Intensive Care Medicine
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Which of the following statements regarding the ICU management of H1N1 Viral pneumonitis in the United Kingdom is correct?
- IV Zanamivir for 5 days is first line treatment.
- Oseltamivir 75mg BD po for 5 days has been found to be adequate treatment for most patients.
- Most patients should be treated with Oseltamivir 150mg BD po/ng for 10 days.
- BIPAP has been found to be the best ventilatory mode to reverse refractory hypoxaemia in patients with respiratory failure.
- High dose systemic steroids are recommended in hypoxic patients with respiratory failure
- The following statements regarding the epidemiology, investigation, management and prognosis of patients with complicated H1N1 viral infection are correct.
- Adults over the age of 65, who smoke, are most at risk of acquiring the infection and progressing to ITU admission.
- Patients with severe disease will characteristically have a high white cell count and an elevated CRP.
- Early therapy with Oseltamivir in patients with 2009 H1N1 reduces the risk of progression to severe disease requiring ICU admission or resulting in death.
- A negative RT PCR on a BAL sample precludes a diagnosis of H1N1 viral pneumonitis.
- Increased levels of CK, thrombocytosis and metabolic alkalosis are associated with a poor prognosis.
- With regard to infection control measures:
- Surgical masks are adequate for routine care of suspected cases.
- Any patient suspected of infection should be isolated immediately.
- Personal protection should be removed in the order: gown, mask, gloves.
- NIV machines need to be switched off prior to removing from a case.
- Droplet exposure to un-immunised staff should prompt prophylactic use of oseltamivir.
INTRODUCTION
This tutorial aims to highlight the management of critically ill patients with H1N1 2009 Influenza A infection which caused the 4th worldwide pandemic in 2009 and is currently causing a seasonal epidemic in the UK and a few other countries in the Northern hemisphere including Egypt. It is written from a UK perspective but the principles translate into worldwide practice
Latest figures from the Health Protection Agency (HPA) indicate that levels of seasonal flu are continuing to increase across the UK with both community and hospital data showing a steady upward trend in the numbers of cases. GP consultation rates are now above the baseline levels in England and Wales (HPA report 23/12/10) The H1N1 2009 virus and B are the predominant circulating viruses with few sporadic A (H3N2) viruses detected in the community.
Severe cases requiring ICU/ECMO admission have been reported; as have 45 deaths, so far, associated with influenza, the majority being due to severe H1N1 2009 virus affecting the young, the pregnant, the obese and those with co-morbidities. Seemingly, this is the same Pandemic 2009 H1N1 (A (H1N1)/California/7/2009 strain that was selected for pandemic influenza vaccines worldwide.
CLINICAL BACKGROUND
H1N1 viral pneumonitis is caused by a novel variant of influenza A (A (H1N1)) which is similar to seasonal influenza but contains segments of genes from pig, bird and human influenza.
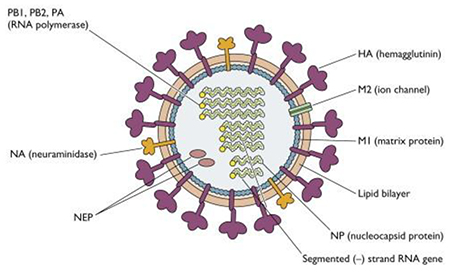
Figure 1.Structure of H1N1 virus. Diagram from http://www.virology.ws
H1N1 is an enveloped, orthomyxovirus whose 8 strands of RNA are joined together by several proteins (PB1,PB2,PA (RNA polymerase) and NP to make up the genetic material responsible for viral replication in host cells. This genetic material is protected by a matrix protein M1 which gives the virus its spherical shape. The envelope is derived from the host cell and within this are embedded glycoproteins (Neuraminidase and Haemagglutinin) which determine the type of influenza virus (A,B or C) and the subtype (A/H1N1). These antigens are the targets for antiviral drugs.
In 2009 it caused a worldwide Pandemic and much of the information we now have about its clinical manifestations, epidemiology and pathogenesis is derived from data collected by the WHO from all countries which experienced the 2009 pandemic.
Epidemiology
The overall case fatality was less than 0.5%, ranging from 0.0004 to 1.47%. Approximately 9 to 31% of hospitalised patients were admitted to an ICU where 14 to 46% of patients died. It affected young people with 32 to 45% of those hospitalised being under the age of 18yrs. People over the age of 60 were relatively spared, presumably due to having cross-protective antibodies from previous exposure to antigenically similar viruses.
In the UK from 17th July 2009 to 3 March 2010 there were 25,785 admissions to hospital with H1N1of whom 9501 were children. Of these, 2326 received critical care : 1863 adults and 463 children, with mean critical length of stay of 7.8 days in adults and 6.1 days in children and 496 ECMO bed days.
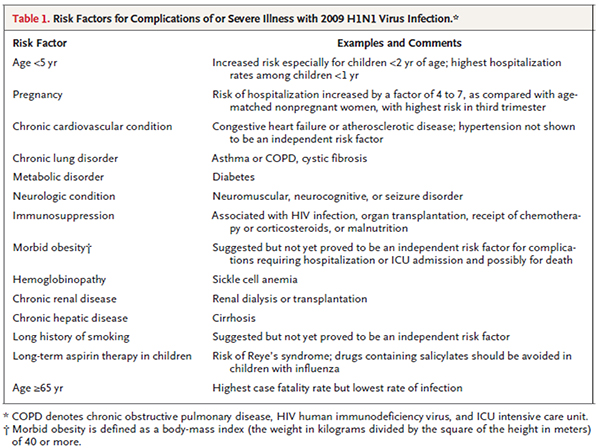
Table 1. Risk factors for complications of severe illness with 2009 H1N1 virus infection. (Reproduced with permission from NEJM 2010:362; 1708-19).
Pregnant women in the 2nd and 3rd trimester and those that are 2 weeks or less postpartum are most at risk (7 to 10% of hospitalised patients, 6 to 9% of ICU and 6 to 10% of patients who died) despite comprising 1 to 2 % of the population.
Pathogenesis
The virus binds to alpha 2,6-linked and alpha 2,3-linked receptors present in upper and distal airways down to the alveolar pneumocytes. In uncomplicated illness viral replication may be more prolonged than in ordinary seasonal influenza, such that on day 8 of the illness nasopharyngeal swabs have yielded viral RNA in 74% of patients and infectious virus in 13% of patients. Nasopharyngeal viral loads decline even more slowly in critically ill patients and viral RNA may be detected in secretions up to 28 days after onset of severe pneumonia in intubated patients and even longer in immunosuppression.
CLINICAL FEATURES AND MANAGEMENT OF SERIOUSLY ILL PATIENTS ADMITTED TO INTENSIVE CARE
Clinical Features
Most patients presenting to intensive care have had a prodromal illness lasting between 1 and 9 days prior to admission. The symptoms prior to admission have been temperature, myalgia, cough and sometimes gastrointestinal symptoms. Many patients at our institution have seen family practitioners and some have been prescribed courses of antibiotics. The principal syndrome leading to ICU admission consists of diffuse viral pneumonitis which is associated with severe hypoxaemia. There may also be ARDS, shock and sometimes renal failure. This syndrome accounted for approximately 49 to 72% of ICU admissions for 2009 H1N1 virus infection worldwide.
Rapid progression was reported to be common , starting on day 4 and 5 after illness onset. Much of the knowledge we now have about this syndrome comes from Canada where 168 critically ill patients with 2009 influenzaA (H1N1) infection were admitted in 38 adult and paediatric ICUs. They were followed up between April and August 2009. In these patients, imaging, including CXRs and CT chests, show diffuse, mixed interstitial and alveolar infiltrates.
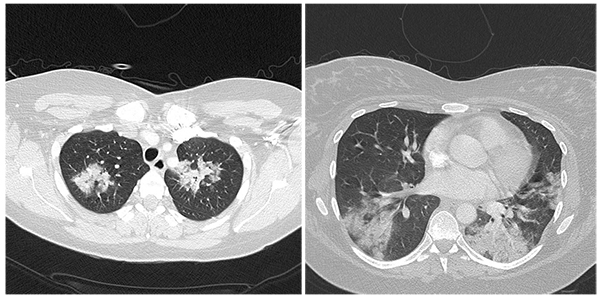
Figure 2. CT showing apical and predominantly basal infiltrates and dense consolidation in a patient with H1N1 pneumonitis.
Occasionally, multi-lobar consolidation has been found particularly in patients with bacterial co- infection. There may also be multiple areas of ground-glass opacities. Lower zones are often more affected on CT scans. Small pleural effusions have been reported.
Of all patients admitted in Canadian ICUs, the majority of whom had the above syndrome, 131 (81%), were mechanically ventilated on the first day of ICU admission, 128 (76.2%) invasively and 55 (32,7%) noninvasively. However 47 (85.4%) who initially received noninvasive ventilation ultimately required invasive ventilation. Approximately one third of patients in Canada in 2009 required vasopressor support on day 1 following ICU admission; in many cases, however, this was due to patients who needed substantial sedation to optimise ventilation due to irritable airways.
Another 1/3 of patients required advanced ventilatory support and rescue therapies for profound hypoxaemic respiratory failure, including high levels of inspired O2 &PEEP, as well as rescue therapies such as airways pressure release ventilation (APRV), high frequency oscillatory ventilation (HFOV), prone position ventilation, neuromuscular blockade, inhaled nitric oxide and extracorporeal membrane oxygenation (ECMO). Ventilatory strategies will be discussed in a little more detail later.
The other important syndromes found in patients with severe H1N1 infection include:
Prolonged exacerbation of asthma and COPD in 14 to 15% of patients.
Secondary bacterial co-infections usually by staphylococcus aureus (often MRSA), streptococcus pneumoniae, strep. pyogenes and occasionally other bacteria. Bacterial co-infections were found in 20 to 24% of ICU patients and in 26 to 38% of patients who died during the 2009 pandemic.
Exacerbations of underlying chronic diseases, e.g. congestive cardiac failure.
Neurological manifestations, including confusion, seizures, unconsciousness, acute or postinfectious encephalopathy, quadriparesis and encephalitis. Patients with influenza A associated encephalitis(IAAE) and concurrent H1N1 2009 virus infection are often reported not to have any CSF pleocytosis and may have protein levels within normal ranges. Brain MRI scans may also be normal (but could equally show nonspecific findings consistent with anoxic brain damage, inflammation, etc.) as well as are EEGs. Diagnosis is often clinical when CNS symptoms are temporally related to chest symptoms and a positive nasopharyngeal swab. Viral RNA has rarely been detected in the CSF or from brain tissue in autopsy specimens despite thorough attempts. There is now a move towards trying to look for other methods of assessing the pathogenesis of IAAE by measuring concentrations of cytokines such as Interleukin 6 (IL6) and tumour necrosis factor alpha (TNF alpha) in cerebrospinal fluid.
Myocarditis
Right ventricular dilation and dysfunction
Myositis and rhabdomyolysis with raised CK
Croup/brochiolitis in the paediatric population
Pathological features
Pathological features at autopsy include diffuse alveolar damage with hyaline membranes and septal oedema, tracheitis, necrotising bronchiolitis, pulmonary vascular congestion, alveolar haemorrhage and pulmonary thromboemboli. Bronchopneumonia with evidence of bacterial co-infection is seen in 26 to 38% of fatal cases.
Laboratory Features
Lab findings at presentation in patients with severe disease typically include normal or low-normal leukocyte counts with lymphopaenia, thrombocytopaenia, elevated serum aminotransferases, CK, creatine and LDH.
Prognostic Features
Poor prognostic indicators include increased levels of creatine kinase, creatinine, LDH, thrombocytopaenia and metabolic acidosis.
The following were found to be independent predictors of mortality in Argentina:
- High Apache 2 score
- Low PO2/FIO2 ratio
- Inotrope use
- Haemodialysis
- Prone positioning
- Pneumonic co-infection with S. penumoniae
Diagnosis
The best method of diagnosis is detection of viral RNA by real-time reverse-transcriptase-polymerase chain-reaction (RT-PCR), which is usually done on samples taken from nasopharyngeal aspirates or swabs, however endotracheal and bronchoscopic aspirates have higher yield in patients with lower respiratory tract illness. One study showed that among patients with detectable H1N1 viral RNA in bronchoscopic samples, 19% had negative upper respiratory tract samples. False negative lower respiratory samples are found in 10%.
Clinical Management
In mid 2009, the Royal College of Anesthetists, the Health Protection Agency and the Intensive Care Society in the UK working in collaboration with collegues from Mexico, the US, Canada, Australia, New Zealand , the WHO and Department of Health (UK), prepared guidelines for the management of critically ill patients admitted to the ICU with severe H1N1 infection. The same general guidelines are still recommended for use in 2010.
Drug Therapy
If there is pneumonia or evidence of clinical progression then Osetalmivir 150mg bd for 10 days without interruptions is recommended. Doses of up to 450mg bd have been used successfully in healthy adults. Intravenous Zanamivir (Relenza) is the preferred option for hospitalised patients with suspected or documented Osetalmivir-resistant 2009 H1N1 virus infection or who are unable to absorb by the enteral route. This is available off-label on a compassionate basis by the manufacturers.If early microbiologic studies do not indicate co-infection, early cessation of antibiotics may be warranted as prophylactic or prolonged courses may select resistant cases which cause problems with ventilated patients later. Nasal and throat swabs and tracheal aspirates may be co taken from ventilated ICU patients to monitor prolonged viral shedding and osetalmivir resistance. The experience of our hospital is that persistent moderate to high fever with usually negative blood cultures persists for several days after admission.
High dose steroids have no role in ARDS.
Prophylactic LMWH should be prescribed.
Ventilatory Management.
Non-Invasive ventilation (NIV) / Continuous positive airway pressure (CPAP)
There are concerns about the usefulness of NIV/CPAP in patients with H1N1. By the time HDU admission occurs, patients are often deteriorating rapidly and it tends not to be successful at avoiding intubation in a majority of patients with hypoxaemia. These patients should be intubated promptly. There are also concerns about aerosol generation promoting transmission of the virus unless patients are in a negative pressure room. There may be a role for NIV in a subset of patients whose progression is slower, or in whom it is the ceiling of care.
Invasive ventilation
Based on current evidence patients with H1N1 should be managed with lung protective ventilation as per ARDS network protocol and if targeted ventilatory goals are not met then rescue therapy should be employed. This means low tidal volume (6ml/kg) open-lung strategy ventilation with PEEP titrated based on FiO2 for goal plateau pressure <30 to 35cmH2O and SpO2 88-90%. Permissive hypercapnia can be employed to avoid higher pressures.
PEEP
High levels of PEEP are often useful in patients with severe hypoxaemia and in the H1N1 pandemic, levels of 16 to 30cm H2O were used with variable responds in terms of oxygenation. It was also noted that patients placed on high PEEP would often desaturate quite profoundly during weaning if small decrements were not used over a prolonged time. Therefore, the recommended strategy is to cautiously and slowly reduce PEEP to less than 20 before weaning FiO2 significantly.
HFOV
High frequency oscillatory ventilation (HFOV) ventilates the lung with low tidal volumes (lower than anatomical dead space) and this avoids volutrauma and barotrauma. The higher mean airway pressure with HFOV translates into higher end-expiratory volume which prevents ateletrauma. HOVF improves oxygenation and lung compliance, however potential complications include retained secretions, mucus plugging, air trapping and airway damage attributable to high gas velocities.
There is no proven mortality benefit over conventional lung protective ventilation methods; however HFOV is useful for rescuing those not responding to conventional methods and in Canada was used on 12% of the H1N1 infected patients during the 2009 pandemic
Airway pressure release ventilation (APRV)
Airway pressure release ventilation is a new mode of mechanical ventilation which is time triggered, time cycled and pressure limited and has been described as continuous positive airway pressure (CPAP) with regular, brief, intermittent releases in airway pressure. However unlike CPAP it facilitates both oxygenation and removal of carbon dioxide. The CPAP level drives oxygenation, while the timed releases aid in CO2 clearance. Spontaneous breathing is possible with APRV, improving carbon dioxide elimination and helping to prevent atelectasis.
APRV should be considered as rescue therapy in patients with persistent hypoxaemia, SpO2 < 88-90% despite conventional ventilation with low tidal volumes (4-8ml/kg)/adequate PEEP with an FiO2 > 0.8 or plateau pressure > 35 cm H2O.
It has no proven mortality benefit over conventional methods. It has proven useful in patients who are not responding to conventional methods in improving oxygenation and eliminating CO2 and improving acidosis.
APRV may not be successful in patients with H1N1 who present with COPD or asthma exacerbations as in these patients the main problem is alveolar emptying which would need to be quite rapid to prevent hyperinflation and gas trapping.
Prone Positioning
This has also been employed in patients with refractory hypoxaemia and ARDS to improve oxygenation. Proposed mechanisms of action include: alveolar recruitment, improvement in V/Q matching from redistribution of ventilation to dorsal lung regions, elimination of the heart’s compressive effects on the lung and better drainage of respiratory secretions.
Current literature has not demonstrated any mortality benefit however up to 70% of patients of patients with ALI/ARDS improve their oxygenation. It is not recommended in haemodynamically unstable patients as it may interfere with CPR. Risks include ET obstruction, loss of central vein access during the manoeuvre and risk of pressure sores. It is difficult to do in obese patients or in those receiving renal replacement therapy.
Recruitment Maneouvres (RM)
These aim to open collapsed lung units and increase functional residual capacity by increasing transpulmonary pressure. RM may be usefully employed on patients on high PEEP who desaturate rapidly on disconnection from the ventilator.
Profound hypoxaemia with normal compliant lungs will respond to moderate pressure and can produce tidal volumes greater than 700mls. In this setting high PEEP or HFO may cause alveolar distension or may worsen oxygen/haemodynamics. Profound hypoxaemia with decreased compliance needs high PEEP and APRV. IPPV duration ranges from 5 to 28 days.
Cardiovascular management
Assessment of fluid responsiveness is crucial along with quantification of right and left ventricular size and function. Inappropriate fluid administration may worsen ventricular function and oxygenation. Therefore, echocardiography along with dynamic cardiovascular monitoring such as oesophageal Doppler, LiDCO or PICCO is useful.
Extracorporeal membrane oxygenation (ECMO)
This is a specialised resource but where available referral for ECMO needs to be considered in the first week of ventilation for refractory hypoxaemia. In the UK there has been some expansion of ECMO capacity outside of the main centre in Leicester but referrals should still be made via the ECMO coordinator, based at Glenfield Hospital.
Extracorporeal Carbon dioxide removal
Devices such as the NovalungTM may be necessary where available where there is significant acidosis due to hypercarbia, or e.g. in the setting of concomitant increased intracranial pressure.
Renal management
Achievement of negative fluid balance by either diuretics or continuous ultrafiltration improves oxygenation. Renal replacement therapy may be required in 10 to 50% of cases and may be the elective mode of fluid balance, even if renal failure is not present.
Nutrition mangement
The use of high calorie feeds to avoid fluid overload is suggested.
Sedation and neurological management
Many patients with H1N1 appear to need high doses of sedation due to a combination of irritable airways and encephalopathy and/or encephalitis. Although CNS imaging and lumbar puncture have not proven very helpful in our hospital, they are generally carried out to exclude other causes of fever and delirium.
INFECTION CONTROL
Healthcare staff should avail of vaccination offered by their hospitals.
Patients with suspected H1N1 should be isolated or cohorted depending on the number of cases on the unit and taking into consideration the number of available cubicles and non-H1N1 cases. Isolation should take place for up to 7 days after illness onset or until 24hrs after resolution of fever and respiratory symptoms (whichever is longer) and for the severely immunocompromised this should be for the duration of the illness.
Use of personal protective equipment for care of patients with H1N1 infection is highly recommended as, not only will it reduce the risk of staff acquiring the infection whilst at work, but together with hand hygiene has been noted to prevent transmission within the hospital.
Aerosol generating procedures (A-GPs) like suctioning, chest physio, intubation, tracheostomy care, bronchoscopy and CPR should be performed in closed single-patient areas with minimal staff present , and operators must wear single use protective gloves, gowns, eye protection and FFP3 masks or 3M respirator masks.
Entry into a cohorted area with no contact with patients requires use of hand hygiene and a surgical mask as a minimum however close contact with a patient requires a plastic apron and gloves to be worn in addition.
Staff do not need to keep changing masks each time they move away from the cohorted areas, however they do need to remove gloves and clean as per standard infection control precautions.
Whilst the order of putting on PPE before entering a cohorted area is not particularly important as long as all the important parts are covered, it is important to remove it in a way that minimizes the potential for cross-contamination. Following an aerosol-generating procedure, before, leaving the room, remove gloves, gown and any eye protection (e.g. goggles) that may have been worn, in that order. After leaving the room, remove the respirator or surgical mask and dispose as clinical waste.

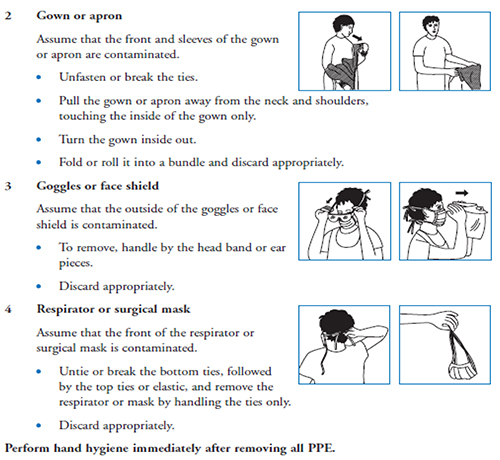
Figure 3. Correct method of donning and doffing personal protective equipment. (Adapted from the DoH UK document “Pandemic influenza: Guidance for infection control in critical care” 2008)
Respiratory care issues
- Disposable patient respiratory equipment must be used whenever possible.
- Closed systems should be used whenever possible, e.g. suction
- All respiratory equipment used on patients, including transport ventilator circuits and manual resuscitation aids, should include a high-efficiency bacteria/viral breathing system filter (BS EN 13328-1)
- The ventilator circuit should not be broken unless absolutely necessary.
- For planned circuit breaks, appropriate PPE & FFP3 respirators should be worn.
Non-invasive ventilation
- Ideally patients should be managed in a negative pressure room
- A non-vented patient mask or helmet should be used
- NIV masks should be applied to the patient’s face and secured before the ventilator is turned on
- Ventilators that function with double-hose tubing( an inspiratory and an expiratory limb) may be advantageous
- The ventilator should be turned off before removal of the close-fitting mask or when lifting the mask away from the face, e.g. for mouth care
- Water humidification should be avoided
SURGE CAPACITY AND ETHICAL ISSUES IN A PANDEMIC.
NHS emergency planning guidance suggests that UK critical care bed capacity could double during a surge and this might need to be sustained for 8 weeks or more. This will entail curtailment of elective operations, conversion of recovery, level 1 and 2 beds to level 3, and rapid training and redeployment of staff.
Once this provision is exhausted, the use of the Sequential Organ Failure Assessment (SOFA) score combined with certain inclusion and exclusion criteria (see appendix) as a triage tool has been suggested by the UK Department of Health (DoH) to ensure consistent and reproducible results.
Using this, patients with a score of >11 and the appendix criteria would not be admitted to ITU and withdrawal of care considered at 48 hours and on subsequent days for SOFA >11 or stable at 8-11.
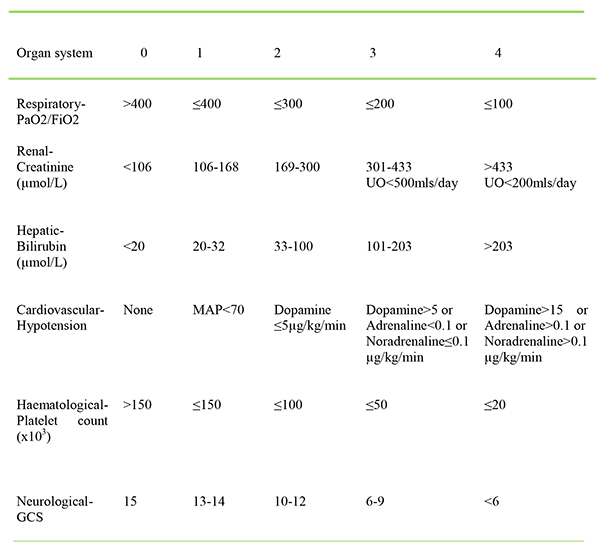
Table 3: SOFA score
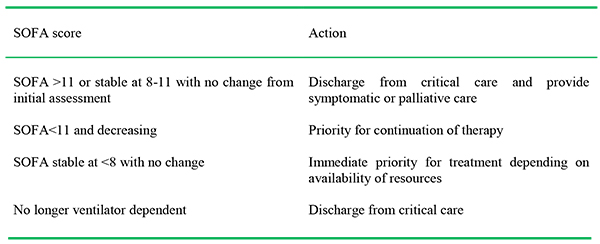
Table 4: Example of 48 hour assessment in critical care.
The 2010 report of the UK Swine flu critical care clinical group suggests that “NHS organisations further develop their local approaches to triage including the maintenance of decision-making groups with membership drawn from across clinical specialties and services provided by the organisation” and that “these groups should continue to meet and rehearse their approach to decision making in advance of any surge in activity.” There are clearly issues with using SOFA in this way. A recent retrospective survey from a UK unit which admitted several cases in 2009, showed that use of the tool in this way would have resulted in withdrawal of care in several patients who went on to survive after an acceptably short duration of ventilation.
Paediatric patients may need to be ventilated on adult units introducing further issues around training, staffing and resource allocation.
At the time of writing it is unclear, but appears unlikely that this winter’s peak, although severe, will stretch capacity to the point where such triage will be required.
APPENDIX 1:
Suggested exclusion criteria for critical care admission in a pandemic:
Severe trauma
Severe burns with two of the following: age >60,inhalational injury, >40% total body surface area affected
Cardiac arrest: unwitnessed, witnessed but unresponsive to electrical therapy, recurrent cardiac arrest
Known, severe progressive baseline cognitive impairment
Known, advanced, untreatable neuromuscular disease
Known, advanced metastatic malignant disease
Known, advanced and irreversible immunocompromise
Severe and irreversible neurological event or condition
End-stage organ failure meeting the following criteria:
Heart: New York Heart Association (NYHA) class III or IV
Lungs: COPD with FEV1 <25% predicted or baseline PaO2<7.33 kPa,
Cystic fibrosis with FEV1 <30% after bronchodilator or baseline PaO2 <7.33 kPa,
Pulmonary fibrosis with VC or TLC <60% or baseline PaO2 <7.33 kpa,
Primary pulmonary hypertension with NYHA class III or IV failure
Liver: Child-Pugh Score >7
IMPORTANT POINTS
- Remember to consider H1N1 in the differential of any persistently febrile respiratory illness and isolate or cohort.
- Once Level 2 dependency is reached, many patients deteriorate rapidly and early intubation and ventilation is recommended.
- Rescue strategies may be needed, including ECMO if available.
- This condition has a high mortality
ANSWERS TO QUESTIONS
Question 1
a. F
b. F
c. T
d. F
e. F
Question 2
a. F
b. F
c. T
d. F
e. T
Question 3
a. F
b. T, if possible
c. F
d. T
e. T
WEBLINKS
REFERENCES and FURTHER READING
- Writing Committee of the WHO Consultation Aspects of Pandemic (H1N1) 2009 Influenza. Clinical aspects of pandemic 2009 influenza A(H1N1) virus infection. NEJM 2010; 362: 1708—19
- Kumar A, Zarychanski R, Pinto R, Cook DJ, Marshall J, Lacroix J et al. Critically ill Patients with 2009 Influenza A (H1N1) Infection in Canada. JAMA 2009; 302(17) 1872—1879
- Pandemic Influenza Infection Control guidance for critical care, DH, April 2008 (available on DH website:www.dh.gov.uk/en/Publicationandstatistics/PublicationsPolicyAndGuidance/DH_ 084178)
- Clare D. Ramsey, Duane Funk, Russel Miller 111, A Kumar: Ventilator Management of hypoxaemic respiratory failure attributable to H1N1 novel swine origin influenza virus. Crit.Care Med 2010 Vol. 38(4) Suppl
- Song Mi Moon, Sung-Han Kim, Min Hee Jeong, Eun Hyee Lee, Tae-Sung Ko. Acute Encephalopathy and Pandemic (H1N1) 2009 Emerging Infections Diseases. www.cdc.gov/eid. vol.16.No.11, November 2010 1811-1813
- Luis Miguel Noriega, Renato J. Verdugo, Rafael Araos, Jose Manuel Munita, Violeta Diaz, Alejandra Marcotti et al .Pandemic Influenza A (H1N1) 2009 with neurological manifestations, a case series. 2010 Blackwell Publishing Ltd, Influenza and Respiratory Viruses, 4, 117-120



