodiesterase inhibitors (e.g. milrinone, amrinone, enoximone)
_-receptor stimulation activates the enzyme adenylate cyclase, which produces cyclic adenosine monophosphate (cAMP) as an intracellular second messenger. cAMP is inactivated by the enzyme phosphodiesterase, therefore phosphodiesterase inhibitors increases intracellular levels of cAMP, with consequent inotropic effects.
There are 5 cateogories of phosphodiesterase enzyme of which type 3 is specific to the myocardial cell and vascular smooth muscle. Caffeine and aminophylline are nonspecific phosphodiesterase inhibitors, whereas milrinone, enoximone and amrinone are all specific phosphodiesterase-3 inhibitors.
They act as “inodilators” increasing cardiac output and causing peripheral vasodilatation, but have less effect on heart rate than dobutamine. They should be used with caution as their vasodilatory effect can result in significant hypotension and usually require coadministration with noradrenaline. They have a role in patients with poorly compliant ventricles and diastolic dysfunction.
Levosimenden
This calcium channel sensitizer is an effective positive inotrope which has been used for the treatment of cardiac failure. It is unlicensed for use in the UK.
Case example (cont)
His confusion and oliguria suggest that the ongoing hypotension is causing inadequate perfusion of his brain and kidneys (he also has ischaemic heart disease and is at risk of ischaemia). This should be reversible, but needs prompt treatment.
You have corrected the patient’s absolute hypovolaemia (i.e. caused by fluid loss through capillary leak throughout the body). He remains hypotensive since he still has relative hypovolaemia, due to profound vasodilatation caused by the septic inflammatory response (he has a low SVR). He needs a vasopressor to correct this. In the UK, this would be noradrenaline (norepinephrine). Start the infusion at 3-5ml/hr and in a patient who is normally normotensive, a target mean arterial pressure (MAP) of 70-80 mmHg would be reasonable. Patients who are normally hypertensive may require a higher target MAP. Where possible insert an arterial line.
By gradually increasing the noradrenaline infusion (by 1ml/hour, every 5 minutes) you achieve a BP of 115/65 mmHg. He gradually becomes less confused and his urine output starts to improve.
Where available, it would be helpful to monitor his cardiac output, using information from a LidCO or PiCCO system. We’d expect his cardiac output to be high, attempting to compensate for the low SVR:

This man has ischaemic heart disease and so monitoring of cardiac output, would help us to avoid causing excessive vasoconstriction, which would increase the afterload to the heart and impair cardiac function.
What further management would you plan for this man?
Resuscitation Endpoints
Each intervention should be followed by reassessment of the patient, in order to decide whether the intervention has improved their clinical condition and should be repeated or whether different strategy should be employed. Reassessment relies on the same tools as cardiovascular system, markers of end organ perfusion, and measurements of metabolic status.
Evidence suggests that early goal-directed treatment of septic shock, using fluids and inotropes to achieve specific end-points in resuscitation, improves survival.2
Central venous oxygen saturation (SCVO2), measured in a sample from a standard central venous cannula in the superior vena cava, was used as an indicator oxygen delivery. Values of SCVO2 below 70% suggest inadequate delivery of oxygen for the demands of the body’s tissues and fluids, blood transfusion and inotropes were used to optimise this. Mortality in the treatment group was 29%, compared to 44% in the control group (absolute risk reduction of 15%).
Case example (cont)
Further management
- Continuous reassessment of clinical signs and investigations:
- His pulse rate settles to 90/minute, urine output improves, his repeat blood gases are better with a falling lactate and improving base excess.
- He has septic shock (Septic shock describes sepsis with hypotension despite adequate fluid resuscitation). Consider further treatments for this: IV steroids, activated protein C, enteral feeding, and tight control of his blood sugar.
- Review the microbiological findings: Streptococcus pneumoniae grows in his sputum, so you stop the macrolide antibiotic, continuing the augmentin.
Special circumstances
Anaphylaxis
Anaphylactic reactions are precipitated by antigen-mediated mast cell degranulation which results in histamine release. This causes vasodilatation and hypovolaemia (due to capillary leak) and results in shock. Anaphylaxis affects multiple systems (respiratory, airway and skin) and must be managed rapidly and aggressively.
Fluid management and adrenaline form the mainstays of immediate resuscitation. Adrenaline has a number of effects in this circumstance; it has both _ and _ effects increasing cardiac output and systemic vascular resistance, but also causes bronchodilation and decreases the further release of vasodilatory mediators from mast cells and basophils. Accepted management in the UK is that advocated by the Association of Anaesthetists of Great Britain and Ireland.3
Summary
The successful management of a patient with cardiovascular compromise relies on thorough early assessment and intervention, with frequent reassessment after subsequent therapies. Remembered that, while you are focused of maintaining blood pressure, cardiac output and oxygen delivery to the patient’s vital organ, you must also diagnose and treat the underlying cause.
References
- Finfer S et al. SAFE Study Investigators. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. The SAFE Study Investigators. New Engl J Med 2004; 350: 2247-56.
- Rivers E et al. Early Goal-Directed Therapy in the Treatment of Severe Sepsis and Septic Shock. NEJM 2001; 345: 1368-77.
- Available at: aagbi.org/publications/guidelines/docs/anaphylaxis03.pdf



