Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article.
- Which of the following statements is correct?
- Hepatorenal syndrome arises only in the context of cirrhosis
- It is associated with splanchnic vasoconstriction
- Cardiac output is always high or high normal
- Urine chemistry is always able to distinguish from acute tubular necrosis (ATN)
- Concerning Type 1 HRS:
- It confers a poorer prognosis after liver transplantation
- The kidneys of an affected patient are unsuitable for transplantation
- A liver and renal transplant may be necessary
- Untreated has a median survival of 3 months
- TIPSS
- Is a treatment for refractory ascites
- Is a treatment for variceal bleeding after unsuccessful band ligation
- Is a treatment for HRS
- May prevent HRS in vulnerable patients
- Terlipressin in HRS
- Is a vasopressin V1 agonist
- Must be given by infusion
- Treats HRS by increasing renal vasoconstriction
- Is more effective than noradrenaline
INTRODUCTION
Renal failure (RF) is common in cirrhotic patients. Intensive care units frequently encounter patients with cirrhosis referred for organ support. Categorisation of renal failure, although sometimes difficult, is important for therapeutic and prognostic reasons and to support decision making.
CAUSES OF RENAL FAILURE IN CIRRHOTIC PATIENTS
Common causes in cirrhotic patients are acute tubular necrosis (ATN), pre-renal failure and hepatorenal syndrome (HRS).
ATN
Acute tubular necrosis is common in patients with sepsis. Clues are found in the history and examination. Urinalysis may show a concentrated urine with urine Na+ <10mmol/L or FeNa <1% and the urinary sediment may show granular and epithelial cell casts.
Pre-renal
Variceal or other gastrointestinal (GI) tract bleeding, sepsis and diuretic treatment for ascites predispose to hypovolaemia and prerenal failure.
Hepatorenal syndrome
HRS is the third commonest cause of renal failure in cirrhosis and is discussed in detail below.
Drugs
Aminoglycosides, NSAIDS, diuretics, ACE-Inhibitors and other antihypertensives may contribute to renal failure especially in the context of sepsis and hypotension.
Glomerulonephritis/vasculitis
Glomerulonephritis, IgA nephropathy and vasculitis can be associated with hepatitis B or C, primary biliary cirrhosis, sclerosing cholangitis and alpha-1 antitrypsin deficiency. These conditions should be diagnosed by renal biopsy when there is an index of suspicion, often with an active urinary sediment.
Post-renal failure
This is a very rare cause of renal failure in cirrhosis and should be picked up on ultrasound scanning.
HEPATORENAL SYNDROME
This is a discrete entity which is essentially a diagnosis of exclusion in cirrhotic patients. It is defined as the occurrence of renal failure in a patient with cirrhosis in the absence of another identifiable cause.
Diagnosis
In the absence of recognised markers, diagnostic criteria for HRS were first defined in 1994 by expert consensus (the International Ascites Club). These comprised:

Table 1: original criteria for defining HRS
The criteria were refined in 2007 as follows in recognition that urine chemistry may be deceptive in HRS.

Table 2: 2007 diagnostic criteria for HRS (modified)
Despite these criteria, HRS is frequently misdiagnosed. Distinguishing between HRS and both pre- renal failure and ATN can be difficult. In the case of pre-renal failure, the urinary chemistry is similar and it is failure to improve with volume expansion which is the hallmark of HRS. Whilst fractional excretion of sodium (FeNa+) is normally more than 2% in ATN, it can be less than 1% in the setting of renal ischaemia in cirrhosis. Granular and epithelial cell casts can be found due to hyperbilirubinaemia, for reasons which are unclear.
Subtypes of hepatorenal syndrome
2 subtypes have traditionally been recognised.
Type 1: cirrhosis and rapidly progressive RF. This rapidly progressive RF causes doubling of creatinine in less than 2 weeks. It may arise spontaneously or in association with treated spontaneous bacterial peritonitis and is usually accompanied by rapid deterioration of liver function.
Type 2: Cirrhosis and subacute RF. This type is characterised by stepwise deterioration of renal function over a longer time period, often in patients with refractory ascites. The median survival is longer than in type 1 HRS and these patients do not frequently come to the attention of general intensive care units.
Recent work expands the list to 4 subtypes, to allow for classification of patients with pre-existing renal disease and in the setting of acute liver failure.
Type 3: cirrhosis with types 1 or 2 HRS superimposed on chronic renal or acute kidney injury.
Type 4: Fulminant liver failure with HRS
It is unclear whether the pathophysiology of renal failure with acute liver failure is shared with that occurring in cirrhosis. The discussion which follows mainly concerns type 1 HRS.
Pathophysiology: peripheral vasodilation hypothesis
The splanchnic circulation of cirrhotic patients is dilated due to mediators including nitric oxide (NO), vasoactive intestinal peptide (VIP), and glucagon which are overproduced by vascular endothelium in the splanchnic and portal circulation and not metabolised by the cirrhotic liver with portosystemic connections. This leads to a reduced systemic vascular resistance (SVR). This is interpreted in turn by the juxtaglomerular apparatus (JGA) of the kidneys as effective hypovolaemia, and the sympathetic nervous system (SNS) and renin-angiotensin-aldosterone system ( RAAS) is activated, leading to intense renal vasoconstriction. ADH is activated, giving rise to the hyponatraemia and ascites associated with cirrhosis. In extreme instances, acute cortical necrosis due to renal artery vasoconstriction can occur. The cardiac output in HRS was traditionally felt to be high, but recent thinking puts cardiac dysfunction on a background of cirrhosis as an important trigger of HRS. This so-called “cirrhotic cardiomyopathy” is distinct from alcoholic and ischaemic cardiomyopathy, both common in this patient group. Vasoconstriction is also observed to occur in the cerebral arterial circulation, which combines with hepatic encephalopathy to depress consciousness.
Intrahepatic vasoconstriction contributes to hepatic dysfunction. Relative adrenal insufficiency due to vasoconstriction is commonplace and should be sought and treated.
Risk factors for Hepatorenal syndrome
Patients with the following conditions are at risk for HRS:
- Spontaneous bacterial peritonitis (SBP)
- Alcoholic hepatitis
- Tense diuretic-resistant ascites, especially after paracentesis
- Recent variceal bleed
- Severe hyponatraemia
- MELD score >18 (see later in text for an explanation)
Histology
The histology of kidneys with hepatorenal syndrome is generally normal, although tubular dysfunction can be seen on electron micrography in longer standing cases.
Presentation
HRS presents rapidly in patients who may have had recent surgery, a GI bleed, acute hepatitis, rising creatinine despite adequate volume restoration or treated SBP. Up to 25% of SBP cases develop HRS despite resolution of infection. The urine output is generally low to normal with severe oliguria being a pre-terminal feature. The urinary chemistry, whilst potentially unreliable, usually shows urinary Na+ <10mmol/L, and concentrated urine in line with pre-renal failure. The urinary sediment is bland.
Prognosis
The prognosis in type-1 HRS is dismal, with a median untreated survival of 2 weeks. Type-2 HRS has a median survival of 6 months in keeping with the slower renal decline. The goal of treatment in established type-1 HRS is to bridge successfully to liver transplantation.
Treatment
Volume restoration
Discontinuation of diuretics and volume expansion with albumin at 1g/kg (5mls 20%/kg), followed by 20-40g/day directed toward appropriate cardiac endpoints is combined with vasoconstriction. Traditionally central venous pressure monitoring has been described in the gastroenterology literature, but dynamic cardiac output is desirable in an intensive care setting. Patients taking chronic beta- blocker therapy for portal pressure control generally have these discontinued. Albumin is preferred on the basis of evidence from small trials comparing it to saline. It may modify endothelial function as well as expanding plasma volume.
Systemic vasoconstriction
Terlipressin is a powerful Vasopressin V1 receptor agonist which causes systemic vasoconstriction and raises blood pressure. It helps to override the splanchnic vasodilatation seen in HRS and thus improve renal perfusion and lower creatinine levels.
Terlipressin itself is inactive but is converted to lysine-vasopressin by tissue enzymes. It is long – acting and generally commenced at a dose of 1 mg qds iv, increasing to a maximum of 2 mg 4-hourly if there is little response in serum creatinine. Octreotide, a somatostatin analogue, can be substituted if there is known coronary disease or other ischaemic contraindication.
Some studies have shown no difference between terlipressin and noradrenaline in terms of efficacy. A recent meta-analysis showed a significant increase in reversal of HRS, but no improvement in survival in patients treated with terlipressin.
In the US, a combination of midrodine, an alpha-agonist and octreotide, has been studied, and is frequently used. The combination is attractive in the outpatient setting of type 2 HRS since midrodine can be administered orally and octreotide subcutaneously.
Renal replacement therapy
Many patients with type 1 and type 4 HRS referred to intensive care units are in established multi- organ failure and renal replacement therapy (RRT) is offered as part of the overall organ support. RRT does not improve prognosis but helps to ameliorate fluid overload, acidosis and hyperkalaemia.
The degree of reversibility of the liver injury (e.g. alcoholic hepatitis) and whether or not the patient is a candidate for liver transplantation will dictate the appropriateness and duration of this treatment.
Expert multidisciplinary assessment involving hepatologists, critical care physicians and transplant surgeons should be carried out, especially if this modality is not to be offered on the grounds of futility.
TIPSS (transjugular intrahepatic portosystemic shunt)
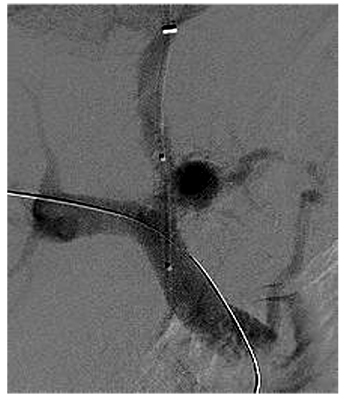
Figure 1. TIPSS procedure. The diagram shows a radiograph of a wire entering the portal vein from above prior to stenting. (Att. Samir@en.wikipedia)
TIPSS can lower portal pressure and prevent splanchnic pooling. There is limited data to suggest TIPSS can reverse HRS or control resistant ascites and reduce refractory variceal bleeding. Survivalwas improved in one small case series to a mean of 5 months.
Many patients with type 1 HRS are too ill to undergo TIPSS and it carries significant complications; notably worsening hepatic failure, cardiac overload due to increased venous return, and encephalopathy.
The Model for End-stage Liver Disease (MELD) score was originally devised to predict survival after TIPSS and it is generally contraindicated in patients with a MELD greater than 18, as median survival is 3 months or less. In view of the risks associated with TIPSS, it is generally used only in patients who are candidates for transplantation rather than as “destination therapy” although this can be considered on an individual basis.
Extracorporeal therapies
Extracorporeal therapies such as the molecular adsorbent recirculation system (MARS) have been shown to reduce ammonia, bilirubin and creatinine levels and this has translated into survival benefit in some studies. Currently, similar to TIPSS, it is primarily a bridging tool to transplant.
Orthotopic liver transplant (OLT)
Patients with type 1 and 4 HRF should be evaluated for OLT. Patients with type 3 HRS or chronic ATN together with HRS may require combined liver and renal transplantation. If appropriate and successful, OLT results in resolution of HRS in the transplanted patient, although over one third of these patients require renal support post-op, and recovery is gradual. On-going renal impairment post transplant may require dose reduction of anti-rejection immunosuppressants such as cyclosporin or tacrolimus. Three-year survival is roughly 60% after OLT, as distinct from 70-80% in patients without HRS.
Prevention of HRS
There is evidence for HRS prevention in certain specific situations:
In patients with cirrhosis and low ascitic protein who had Child –Pugh scores >9 and bilirubin >51.3 micromol/L; or creatinine> 106mmol/L or sodium<130mmol/L, quinolone administration (norfloxacin) was associated with a decreased probability of developing SBP and HRS, and improved survival at 3 months and 1 year.
In established SBP, albumin at 1.5 g/kg at the time of diagnosis and 1g/kg on day 3 of antibiotics may reduce the incidence of renal failure and improve 3 month mortality.
In large volume paracentesis (LVP), attention must be paid to volume replacement. Albumin 20% 250mls for every 5L is usually recommended.
In patients with alcoholic hepatitis, administration of the tumour necrosis factor (TNF) inhibitor pentoxifylline at 400 mg tds reduces the occurrence of HRS.
The organ donor with HRS
The presence of hepatorenal syndrome in an organ donor does not preclude transplantation of their kidneys to a patient requiring this, as the histologically normal kidneys have a high success rate once transplanted into a recipient without liver disease. Clearly, diagnostic accuracy is essential as other causes of renal failure such as ATN or intrinsic renal disease will function far less well after transplantation.
IMPORTANT POINTS
- HRS type 1 is generally fatal without transplant
- The diagnosis can be difficult despite clear criteria
- Preventive measures are worthwhile in at-risk patients
ANSWERS TO QUESTIONS
-
- T (although some classifications include renal failure with acute liver failure as type 4 HRS)
- F it is characterised by splanchnic vasodilatation
- F myocardial dysfunction is common
- F (although these are classically distinct, it is not invariable)
-
- T renal recovery is slower and less complete
- F
- T (if dual pathology present)
- F
-
- T
- T by reducing portal pressure
- T (if used for treatment of ascites)
- T
-
- T (As distinct from V2 receptors, which are mainly concerned with renal water resorption)
- F (it is long acting and can be given as peripheral boluses))
- F
- F (it is probably equivalent, but insufficient data exists)
REFERENCES AND FURTHER READING
- Burton Rose, M.D.,Hepatorenal syndrome; Up to date;2010; ed.
- Arroyo, V., Fernandez, J., Gines, P.,Pathogenesis and treatment of hepatorenal syndrome;, Semin Liver dis;2008;28:81-95.



