Intensive Care Medicine
QUESTIONS
Before continuing, try to answer the following true/false questions. The answers can be found in the text and are provided at the end of the article.
- When compared to conventional ventilation for patients with acute respiratory distress syndrome, high frequency oscillatory ventilation (HFOV) is associated with a reduction in mortality rate.
- In HFOV both inspiration and expiration are active processes.
- Mean airway pressure (MAWP) is characteristically lower in HFOV compared to conventional ventilation.
- Respiratory rates of up to 600 breaths per minute may be employed in HFOV.
- Increasing frequency (respiratory rate) of HFOV is associated with a fall in arterial CO2 concentration.
- Sudden loss of chest wiggle factor may indicate total airway obstruction.
INTRODUCTION
High frequency oscillatory ventilation (HFOV) is an alternative form of mechanical ventilation that can be delivered on critical care units. Unlike conventional ventilation (CV), HFOV relies on the rapid delivery of tidal volumes that are smaller than dead space. Typical tidal volumes on HFOV are 1-3 ml/kg. HFOV has been successfully used in neonates and paediatrics since 1983. Studies have shown higher survival rates for patients in these groups with acute respiratory distress syndrome (ARDS).1, 2 However, the evidence base for the use of HFOV in adults is limited. To date, studies have shown HFOV is safe and equivalent to CV in adult patients with ARDS, but no mortality benefit has been demonstrated. 3, 4 Of note, whether HFOV is superior to CV in adult patients with ARDS is the subject of two current randomised controlled trials, one in the UK and one in Canada. 5, 6
Despite this lack of a documented mortality benefit there is growing use of HFOV in adult critical care units throughout the UK. In the last five years 26 ventilators were sold in the UK, 10 of which were purchased in 2011 alone 5 . This article will review the basic principles of HFOV and provide practical advice on the selection and clinical management of adult patients.
BASIC PRINCIPLES
Theory
HFOV utilises much higher frequencies than CV (120 – 600 breaths per minute versus up to 40 breaths per minute for CV). This allows the use of tidal volumes that with conventional ventilation would lead to rising CO2 levels. The principle goals for ventilating a patient with ARDS using HFOV are to prevent ventilator induced lung injury (VILI) and to achieve adequate ventilation and gas exchange with as low a fractional inspired oxygen concentration (FiO2) as possible.
The key features of HFOV that are thought to be responsible for decreasing the incidence and severity of VILI when compared with CV are explained below.
Smaller tidal volumes – This helps prevent volutrauma caused by alveolar over-distension.
Higher mean airway pressure (MAWP) – The continuously high distending pressure provides improved oxygenation via improved alveolar recruitment.
Smaller differences between inspiratory and expiratory pressures – This helps prevent atelectotrauma associated with cyclical alveolar collapse and distension that can be a feature of CV.
Lower peak pressures – This helps reduce barotrauma.
Physiology
Gas transport on HFOV is thought to occur via 5 mechanisms, as suggested by Weavind and Wenker in 2000. 7 The individual contribution of each mechanism to overall gas exchange remains debated:
- Bulk flow. This is the predominant mechanism of gas transport seen in CV and it plays a part in HFOV, providing gas delivery to proximal alveoli with low regional dead space volumes.
- Pendelluft. This refers to inter-regional gas mixing between alveolar units whereby there is transient movement of gas out of some alveoli and into others when flow stops at the end of inspiration and in the opposite direction at the end of expiration. This occurs where regions of the lung differ in compliance or airway resistance so that their time constants of filling in response to changes in trans-pulmonary pressure are not the same.
- Taylor dispersion. This leads to mixing of fresh and residual gases along the front of a flow of gas through a tube, due to the interaction of the axial velocity profile and radial concentration gradient.
- Coaxial flow. This occurs when gas in the centre flows inward and the gas on the periphery flows outward. It is attributed to the asymmetry between inspiratory and expiratory velocity profiles.
- Augmented molecular diffusion. This occurs at the alveolar level as a result of the added kinetic energy supplied by the oscillations.
Oxygenation on HFOV is similar to conventional ventilation (CV). It is dependent on two set variables; FiO2 and MAWP.
CO2 clearance on HFOV differs however. During conventional ventilation CO2 removal is dependent on minute volume, the product of tidal volume (Vt) and respiratory rate. An increase in minute volume leads to an increase in CO2 clearance. CO2 clearance on HFOV is also dependent on the frequency of oscillations and tidal volume (Vt). However, as will be explained later, a decrease in frequency on HFOV leads to larger tidal volumes and subsequent increased CO2 clearance. This is the opposite of CV. Cycle volume is analogous to tidal volume and is a function of amplitude of oscillation. This can be set on the Vision alpha Novalung ventilator (Respironics). Increasing the cycle volume will increase CO2 clearance.
THE VENTILATOR
A range of high frequency oscillatory ventilators are commercially available. The 2 in use for adult patients in the UK are the Vision alpha Novalung (Respironics) and the SensorMedics Model 3100B (Sensor medics corporation). The remainder of this article will focus on the Novalung but the general principles are common to all oscillatory ventilators.
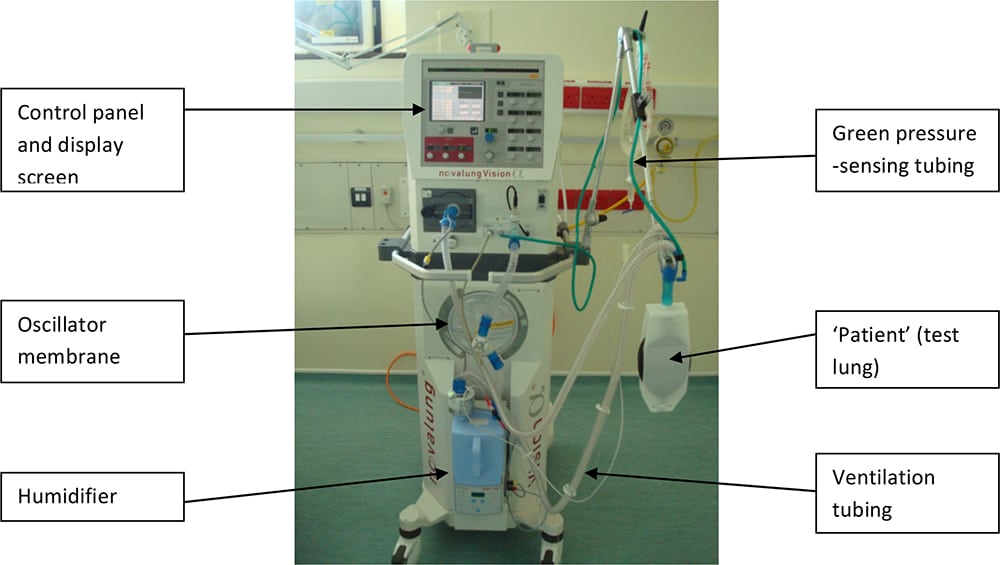
Figure 1: Picture of Novalung vision ventilator currently used in the OSCAR trial 5
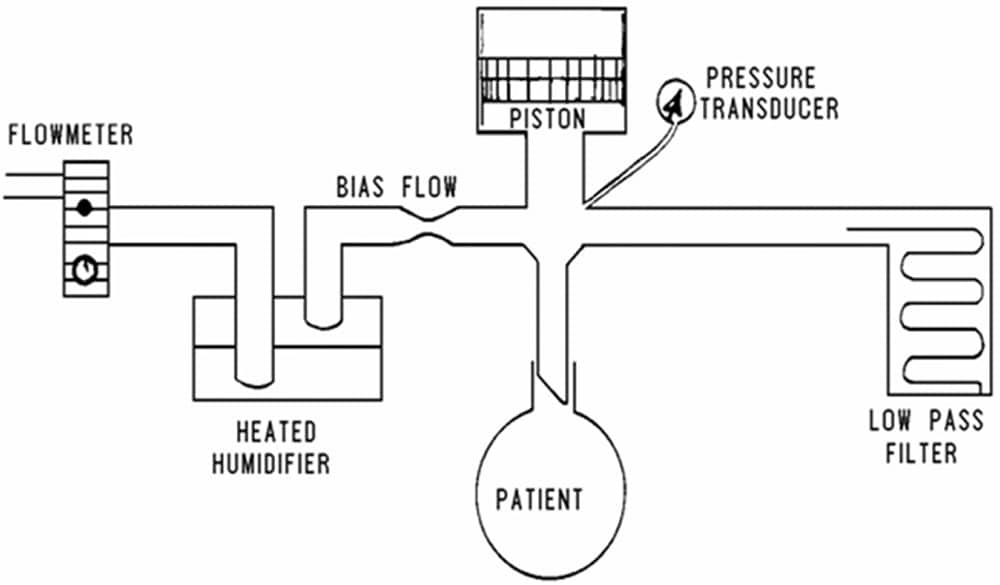
Figure 2: Schematic representation of component parts of HFOV ventilator 4
HFOV ventilators for both adults and paediatrics follow the same basic principles. A flow of warmed humidified gas (bias flow) is run across the proximal end of the endo-tracheal (ET) tube at 20-40 l min-1 . An oscillatory piston pump vibrates this bias flow of gas so that a portion of the gas is pumped into and out of the patient. Thus, in contrast to CV, inspiration and expiration are both active processes in HFOV. The frequency of the vibration is usually between 3 – 10 Hz. As 1 Hz = 1 cycle per second, patients on HFOV receive respiratory rates between 180 – 600 breaths per minute. Occasionally a bias flow above the normal value of 20 l min-1 may be needed. Examples where a higher bias flow may be required are during suctioning, after introduction of a cuff leak in order to maintain MAWP or in the presence of specific pathology such as a broncho-pleural fistula.
Cycle volume is synonymous with tidal volume delivered. This is controlled by the distance the piston pump moves in and out, the amplitude of oscillations. On the Novalung a specific cycle volume can be set from within a range that is dependent on frequency. Altering cycle volume is another way, other than changes in frequency, in which CO2 clearance can be altered. Vt may however vary unpredictably, particularly if a patient is making respiratory effort or there is a change in compliance such as pneumothorax or partial/total obstruction of the patient’s ET tube. A clinically useful measure of tidal volume is the ‘chest wiggle factor’ (CWF). This refers to visible movement of the patient generated by the oscillations. Under normal circumstances such movement should be visible from chest to mid-thigh during HFOV. CWF may be used as a surrogate marker of Vt and sudden changes in observed CWF should prompt urgent patient evaluation, as discussed later in the text. Reducing frequency on HFOV leads to increased amplitude. This explains why in contrast to CV, a reduction in ventilatory rate during HFOV is associated with increased CO2 clearance.
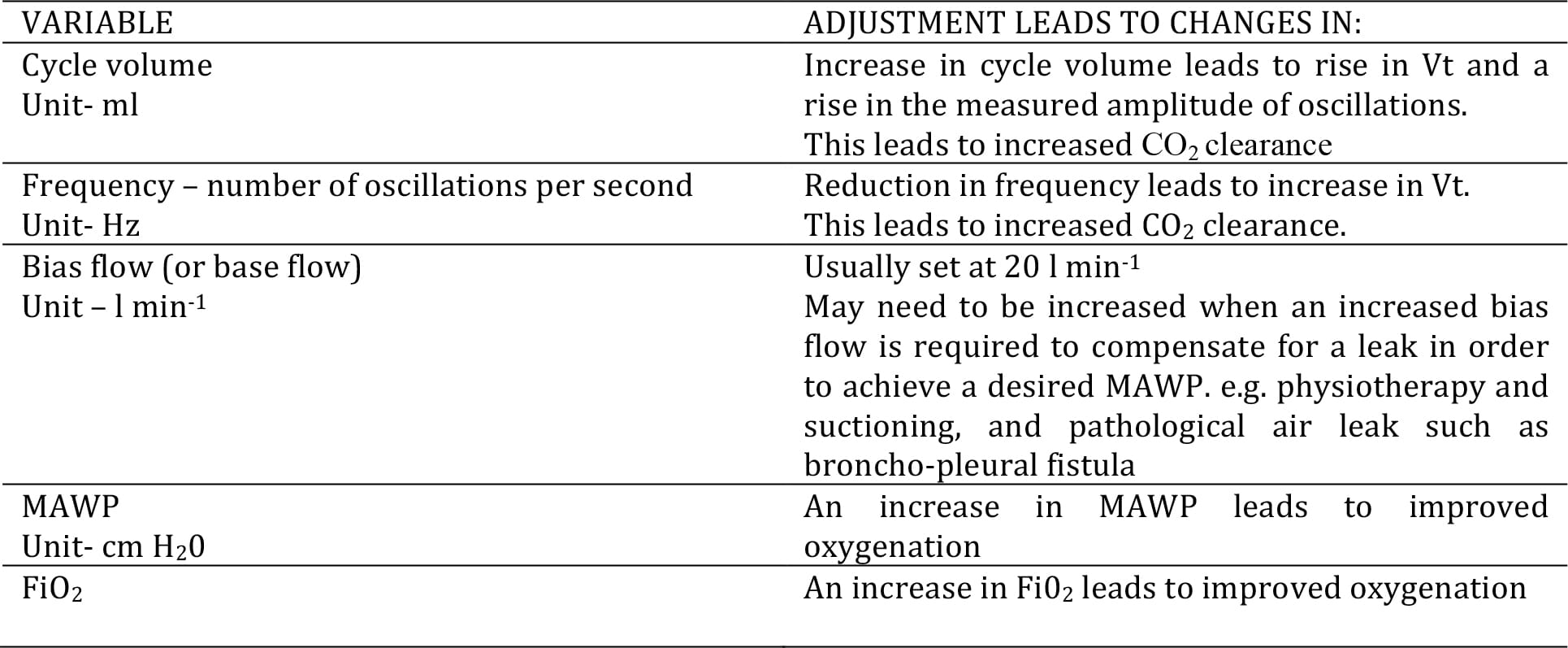
Table 1: Adjustment of HFOV variables
PATIENT SELECTION
Potential indications
Recently there has been an increase in the use of HFOV in critical care units in the developed world. Early studies by Derdak3 , Fort8 , Mehta9 and Matthias10 have focused on the use of HFOV as rescue therapy in patients failing conventional ventilation. The main potential indications for use of HFOV in adult critical care patients remain the treatment of patients with ARDS or broncho-pleural fistula. Current evidence suggests that HFOV is equivalent to CV without significant improved outcomes for both these groups of patients. In patients with refractory broncho-pleural fistula, HFOV may reduce leak size and promote healing. 11, 12 However it is unlikely that this approach will ever be investigated in a clinical trial appropriately powered to detect a mortality benefit. Regarding the use of HFOV in patients with ARDS it is hoped that the two current trials investigating the use of HFOV in adult patients with ARDS will help identify whether this patient group may benefit from HFOV.5, 6 In addition, these trials may provide some information to help identify whether patients may benefit more if HFOV is commenced early. In contrast to neonatal practice, HFOV is not however recommended as a first line ventilatory strategy in adults.
PRACTICAL MANAGEMENT
Initial patient preparation
There are many areas of consideration that should be addressed prior to initiating HFOV. These include patient and equipment preparation and staff and family briefing. As HFOV remains a less common mode of ventilation use of a guideline is strongly recommended. Many such guidelines exist. On our unit we use the OSCAR study guideline. This is may be downloaded from the OSCAR website (see link in references):
An alternative guideline was published by Fessler et al.13 The following points should be considered prior to starting HFOV.
- Suction the airway and check ET tube patency.
- Consider changing ET tube if small tube size, i.e. less than # 8.0.
- Bronchoscopy, if indicated, should take place prior to starting HFOV.
- Ensure adequate sedation/ analgesia while still on CV.
- Neuromuscular blockade should be considered but may not needed for initiation or duration of HFOV.
- Intravascular volume status should be assessed. The higher MAWP used with HFOV may lead to reduced preload and hypotension. Supplemental fluid or vasoactive drug therapy may be required.
TIP: Perform an inspiratory pause whilst the patient is receiving CV to try to identify hypovolaemia prior to starting the HFOV therapy. Hypovolaemia should be suspected if an inspiratory pause is associated with hypotension. - Any trips planned for off-unit imaging or interventions should be performed prior to stating HFOV if possible as the ventilator is not portable.
Suggested initial HFOV ventilator settings:
FiO2. 1.0
Frequency. An increase in PaCO2 is often observed after commencing HFOV. Particular attention must be paid to selecting an appropriate starting frequency if an increase in PaCO2 is likely to be poorly tolerated. We have found the starting frequency of 10 Hz recommended in the OSCAR study guidelines often to be too high. A lower starting frequency of 8 Hz or less may be more appropriate. Alternatively there may be some place for the use of buffering with sodium bicarbonate, but this is controversial. 14, 15
Mean airway pressure (MAWP). One of the basic principles of HFOV is ventilation with a constant high distending pressure. Initial MAWP is set at 5 cmH2O more than the MAWP on CV.
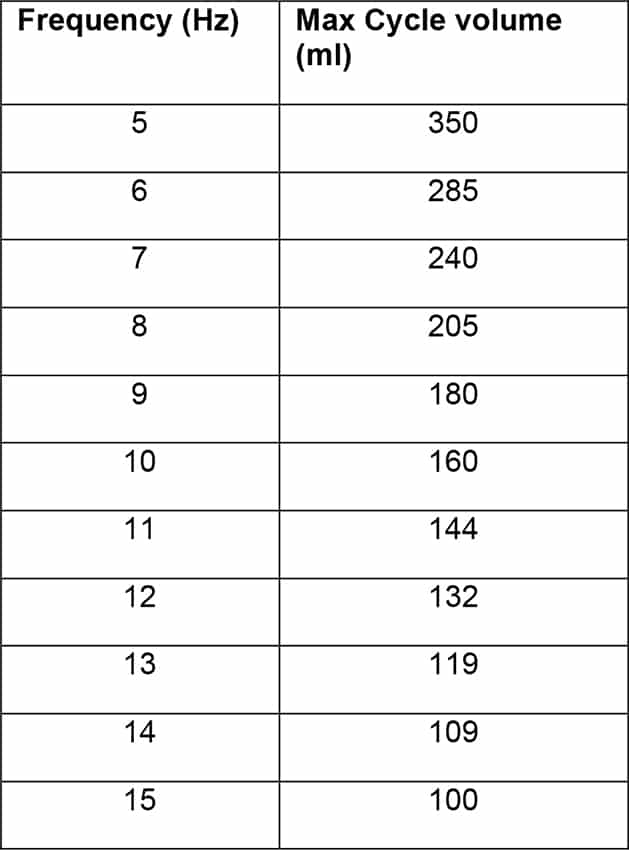
Cycle volume. This correct initial cycle volume varies with frequency selected. If a starting frequency of 8 Hz is selected on the Novalung appropriate cycle volume is 170ml.
Base flow (Bias flow). 20 lmin-1

Figure 3: Vision Alpha ventilator control panel with close up of data screen which includes graphical representation of oscillations
If oxygenation is poor prior to commencing HFOV it is advisable to perform a recruitment manoeuvre such as an inspiratory hold, 30-40 cm H20 for 40 seconds. This should be aborted if haemodynamic instability occurs and remedial action taken prior to commencing HFOV.
The HFO ventilator should then be started and an arterial blood gas (ABG) performed 15 minutes after starting HFOV to guide further management. For the Novalung it is important to remember that any changes to HFOV settings need to be confirmed by pressing the accept button, displayed in figure 3. Flow diagram 1 contains a simplified algorithm for initial settings and continued management.
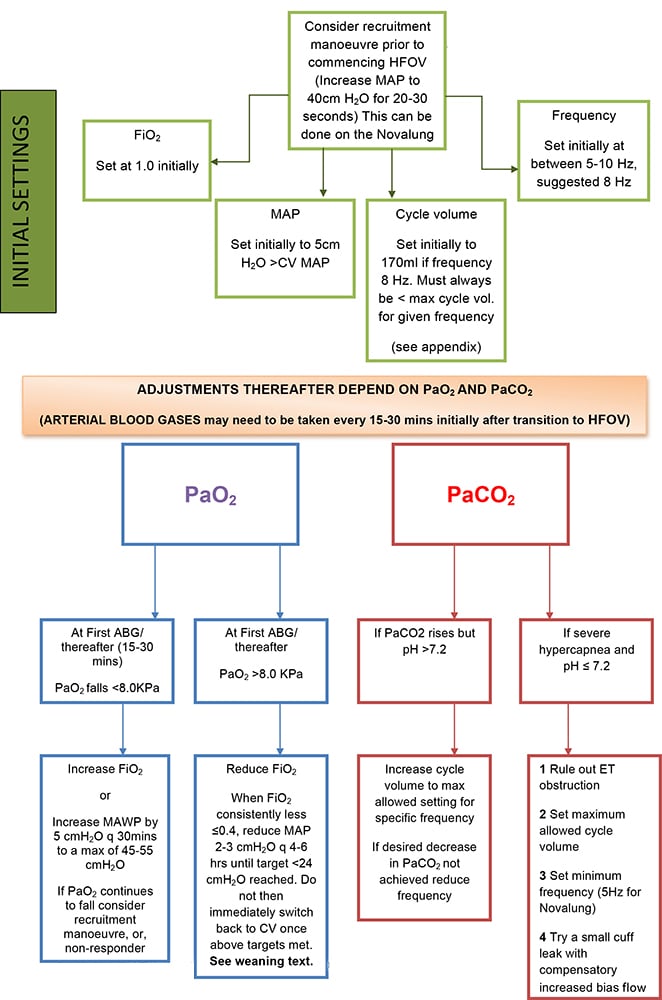
Flow diagram 1: Initial HFOV settings and management
PATIENT CARE DURING HFOV
In addition to standard nursing and medical care specific attention should be paid to the following points for patients receiving HFOV.
General considerations
Set up/ Nursing
Patients on HFOV require one to one nursing
In the absence of any contra-indications the following targets are appropriate:
- Sp02 > 87%
- Any PaCO2 associated with a pH > 7.2.
A closed suction system should be used to minimise airway disconnections and subsequent de-recruitment. Suctioning may be performed in patients on HFOV. Temporarily increasing bias flow before and during suctioning may help prevent a decrease in MAWP and de-recruitment.
Inspired gases should be humidified to help prevent necrotizing tracheobronchitis.
Good communication and adequate explanation to relatives are required due to the unusual appearance of a patient receiving HFOV.
Monitoring / Examination
Prompt action is required in patients on HFOV who experience a rapid increase in PaCO2 as improvements in PaCO2 do not occur as quickly after changes on HFOV compared to CV.
End tidal CO2 monitoring cannot reliably be used.
Auscultation of the lungs/ heart or abdomen is not possible due to background ventilator noise.
Chest wiggle factor should be regularly assessed. ‘Wiggle’ should be equal on both sides of the chest. Close monitoring will afford early recognition of ET tube displacement, airway obstruction with secretions or pneumothorax. CWF should be re-assessed after any patient re-positioning.
Special procedures / specific cases
It may be necessary to revert to conventional ventilation for certain procedures, such as echocardiography, and central line insertion. Doppler cardiac output monitoring is unreliable during HFOV.
In patients with large air leaks and poor oxygenation a maximum bias flow of 40 l/min may be required in order to achieve desired MAWP.
Bronchoscopy is not well tolerated in patients on HFOV as it results in reduced MAWP.
Oscillatory ventilators are not portable therefore patients cannot undergo transfer whilst on HFOV.
Delivery of nebulised drugs to patients on HFOV requires a specific nebuliser but is still unreliable. If bronchodilators are required they should probably be administered intravenously.
Emergency scenarios associated with HFOV
Hypotension may be caused by increased MAWP on HFOV causing reduced cardiac preload. This is exacerbated by coexistent hypovolaemia. Careful assessment of fluid status is essential. Additional fluid boluses and / or vasopressors may be required.
Subtotal obstruction of ET tube will lead to progressive hypercapnea. Abrupt increases in PaCO2, loss of chest wiggle factor and a change in observed amplitude may all indicate an obstructed airway. A suction catheter should be passed immediately to check ET tube patency. If ET tube is obstructed it should be replaced.
Pneumothorax and ET tube displacement are difficult to recognise in patients receiving HFOV. A high index of suspicion is required for both clinical situations. There may be a change in amplitude, loss of oscillations on the side of pneumothorax or area no longer ventilated after ET tube displacement as well as an abrupt rise in CO2. Hypoxia and hypotension may also ensue.
If PaCO2 is dangerously high ET tube obstruction must be ruled out. The patient may then be disconnected from HFOV and manually hyperventilated with a PEEP equipped bag (i.e. Water’s circuit). When recommencing HFOV the lowest frequency and highest cycle volume should be set. Introducing a small cuff leak may also be necessary. The latter can be performed by slowly deflating the cuff pressure until the mean airway pressure drops by 5 cmH20. Then the bias flow should be increased until the MAWP returns to previous value. As it is difficult to achieve a rapid drop in PaC02 on HFOV, close monitoring of PaCO2, anticipation of problems and prompt action are all required
Cardiac arrest. In the event of cardiac arrest, HFOV should be discontinued and manual ventilation commenced, preferably with a PEEP equipped bag (Water’s circuit).
Weaning from HFOV
Whilst neonates can be transitioned directly to extubation from HFOV, adults need to be weaned back to CV first. The first priority in weaning HFOV is to decrease Fi02 from toxic levels. Whilst some changes in Fi02 may be guided by Sp02 rather than PaO2, all changes of frequency and cycle volume should be guided by arterial blood gas sampling. Regular arterial blood gas sampling is therefore required. Regarding oxygenation, once oxygen saturations of ≥90% are consistently achieved with an Fi02 ≤ 0.4, decreases in MAWP may be made. Reductions in MAWP of 2-3 cmH20 steps should be trialled. Conversion back to conventional ventilation may be considered once Fi02 requirement is ≤ 0.4 and MAWP is <24 cm H20. It is our practice to continue HFOV for at least 12 hours and up to 24 hours after these targets have been achieved. This is because we have found that immediately converting to CV once the above targets have been met is often associated with deterioration in gas exchange. The Novalung ventilator can deliver both conventional pressure and volume modes of conventional ventilation. Therefore to begin with we provide CV via the Novalung, prior to switching to our usual ICU ventilator at a later time.
SUMMARY
HFOV is increasingly being used in critical care units for the treatment of patients with ARDS. It has been shown to be both safe and effective in improving the refractory hypoxaemia associated with this condition. It remains to be seen however when compared to conventional ventilation whether the theoretical advantages of HFOV translate into significantly improved patient outcomes. The results of 2 prospective randomised trials are eagerly awaited.
QUESTION ANSWERS
- False. This has not been proven in adequate clinical trials.
- True
- False
- True
- False
- True
REFERENCES
- Yildizads D, Yapizioglu H, Bayram I et al.: High frequency oscillatory ventilation for acute respiratory distress syndrome. Indian Journal of Paediatrics Sept 2009, vol./is. 76/9(921-7), 0019-5456
- Kessel I, Waisman D, Barnett-Guring O et al : Benefits of High frequency Oscillatory ventilation in premature infants. Israeli Medical Association Journal March 2010, vol/is. 12/3(144-9), 1565-1083 (2010 March)
- Derdak S, Mehata S, Stewart TE, Smith T, Rogers M, Buchman TG, Carlin B, Lawson S, Cranton J : The Multicenter Oscillatory Ventilation for ARDS Trial ( MOAT) study investigators : High-frequency oscillatory ventilation for ARDS in adults: a randomised controlled trial. Am J Respir.Crit.Care Med 2002, 166:801-808
- Ritacca FV, Stewart TE : High frequency oscillatory ventilation in adults a review of the literature and practical applications. Critical Care 2003, 7:385-390
- High Frequency Oscillation in ARDS trial (OSCAR), www.oscar-trial.org. Guidelines available from: http://www.oscar-trial.org/pdf/v7-abbreviated-manual–_kpa_dec-09.pdf
- The Oscillation for Acute Respiratory Distress Syndrome (ARDS) Treated Early (OSCILLATE) Trial, Canadian Critical Care Trials Group. (http://www.oscillatetrial.com/)
- L. Weavind, O.C. Wenker: Newer Modes Of Ventilation: An Overview. The Internet Journal of Anesthesiology. 2000 Volume 4 Number 4
- Fort P, Farmer C, Westerman J, et al. High frequency oscillatory ventilation for acute respiratory distress syndrome – a pilot study. Crit. Care Med 1997; 25:937-47
- Mehta S, Lapinsky SE, Hakett DC, et al. Prospective trial of high frequency oscillation in adults with acute respiratory distress syndrome. Crit. Care Med 2001; 29:1360-9
- Matthias D, Weiler N, Heinrichs W, et al. High frequency oscillatory ventilation in acute respiratory distress syndrome. Intensive Care Med 2003; 29:1656-65
- Imai Y, Slutsky AS. High frequency oscillatory ventilation and ventilator induced lung injury. Crit. Care Med 2005; 33(3):s129-s134
- Galvin I, Krishnamoorthy R, Saad RSU : Management of advanced acute respiratory distress syndrome complicated by bilateral pneumothoraces with high frequency oscillator ventilation in the adult. B.J Anaesth. 2004; 93:454-6
- Fessler HE, Derdak S, Ferguson ND, Hager DN, Kacmarek RM, Thompson BT, Brower RG. A protocol for high-frequency oscillatory ventilation in adults: results from a roundtable discussion. Crit Care Med. 2007 Jul;35(7):1649-54
- Laffey JG, Engelberts D, Kavanagh BP. Buffering hypercapnic acidosis worsens acute lung injury. Am J Respir Crit Care Med 2000; 161: 141-146
- Kallet RH, Jasmer RM, Luce JM, et al: The treatment of acidosis in acute lung injury with tris-hydroxymethyl aminomethane (THAM). Am J Respir Crit Care Med 2000;161(4 Pt 1):1149–1153
APPENDIX
Table showing maximum cycle volume (ml) for each frequency (Hz) 5 as referred to in HFOV management flow diagram 1: