Basic Sciences
KEY POINTS
- Dexamethasone is a synthetic corticosteroid that acts on the hypothalamic-pituitary-adrenal axis to produce a range of systemic effects.
- Compared with other exogenous steroids, such as hydrocortisone, dexamethasone is relatively potent and long acting, with negligible mineralocorticoid activity.
- Side effects of dexamethasone are the same as other corticosteroids and are more common with prolonged use and higher doses.
- Dexamethasone can be used to supplement those with adrenal insufficiency intraoperatively to prevent an acute adrenal crisis.
INTRODUCTION
There has been a recent increased interest in the glucocorticoid drug dexamethasone with its use in the treatment of COVID-19 patients, in addition to its historical use as an antiemetic in surgical patients. In this article, we explain the pharmacokinetics of the drug as well as its indications and side effects, not only in anaesthesia but also in other aspects of medicine.
PHYSIOLOGY OF CORTICOSTEROIDS
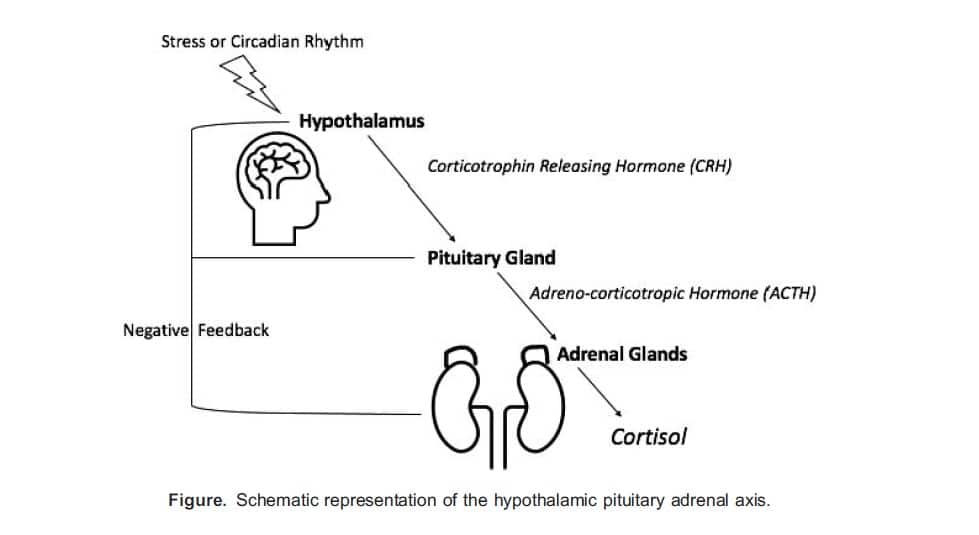
The hypothalamic-pituitary-adrenal axis is a negative-feedback mechanism that controls and regulates the release of cortisol (Figure). Corticotrophin-releasing hormone (CRH) is produced in the hypothalamus. Its release is triggered by stimulation from various centres of the brain in response to physiological and emotional stress or naturally as part of the circadian rhythm. CRH triggers the release of adrenocorticotropic hormone (ACTH) from the anterior pituitary gland. ACTH acts on the adrenal glands to stimulate the release of cortisol from the zona fasciculata. Cortisol will then feedback on the anterior pituitary and hypothalamus to downregulate the release of ACTH and CRH.
Cortisol travels largely bound to cortisol-binding globulin and albumin. Because more than 90% is protein bound, its half-life is relatively long, up to 90 minutes. Cortisol is crucial in maintaining homeostasis through carbohydrate, fat and protein metabolism and has other widespread effects throughout the body (Table 1). Cortisol release is controlled by both a diurnal circadian and ultradian rhythm, with levels highest in the early morning. Normal levels are 20-25 mg/d.
DEXAMETHASONE
Dexamethasone (9a-fluoro-16a-methylprednisolone) is a long-acting synthetic corticosteroid with negligible mineralocorticoid activity. It is characterised by the fluorine atom at C9 on the steroid ring. It is 25 times more potent than hydrocortisone. Because of its intracellular action, the pharmacologic effects may persist even after plasma concentrations drop. It may take up to 8 hours for these effects to manifest, and they may last for more than 36 hours.
Pharmacokinetic Properties
Routes of Administration and Absorption
Dexamethasone is available as an oral preparation with 80% bioavailability. It may also be administered topically, intravenously or intramuscularly. Itis highly protein bound.

Distribution
Dexamethasone rapidly distributes across all tissues. It crosses the placenta and enters breast milk. Repeated, prolonged administration is associated with an increased risk of intrauterine growth restriction.1
Metabolism and Excretion
Dexamethasone, like other corticosteroids, undergoes extensive metabolism in the liver by microsomal enzymes. It is further conjugated to glucuronic acid and sulphates, which are excreted renally as inactive metabolites.
Mechanism of Action
Steroids reduce inflammation and are immunosuppressant. They cross the cell membrane and bind to a glucocorticoid receptor. The complex moves into the cell nucleus, where it will both suppress and stimulate transcription. This leads to a reduction in proinflammatory mediators such as eosinophils, mast cells, macrophages, interleukins and phospholipase A2. There is an upregulation of anti-inflammatory mediators,which ultimately reduces prostaglandin and leukotriene synthesis.
INDICATIONS: ANAESTHTIC USES OF DEXAMETHASONE
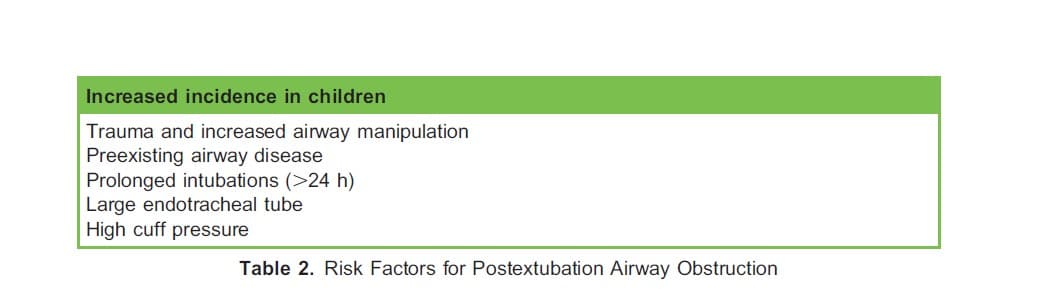
Postextubation Airway Obstruction
Endotracheal intubation can cause laryngeal oedema and mucosal ulceration as a result of local pressure and ischaemia. Postextubation airway obstruction (PEAO) is a serious complication with an incidence of 6-37%, with up to 17% of patients requiring reintubation.2 It often presents as stridor following extubation. There are several risk factors for PEAO, as highlighted in Table 2. When compared with a placebo, 4 doses of dexamethasone 5 mg intravenously (IV) every 6 hours prior to extubation has been shown to increase cuff leak volumes and reduce the incidence of stridor and reintubation without adverse features.2 Other steroids, such as methylprednisolone, have shown a similar effect.
Postoperative Nausea and Vomiting
Postoperative nausea and vomiting (PONV) is a common anaesthetic side effect that affects one-third of patients. It may delay recovery, prolonghospital stay and lead to serious consequences such as aspiration pneumonia and wound dehiscence. The use of intraoperative dexamethasone is well established in reducing the incidence and severity of PONV.3–5 Recommended doses are 4-8 mg in adults and 0.15 mg/kg in children. Evidence shows that it is as effective as ondansetron and marginally better than cyclizine and droperidol.3,4
The mechanism behind this effect is poorly understood but is likely multifactorial. It is postulated that the effect is due to a combination ofreduced inflammatory and emetogenic mediator production such as prostaglandins, gut serotonin and neural 5-HT. There is a potential mild analgesic effect that may be opioid sparing, but the clinical significance of this remains questionable.
There have been case reports of awake patients reporting unpleasant and unusual perineal sensations when given IV dexamethasone. It is therefore common practice to administer it after induction of general anaesthesia.5
Perineural Adjuncts
Dexamethasone has been shown to reduce the onset time and increase the duration of analgesia when administered alongside a local anaesthetic during regional anaesthesia.6 This may be a result of reduced regional blood flow and inflammation, thus slowing local anaesthetic absorption. Membrane excitability of nociceptor C-fibres may be reduced by dexamethasone.
When comparing IV and perineural dexamethasone, analgesia was prolonged in the perineural group as well as sensory and motor block. However, postoperative opiate requirements were unchanged.6 No optimum dose has been elucidated. The risk of neural toxicity remains unclear, and therefore, its use requires further research.6
INDICATIONS: NONANAESTHETIC USES SARS-CoV-2: ‘COVID-19’
SARS-CoV-2 can progress to COVID-19 pneumonia. The mortality rate for those admitted to hospital is 26%, which increases to 37% if invasive ventilation is required.7 The pathological process is immune mediated and characterised by alveolar damage and inflammatory infiltrates. SARS-CoV-2 inhibits the histone deacetylase (HDAC2) transport into cells’ nuclei. HDAC2 mediates the cells’ release of inflammatory cytokines. Dexamethasone activates HDAC2 and directly counters this action.
The RECOVERY trial looked at whether dexamethasone would reduce 28-day mortality by virtue of its anti-inflammatory activity. The study concluded that a 10-day course of 6 mg daily provides a significant survival benefit in patients requiring respiratory support. The benefit was greatest in those requiring invasive mechanical ventilation, as the age-adjusted absolute 28-day mortality was reduced by 12.3%.7 There was amore modest effect in those only requiring oxygen supplementation, with a reduction of 4.2%.7
In addition, dexamethasone shortened hospital stays (median 12- vs 13-day duration) and reduced the need for invasive ventilation, particularly in those requiring oxygen therapy.7 Patients symptomatic for longer than 7 days prior to receiving dexamethasone appeared tobenefit most. However, there is potential for harm in patients not requiring oxygen therapy.
Together, these findings may reflect the pathophysiology of COVID-19 pneumonia whereby viral replication is highest early in the disease before transitioning to a phase of immunological dysfunction. Therefore, dexamethasone may augment viral replication if given too early but combat the proinflammatory response in severe COVID-19 pneumonia.7
Acute Laryngotracheobronchitis: ‘Croup’
Croup is a common viral respiratory illness, affecting children aged 6 months to 3 years in the colder seasons. Typically, it does not require hospital admission, and symptoms resolve within 48 hours; however, upper airway obstruction and respiratory arrest can occur.8
Dexamethasone at a dose of 150 lg/kg orally, intramuscularly or IV has been shown to reduce hospital stay and readmission rates. There is no difference in efficacy between routes of administration.9 The clinical effect becomes apparent 6 hours after administration. Nebulised budesonide 2 mg is an alternative, as both have been shown to reduce hospital stay and readmission rates.
It is crucial to exclude alternative causes of upper airway obstruction such as epiglottitis, uvulitis, severe tonsilitis, bacterial tracheitis and foreign-body inhalation. The use of dexamethasone is not indicated in any of these conditions.8
Bacterial Meningitis
Antibiotic therapy causes bacterial cell lysis within the cerebrospinal fluid, and the subsequent inflammatory response causes disruption to the affected neural tissue. Dexamethasone diminishes this response. The first dose of dexamethasone should be administered prior to the first dose of antibiotics; however, it is acceptable to give it within 4 hours. There is no benefit in starting dexamethasone more than 12 hours after the first dose of antibiotic. Dexamethasone therapy should be stopped if meningococcal septicaemia develops.10
High-dose dexamethasone (0.15 mg/kg in children, 10 mg in adults, 4 times per day) has been shown to improve outcomes and reduce mortality by 10%10 in patients with Streptococcus pneumoniae. There is no outcome benefit with the use of dexamethasone if Neisseria meningitidis is the causative organism.10
Oncology
Dexamethasone is used extensively within oncology and palliative care, as it combats many symptoms of terminal illness and cancer (Table3).11
Concerns have been raised about the potential for dexamethasone to hinder apoptosis of cancer cells by dampening the immune response and thereby increasing the risk of tumour recurrence. However, clinical evidence supporting this theory is conflicting and limited in scope.12,13As present, there are no consensus recommendations regarding the use and safety of perioperative dexamethasone in cancer patients.
Tumour Lysis Syndrome
Case reports have implicated dexamethasone as a causative agent for tumour lysis syndrome (TLS), resulting in the death of 2 people.14,15 This oncological emergency is characterised by a significant release of harmful substances, such as potassium, phosphate and uric acid, fromrapid cancer cell lysis. This may result in severe metabolic disturbances, most notably

hyperkalaemia and hypocalcaemia and the associated clinical sequelae such as dysrhythmias, vomiting, tetany, syncope and death. TLS is more commonly associated with cytotoxic agents and with those suffering from high-grade lymphomas and acute lymphoblastic leukaemia. Dexamethasone-induced TLS appears to be a rare phenomenon, but its use in those with suspected or known, high-risk cancers should be carefully considered.
CAUTIONS AND ADVERSE EFFECTS
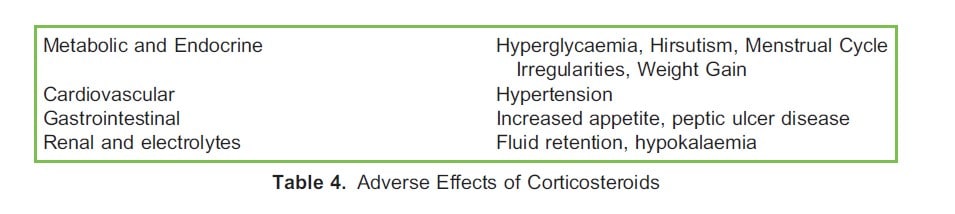
The adverse effects of dexamethasone are common to all corticosteroids (Table 4).1 These become increasingly apparent with higher doses and prolonged courses.
Because of the potential life-saving nature of corticosteroids, there are no absolute contraindications. However, given their side effect profile, it is important to be cautious in the following scenarios:
Psychiatric Reactions
Dexamethasone is linked to neuropsychiatric side effects including euphoria, hypomania, mania, psychosis, cognitive impairment or depression. While the pathophysiology is thought to be multifactorial, it is hypothesised that it is a result of increased dopamine levels.5 There is a 28% incidence of mild-moderate reactions and 5-10% incidence of severe reactions.16 Higher doses increase the incidence but have no relation to onset, severity or duration of symptoms.16 No other risk factors have been identified. Most cases are reported to start within the first 2 weeks of initiating treatment, and symptoms generally resolve on reducing or stopping treatment.16 A pre-existing psychiatric disorder does not increase the risk of an acute reaction, but this cohort of patients should be appropriately counselled.
Vaccines
The administration of live vaccines is contraindicated for patients receiving immunosuppressive doses, as the expected antibody response may not be stimulated.1 This would result in a lack of immunisation and potentially a severe reaction to the live organism.
Infections
Increased susceptibility and severity of acute infections is associated with steroid immunosuppression.1 For example, chickenpox or measles may present with fulminant disease. In addition, fungal infections may be aggravated and latent tuberculosis reactivated.1 A single dose of intraoperative dexamethasone has not been found to significantly increase postoperative infection.3
Hyperglycaemia
Perioperative hyperglycaemia, from any cause, is associated with increased morbidity and mortality. This has justifiably raised concerns about the use of intraoperative dexamethasone, especially in those with pre-existing diabetes mellitus.
However, a recent, large study has shown that whilst postoperative hyperglycaemia is more likely in patients who receive a single intraoperative dose, this resolves within 24 hours.17 Furthermore, a single dose of dexamethasone has been shown to shorten the length of hospital stay and improve 90-day mortality in both patients with diabetes and those without diabetics.17 Another large noninferiority trial found that a single dose of intraoperative dexamethasone was noninferior to a placebo group for surgical site infections within 30 days of nonurgent and noncardiac operations.18 Closer postoperative blood glucose monitoring is generally recommended in those who have received dexamethasone, especially if they have preexisting diabetes.17
Intraoperative and Postoperative Steroid Replacement
Surgical stress and illness may precipitate an acute adrenal crisis in those with adrenal insufficiency. Primary adrenal insufficiency is a dysfunction of the adrenal glands, and secondary insufficiency is a dysfunction of the central release of ACTH or CRH. The most common cause of secondary adrenal insufficiency is exogenous steroid use. Patients with primary adrenal insufficiency require both glucocorticoid and mineralocorticoid replacement during the perioperative period, hence why hydrocortisone is suitable whereas dexamethasone, with negligible mineral ocorticoid activity, is not. However, dexamethasone can be used for intraoperative replacement for patients is taking 5 mg or greater ofprednisolone.19
CONCLUSION
Dexamethasone is a versatile drug with indications across many medical specialties. Recently, it has risen to prominence with evidence of its effectiveness in treating severe cases of COVID-19 pneumonia. Whilst generally considered a safe drug, with widespread use, care should betaken in certain patient groups to avoid potential adverse reactions.
REFERENCES
1. British National Formulary. https://bnf.nice.org.uk/drug/dexamethasone.html
2. Lee CH, Peng MJ, Wu CL. Dexamethasone to prevent postextubation airway obstruction in adults: a prospective, randomized, double-blind, placebo-controlled study. Crit Care. 2007;11(4):R72.
3. Kakodkar PS. Routine use of dexamethasone for postoperative nausea and vomiting: the case for. Anaesthesia. 2013;68(9):889-891.
4. Gan Tong J et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Anal. 2014;118(1):85-113.
5. Bartlett R, Hartle AJ. Routine use of dexamethasone for postoperative nausea and vomiting: the case against. Anaesthesia. 2013;68(9):892-896.
6. Heesen M, Klimek M, Imberger G, Hoeks SE, Rossaint R, Straube S. Co-administration of dexamethasone with peripheral nerve block: intravenous vs perineural application: systematic review, meta-analysis, meta-regression and trial-sequential analysis. Br J Anaesth. 2018;120(2):12e227.
7. The RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693-704.
8. Davies I, Jenkins I. Paediatric airway infections. BJA Educ. 2017;17(10):341-345.
9. Johnson DW. Croup. BMJ Clin Evid. 2014;2014:0321.
10. De Gans J, Van de Beek D. Dexamethasone in adults with bacterial meningitis. N Engl J Med. 2002;347(20):1549-1556.
11. Palliative drugs. https://www.palliativedrugs.com/download/090331_GUIDELINES_FOR_USE_OF_STEROIDS_IN_CANCER_PATIENTS.pdf
12. Kim MH, Kim DW, Park S, et al. Single dose of dexamethasone is not associated with postoperative recurrence and mortality in breast cancer patients: a propensity-matched cohort study. BMC Cancer. 2018;19(1):251. https://doi.org/10. 1186/s12885-019-5451-5
13. Singh PP, Lemanu DP, Taylor MHG, Hill AG. Association between preoperative glucocorticoids and long-term survival and cancer recurrence after colectomy: follow-up analysis of a previous randomized controlled trial. Br J Anaesth. 2014;113(suppl 1):i68-i73.
14. McDonnell C, Barlow R, Campisi P, Grant R, Malkin D. Fatal peri-operative acute tumour lysis syndrome precipitated by dexamethasone. Anaesthesia. 2008;63(6):652-655.
15. Malik IA, Abubakar S, Alam F, Khan A. Dexamethasone-induced tumor lysis syndrome in high-grade non-Hodgkin’s lymphoma. South Med J. 1994;87:409.
16. Kenna HA, Poon AW, de Los Angeles CP, Koran LM. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
17. Herbst R, Telford OT, Hunting J, et al. The effects of perioperative dexamethasone on glycaemic control and postoperative outcomes. Endocr Pract. 2020;26(2:218-225.
18. Corcoran et al. Dexamethasone and surgical-site infection. N Engl J Med. 2021;384:1731-1741.



