Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- The cell-mediated immune system:
- Can be responsible for autoimmune diseases
- Involves activation of lymphocytes in the periphery
- Induces proliferation of plasma cells
- Results in increased IgA synthesis
- Causes large amounts of histamine release
- Regarding B-cells:
- They are so named as they mature in the bone marrow
- They display receptors of similar morphology to the immunoglobulin produced by that cell
- Give rise to NK-cells
- Are only activated by T-cells
- Produce more than one clone of antibody
- Immunoglobulin:
- Is secreted by T-cells
- Is present in the IgA isoform on mucous membranes
- Consists of one heavy chain and two light chains
- Specificity for antigen depends on the light chains
- IgG is responsible for mast cell degranulation
Key Points
- The innate immune response is nonspecific and destroys pathogens mainly through phagocytosis and activation of complement.
- The acquired immune system is a specific response to a certain pathogen and is principally brought about by T-cells, B-cells, immunoglobulin and complement.
- T-cells can be CD8+ or CD4+, the former are known as cytotoxic T-cells and the latter become T-helper cells.
- B-cells secrete immunoglobulin. This binds to antigen on pathogens and labels the cell for destruction by complement or phagocytic cells.
INTRODUCTION
Immunity describes the ability to fight infection and is dependent on a highly functional immune system. As anaesthetists we challenge the immune system through our everyday practice, whether this is during the placement of intravascular devices, tracheal intubation or the administration of drugs. It is important to have an understanding of the fundamental science behind immunology, so we can reduce the risk to our patients and understand why some patients may be particularly prone to infections.
The aim of this first tutorial is to provide an overview of the immune response whilst the second tutorial will concentrate on immunology in clinical practice.
The immune response can be broadly categorised into two systems: innate immunity and acquired immunity. As the body’s first line of defence, the innate immune system comprises physical barriers and some cellular defences. Pathogens that evade the innate system subsequently encounter the acquired immune response, which is a specific system of cellular and humoral responses that have developed during an individual’s lifetime.
INNATE IMMUNITY
ANATOMICAL BARRIERS
Barrier mechanisms are responsible for the first line of defence against pathogens and the most significant barrier is the skin1. The skin provides a surface that is difficult to penetrate and provides excellent defence against infection. It explains why there is an increased incidence of both local and systemic infections when this barrier is compromised, for example in patients with skin diseases or burns.
Pathogens can gain access to the body through other anatomical areas. The respiratory, gastrointestinal and genitorurinary tracts are sites where specialised mucous membranes play an important role as a barrier in preventing infection. Importantly, however, it is not only physical and chemical mechanisms that are involved. There are essential immunological factors that defend against infection at this level. Immunoglobulin A (IgA) is an antibody secreted by mucosal cells that binds to pathogens and their toxins to neutralise and deactivate them before they are able to invade.
CELLS AND PROTEINS
The cells and proteins of the innate immune system provide a line of defence that is present from birth. Unlike the acquired system, their response is not altered by subsequent invasion and they do not have a ‘memory’. If an anatomical barrier has broken down, pathogens will be able to enter the underlying tissues. At the tissue level, the innate immune system provides non-specific defences that prevent microbial growth and subsequent infection. This system responds to invasion through cellular and complement protein responses. The latter will be discussed at the end of this tutorial.
The cellular response1,2,3 is bought about by leucocytes, which can be granulocytes (neutrophils, eosinophils and basophils) or agranulocytes (monocytes and lymphocytes). The innate system aims to destroy the invading pathogens rapidly to prevent spread of infection. An important tool in its armoury is phagocytosis (Figure 1). This describes the process by which cells engulf foreign material and kill microbes. Macrophages and neutrophils are the primary phagocytic cells of the innate system but other cells also play a role.
Macrophages are the primary phagocytic cells and are one of two cell lines of cells that mature from monocytes. Monocytes also differentiate into dendritic cells. These have limited phagocytic activity but do play a crucial role in linking the innate system with the acquired immune response through their role as antigen presenting cells. The importance of this will be discussed later in the tutorial.
In order for phagocytosis to take place, the pathogen must first be recognised as being ‘foreign’. This occurs through interactions between specialised cell-surface receptors on the macrophage with ‘pathogen associated molecular patterns’ (PAMPs) which are expressed by microbes. An example of this interaction is the toll-like receptor (TLR) expressed by macrophages recognising lipopolysaccharide (LPS) on gram negative bacteria.
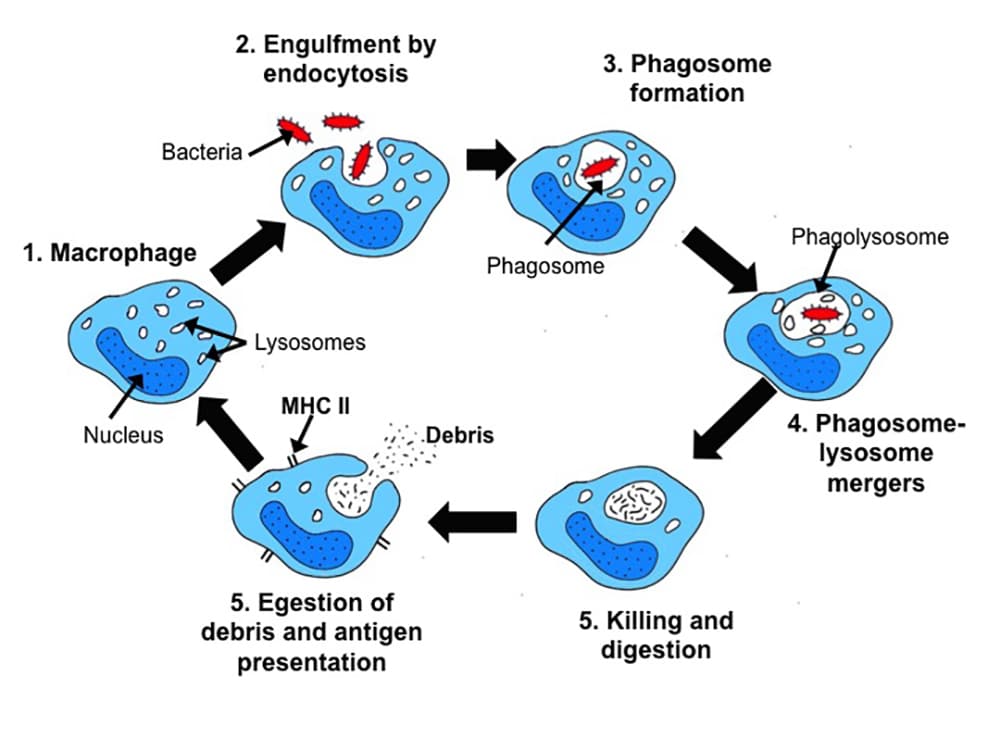
Figure 1: Interaction of the microbe with the macrophage stimulates phagocytosis, the phagosome fuses with macrophage lysosomes which acidify and digest the microbe. The macrophage presents microbial particles on its surface in association with MHC class II which allows activation of other immune cells.^3
A second class of phagocytes is the polymorphonuclear leucocytes, more commonly known as neutrophils. These cells are recruited early in infection by macrophages through release of pro-inflammatory cytokines such as Interleukin- 1ß (IL- 1ß), IL-6, IL-12 and IL-18. This serves to increase the number of phagocytic cells in the tissue to help destroy pathogens. In addition to the cytokines listed above, macrophages release tumour necrosis factor-α (TNF-α), prostaglandins, leukotrienes and platelet activating factor. These mediators induce the changes seen in inflammation such as increased vascular permeability, clotting and pain. The purpose of this response is to prevent both the spread of infection via the blood and to recruit further immune cells.
Not all invaders are extracellular. Viruses infect host cells and can evade phagocytosis by macrophages. The innate immune system is able to combat intracellular infections through another cell line – the natural killer (NK) cell. Through the pro-inflammatory cytokines, these cells are recruited to the site of infection and identify infected cells through the display of altered or absent major histocompatibility (MHC) class 1 complexes (Figure 2).
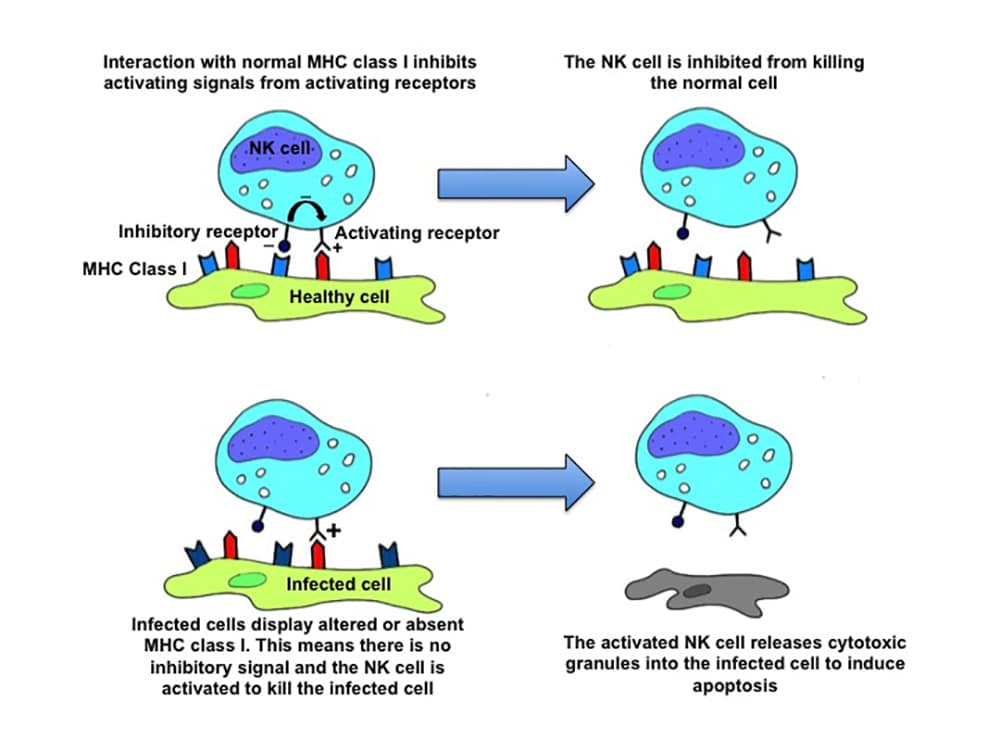
Figure 2: Healthy cells display normal MHC complexes and will not activate NK cell killing. Infected cells display altered or abnormal MHC and will trigger cell killing by NK cells.^1
ACQUIRED IMMUNITY
Acquired immunity describes responses to infection that have been ‘learnt’ by the immune system over time. Innate immunity cannot always control an infection alone and therefore works in unison with the highly specialised cells and proteins of the acquired immune response.
Following primary pathogenic exposure, the acquired response takes time to develop. However, due to the memory of this system, subsequent infections with the same pathogen lead to a rapid response. We take advantage of this system when we vaccinate individuals.
The acquired system is broadly categorised as having a humoral component brought about by B-lymphocytes (B-cells) and a cellular component brought about by T-lymphocytes (T-cells). As lymphocytes, these cells both mature from haemopoietic stem cells in the bone marrow but are sub-classified according to their site of differentiation; T-cells develop in the thymus, whereas B-cells develop in bone marrow.
T and B cells express highly specific cell surface receptors. The mammalian genome codes for millions of different T and B cell receptors. During development, cells are selected for the ability of these receptors to recognise specific antigens. Importantly, any cell that responds strongly to ‘self’ proteins are deleted to prevent immune attack on the body’s own cells; this is a process that may be defective in auto-immune diseases. Cells that respond strongly to non-self peptides then remain ‘naïve’ until they are activated during future infection in secondary lymphoid tissues of the spleen, lymph nodes or mucosa-associated lymphoid tissue (MALT).
T-Cells
T-cells have a role in the defence against both intra-cellular pathogens (such as viruses) and extra-cellular pathogens (such as bacteria). These cells are selected for the ability of their T-cell receptor (TCR) to recognise MHC in association with foreign peptide.
MHC complexes are displayed by all mammalian cells, but are specific to an individual. MHC class I is expressed by all nucleated cells and MHC class II is expressed only by cells of the immune system, predominantly antigen presenting cells. It is the interaction of the T-cell receptor with MHC class I or class II that gives rise to the major classification of these cells; CD4+ and CD8+ T-cells. Immature T-cells express both CD8 and CD4 co-receptors which have affinity for MHC class I and class II respectively. During development in the thymus, T-cells are exposed to antigenic peptide associated with MHC class I or class II. If the TCR associates strongly with MHC class I, the thymocyte will retain its CD8 receptor but lose its CD4 receptor. The converse occurs if the TCR has affinity for MHC class II. Following development in the thymus, naÏve T cells reside within secondary lymphoid tissue as either CD8+ or CD4+ cells where they wait to be activated.
Activation of naÏve T-cells occurs in a process called ‘antigen presentation’. Dendritic cells and macrophages involved in the innate response migrate to lymphoid tissue where they present antigen in association with MHC, this is why they are called antigen presenting cells (APCs). CD8+ T-cells are activated if their specific peptide is presented with MHC class I and CD4+ cells are activated if it is presented by MHC class II. Activation of CD8+ T-cells creates cytotoxic T-cells. As MHC class I is present on all nucleated cells, if a cell is virally infected, it will display foreign peptide on MHC. CD8+ cells specific for this antigen will kill these cells by releasing cytoplasmic vesicles into the infected cell. These vesicles contain caspases that lead to programmed cell death by apoptosis.
CD4+ T-cells have the potential to become one of four classes of cell; T-helper 1 (Th1), T-helper 2 (Th2), T-helper 17 (Th17) or T-regulatory. The formation of each sub-population depends upon the cytokine environment in which the T-cell is presented antigen and each population has a different role in inflammation. The table below depicts the differentiation of the classes of CD4+cells. It is important to note the inhibitory actions of each class on each other as in health this allows effective defence against pathogens. Polarised responses can lead to hypersensitivity reactions; these will be discussed in tutorial two.
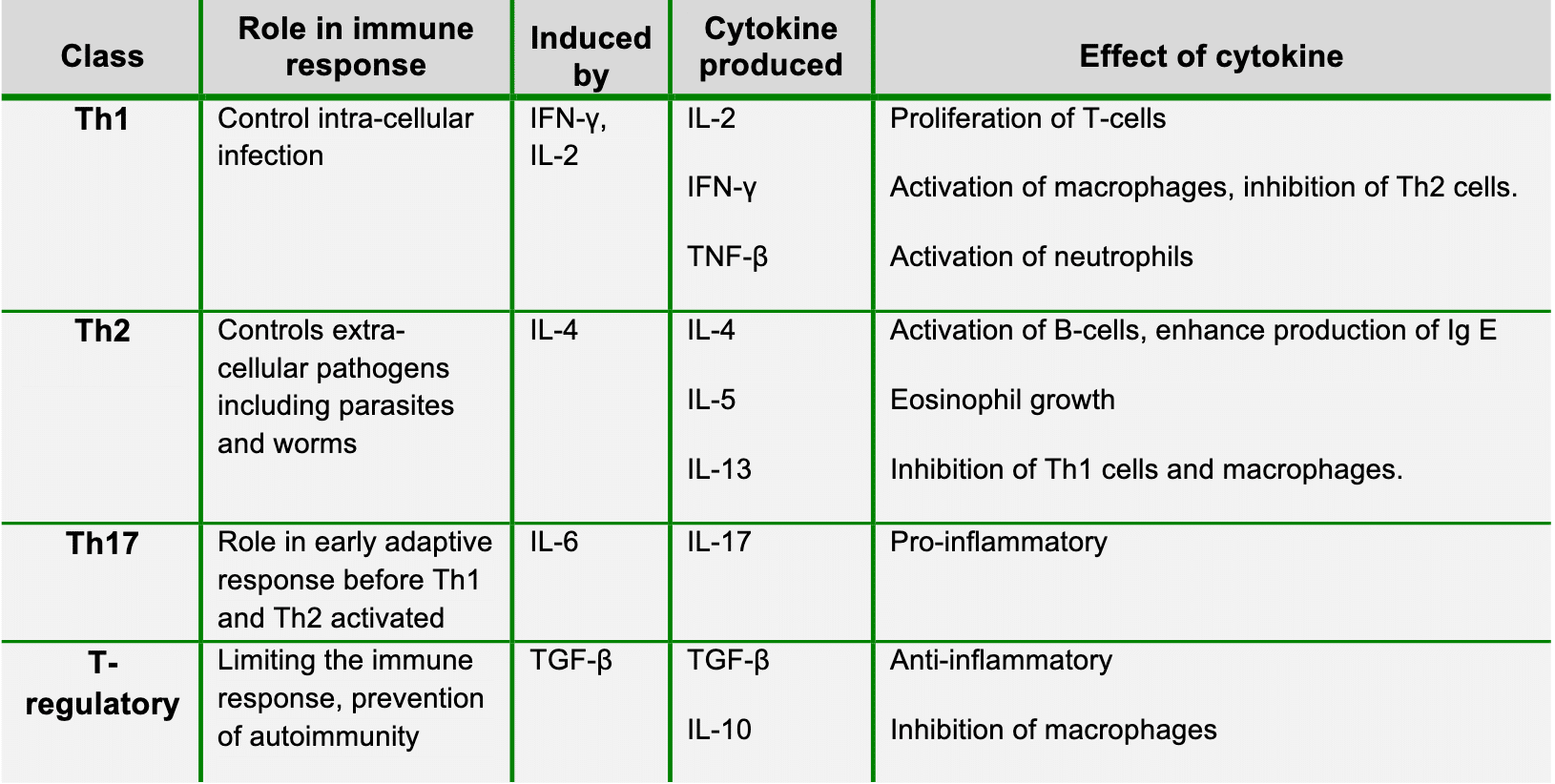
Table 1: Sub-classification of CD4+ T-cells 1,2,4 Interferon (IFN), Tumour-growth factor (TGF), Tumour-necrosis factor (TNF).
NKT-cells are a further subclass of T cell independent of the CD4+ classification. These cells are able to recognise foreign material independent of MHC and release IL-4 and IFN-γ, although they appear to favour a Th2 type response. They are not to be confused with NK cells of the innate response.
B-cells1,2,4
B-cells are involved in the humoral immune response, this means they are the immune cells involved in production of immunoglobulin (Ig) or antibodies and their role is to protect against extra-cellular pathogens.
B-cells display a highly specific B-Cell receptor (BCR) with affinity for one antigen only. This receptor is the membrane bound form of the immunoglobulin molecule that would be secreted by the B-cell when activated. When the BCR interacts with its antigen, it internalises the receptor-antigen complex, processes the antigen and then displays antigenic peptides in association with MHC class II at its cell surface. Through this display, the B-cell acts as an APC to helper Tcells that have been activated by the same pathogen. The T-cells then release the cytokines IL-2, IL-4, IL-10 and IL-13 which induce B-cell activation. Rarely, some B-cells can be activated without T-cell help. This only occurs in the presence of unusual antigens that can directly induce cell signalling pathways on interaction with the BCR.
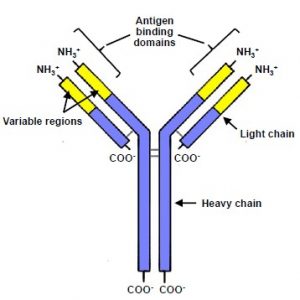
Figure 3: Structure of immunoglobulin5
Following activation, B-cells proliferate and differentiate into a large number of identical plasma and memory cells in a process known as clonal expansion. Plasma cells are responsible for the secretion of immunoglobulin whereas memory cells enter a resting state and are involved in producing a rapid, vigorous response to subsequent infection with that particular pathogen.
Immunoglobulin 1,2,4
Immunoglobulin is a protein complex that consists of two identical heavy chains and two identical light chains. At the amino-end of the molecule there is a binding site that is highly specific for its antigen. This region is called the variable region as this is where the diversity in the heavy and light chains leads to specificity to different antigens. The carboxy- terminal of the peptide is known as the constant region and is responsible for interaction with antibody receptors on neutrophils, macrophages and mast cells, further it is able to induce activation of the complement cascade.
Immunoglobulin molecules can be classified into five isoforms depending on the type of the heavy chain (Table 2)

Table 2: Immunoglobulin isoforms 4
As shown in table 2, immunoglobulin has a diverse role in the defence against both intracellular and extracellular infection. The role of IgE in the activation of mast cells, basophils and eosinophils cannot be overlooked. IgE is produced by activated B-cells. Although it is the least abundant immunoglobulin, it is vital for combatting parasitic infection. Mast cells, basophils and eosinophils are granulocytes with intracellular granules containing inflammatory mediators including histamine, leukotrienes and TNF. These cells express Fc receptors (FcR) that are specific for IgE. When the IgE: FcR complex is cross-linked by antigen, the cells degranulate to cause inflammation and assist with destruction of parasites. This process also contributes to allergy and hypersensitivity. We will discuss this further in tutorial two.
Whilst the cellular response is critical to an effective innate response, a second protein dependent mechanism known as the complement system has an equally important role.
Complement Response1,2,4
The liver produces a number of complement proteins that are involved in both innate and acquired immunity. Here, the chain of events is activated by microbial invasion and the process is known as the complement cascade. There are two main ways by which the complement system destroys microbes; opsonisation and the complement cascade.
Opsonisation describes the labelling of foreign pathogens by complement proteins. Once bound to a pathogen, complement proteins interact with specific complement receptors expressed by macrophages and induce phagocytosis. Binding of complement proteins also leads to the activation of the complement cascade. This is a more complex process of protein activation which directly induces microbial death through the formation of a membrane attack complex. This complex effectively ‘punches a hole’ to disrupt the cell wall and kill the microbe.
The complement cascade can be activated by three pathways; classical, lectin and alternative. Each pathway initially involves different complement proteins, but all lead to the hydrolysis of complement protein C3 into C3a and C3b. C3a acts as an inflammatory cytokine whilst C3b acts as an opsonin or initiator of the membrane attack pathway.
The exact stages of the complement cascade are complex and are largely outside of the scope of this tutorial, however three protein fragments within this cascade are important to consider. Complement proteins C3a, C4a and C5a work as potent inflammatory cytokines causing smooth muscle contraction, vascular permeability and degranulation of mast cells and basophils. These proteins are known as anaphylatoxins due to their ability to induce anaphylactic symptoms independent of immunoglobulin E. Anaphylaxis will be covered in greater detail in tutorial two.
SUMMARY
The immune system is a complex network of cells and proteins that are able to protect against infection. The innate response is largely brought about by phagocytes and complement proteins whereas the acquired system relies on a more specialised cellular and humoral response bought about by T and B-cells. The two systems are closely interlinked and work synergistically to assist in fighting foreign invaders.
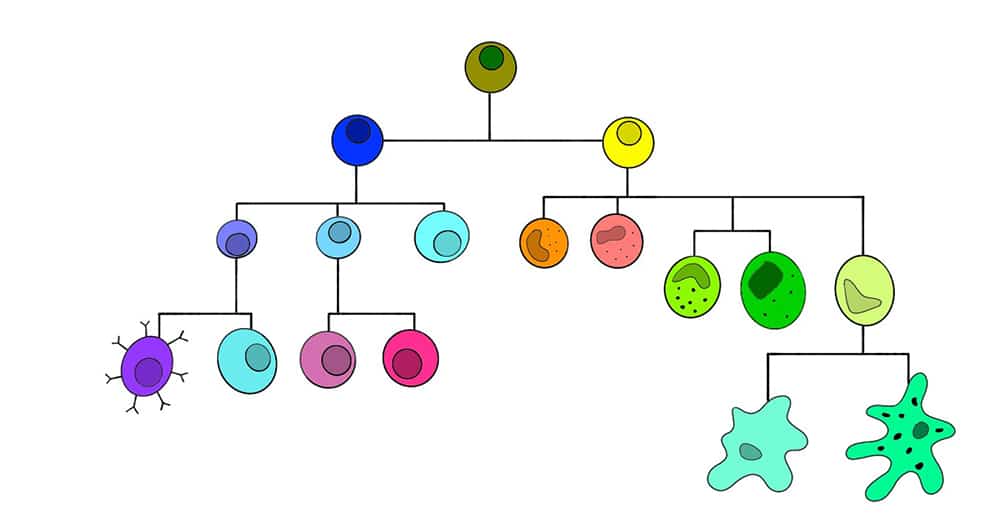
Figure 4: Cells of the immune system3
MCQ ANSWERS
- …
- True: a defect in the cell-mediated immune system can lead to the inappropriate destruction of the body’s own cells and thus the development of some autoimmune disease processes.
- True: the activation of B-lymphocytes occurs in the bone marrow and T-lymphocytes in the thymus.
- False: plasma cells are part of the humoral immune response, not the cell-mediated immune system.
- False: immunoglobulins are not involved in the cell-mediated immune response. IgA is a mucosal immunoglobulin.
- False: histamine is released by mast cells when activated by IgE, which is not part of the cell-mediated immune system.
- …
- True: B-cells are named after bone marrow, as this is where they mature. T-cells mature in the thymus.
- True: the shape of the cell receptors on the surface of the B-cells is similar to the immunoglobins that the cell then produces. This ensures that the immunoglobulin will bind to the same antigen identified by the B-cell.
- False: NK cells are part of the innate immune response, separate to the humoral response
- False: B-cells are mainly activated by T-cells. Occasionally, some antigens are able to activate B-cells directly.
- False: each individual B-cell only produces one type of antibody
- …
- False: immunogloblulin is produced by plasma cells, which are B-cells.
- True: IgA is a type of immunoglobulin that is found in mucous membranes.
- False: each immunoglobulin consists of two heavy chains and two light chains.
- False: specificity for antigens is dependant on the variable region of both heavy and light chains.
- False: IgE causes mast cell degranulation.
REFERENCES
- Murphy K, Travers P, Walport M; Immunobiology, 7th Ed. New York, Garland Science, 2008.
- Todd I, Spickett G; Lecture Notes Immunology, 6th Ed. Oxford, Wiley-Blackwell, 2010
- Todar, P. (2012). Immune Defense against Bacterial Pathogens: Innate Immunity. Available: http://textbookofbacteriology.net/innate.html Last accessed 2nd July 2015.
- Male, D; Immunology An Illustrated Outline, 3rd Ed. London, Mosby-Wolfe, 1998.
- Biochemistry Department, University of Otago. (1999). Biochemistry 111. Available: http://biocadmin.otago.ac.nz/fmi/xsl/bioc/learnbitslecture.xsl?-db=BIOCweb.fp7&-lay=Lectures&-recid=3970&-find Last accessed 2nd July 2015.



