Obstetrics Anaesthesia
KEY POINTS
- Intraoperative cell salvage is likely cost effective for pregnant women with risk factors for postpartum haemorrhage
- Intraoperative cell salvage does not increase the risk of amniotic fluid embolus, infection or coagulopathy
- Intraoperative cell salvage is accepted by some Jehovah’s Witnesses
- For Rhesus negative women, the anti-D immunoglobulin dose required should be determined with a Kleihauer-Betke test, and administered within 72 hours
INTRODUCTION
Obstetric haemorrhage is a leading cause of maternal morbidity and mortality worldwide (1). The incidence and severity of postpartum haemorrhage (PPH) is increasing; rates of PPH in Canada rose from 5.1% to 6.2% (an increase of 22%) between 2003 and 2010, while a PPH requiring a blood transfusion rose from 37 to 50.4 per 10 000 deliveries (an increase of 37%) over the same time period (1,2).
Intraoperative cell salvage (ICS) offers an alternative or adjunct to allogenic (donor) blood transfusion during an obstetric haemorrhage and is recommend in guidelines published by The Association of Anaesthetists of Great Britain and Ireland (AABGI), Obstetric Anaesthetists’ Association (OAA) and the National Institute for Health and Care Excellence (NICE) in the UK (3,4). The SALVO group’s randomized control trial found routine use of ICS during caesarean section (CS) did not significantly change the rate of postoperative blood transfusion and patients’ recovery was clinically similar (5). However, in studies where the average volume of blood returned to the patient using ICS was larger (360 mL vs. 260 mL), albeit in a smaller non-randomized study population, the postoperative haemoglobin was higher and hospital stay shorter (5,6). This suggests that ICS may be most beneficial in women with risk factors for PPH during CS. In obstetrics, ICS has been shown to be safe, beneficial and cost effective (5,7).
This tutorial aims to address the indications, contraindications, advantages, disadvantages, risks and principles of ICS in obstetrics.
INDICATIONS FOR INTRAOPERATIVE CELL SALVAGE
Indications for ICS may be liberal or restrictive but should be considered for women at risk of PPH during CS; the AAGBI, Royal College of Obstetrician and Gynaecologists and NICE define this as an estimated blood loss (EBL) greater than 1000 mL (3,4). ICS becomes most cost effective when there is a high probability of red cell transfusion e.g. a repeat CS in the setting of a known placenta praevia, severe preoperative anaemia, or known abnormal placentation (8). Women with an EBL less than 1000 mL may benefit from ICS, but to a smaller extent (5,6). Table 1. shows examples for when ICS should be considered, however usage varies depending on local protocols and resources. Severity of PPH can be difficult to predict, the potential benefit of ICS increases as risk factors accumulate.
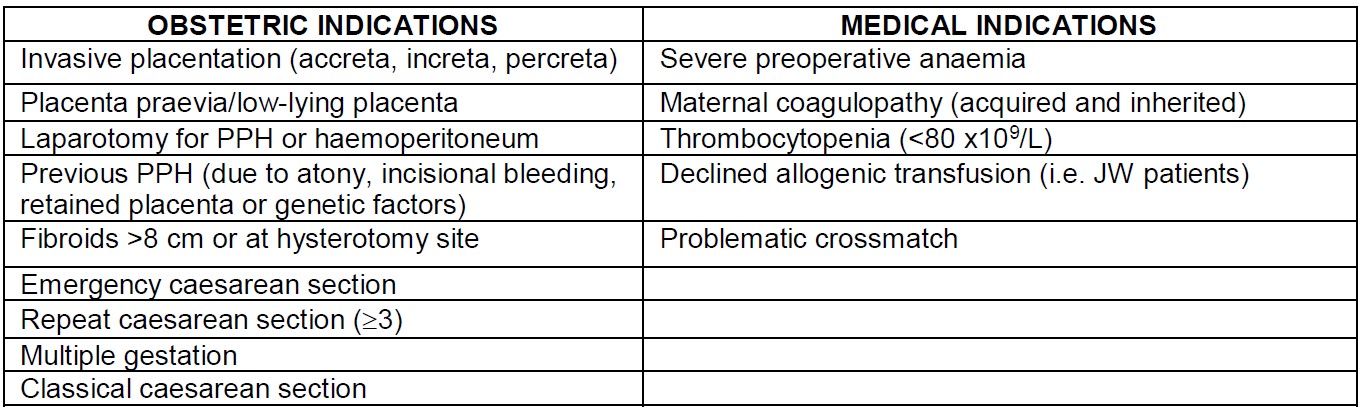
Table 1. Obstetric and medical indications for intraoperative cell salvage at caesarean section (PPH = postpartum haemorrhage, JW = Jehovah’s Witness).
CONTRAINDICATIONS FOR INTRAOPERATIVE CELL SALVAGE
- Biological contaminants
- Faeces (Note: urine and amniotic fluid are safe)
- Medical contaminants
- Haemostatic agents (Gelfoam™, Avitene™, Surgicel™, Floseal™ etc.)
- Medication not appropriate for intravenous administration
- History of heparin induced thrombocytopenia (9)
- Malignancy
- Homozygous sickle cell anaemia
Reinfusion of salvaged blood in patients with a malignancy presents a theoretical concern of disseminating malignant cells, including gestational trophoblastic neoplasms. ICS has been safely used in urologic malignancy but comprehensive safety data in obstetrics and other malignancies are lacking (10).
ICS is not recommended in women who are homozygous for sickle cell anaemia as the hypoxic conditions in the collecting reservoir can induce sickling in up to 50% of collected erythrocytes (11). In life-threatening scenarios, ICS can be considered for patients who are heterozygotes. If ICS is required for a woman with sickle cell disease an urgent blood smear should be completed on the collected blood to determine the degree of sickling prior to reinfusion (7,11).
ADVANTAGES AND DISADVANTAGES OF INTRAOPERATIVE CELL SALVAGE
Advantages
- No risk of allogenic transfusion reactions or blood borne infections
- Avoids immune-modulating effects of allogenic transfusion and associated nosocomial infection (12)
- Immediate availability of blood
- When crossmatching blood is problematic (i.e. when antibodies are present)
- Accepted by some Jehovah’s Witnesses
- Safe administration along with medications (such as uterotonics and tranexamic acid)
- Salvaged blood is more physiological than stored blood (see Table 2. below)
- Temperature, 2,3-DPG, pH and potassium are closer to maternal values (7,13,14).
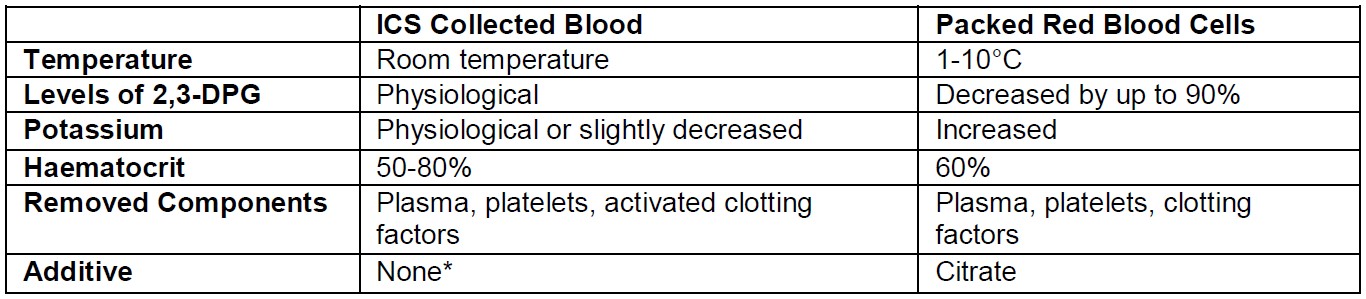
Table 2. Comparison of salvaged blood and donated red blood cells (ICS = intraoperative cell salvage, 2,3-DPG = 2,3- diphosphoglycerate). *Adequate washout will remove all but trace amounts of heparin (<10 units remaining) in reinfused blood (16)
Disadvantages
- Capital costs of cell salvage device and ongoing cost of the disposables
- Training costs
- Set-up time may limit utility during a stat caesarean section
- The volume of salvaged blood may be insufficient and the patient may need to be transfused with allogenic blood in addition to salvaged blood
Costs can be reduced by setting up the collection circuit when indicated, and only using the reinfusion circuit when required.
RISKS OF INTRAOPERATIVE CELL SALVAGE
Historically there has been concern about using ICS in pregnant women because of the theoretical risk of amniotic fluid embolus (AFE). Previously believed to be an embolic phenomenon of foetal squamous cells, AFE is now considered an anaphylactoid reaction to foetal antigens. The same levels of foetal squamous cells are present in salvaged blood as normally seen in maternal blood at the time of placental separation (13). There have been no reported cases of AFE attributed to ICS use in obstetrics (5,7,13).
Alloimmunisation occurs when a Rhesus negative woman creates antibodies to the D antigen present on Rhesus positive foetal erythrocytes. All mothers are exposed to foetal cells during delivery. Foetal cell burden should be quantified with a Kleihauer-Betke test that can then be used to determine the maternal dose of Anti-D immunoglobulin that requires administration within 72 hours of delivery.
Other risks with ICS mirror those for the general surgical population (10), which include hypotension with the use of a leukocyte depletion filter and risk of bacterial contamination. Hypotension associated with leukocyte depletion filters is rare and believed to be due to release of cytokines from filtered leukocytes. Case reports have shown that the hypotension resolves when re-infusion is stopped (13). Bacterial contamination of post-wash unfiltered and filtered samples is minimal (14), and when antibiotic prophylaxis is used, there does not appear to be an increase in infectious morbidity with ICS (15).
THE CELL SALVAGE CIRCUIT
The cell salvage circuit contains a collection reservoir, processor and reinfusion line (Figure 1). The collection reservoir can be set-up independently from the reinfusion circuit.
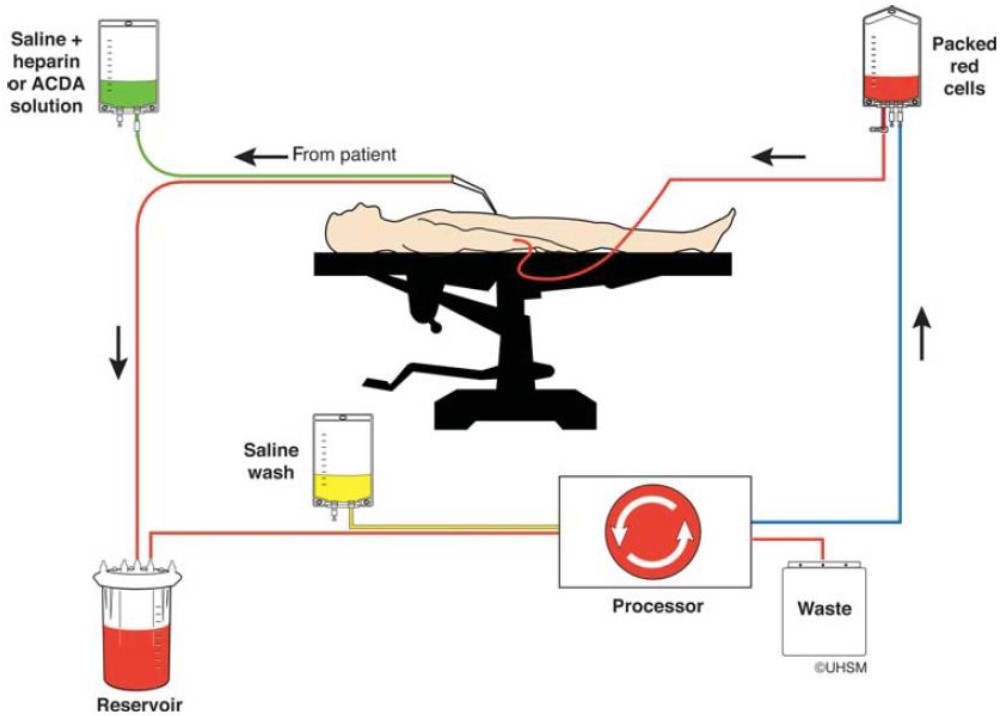
Figure 1. Intraoperative cell salvage (reproduced with permission from UK Cell Salvage Action Group). 1) The red blood cells are harvested from the operative field and/or swabs. 2) Blood is then heparinized, filtered and washed with saline then collected in a reservoir. 3) If the volume of collected blood is sufficient, the blood is then processed by differential centrifugation, suspended in saline and re-infused.
Blood is harvested from the surgical field with a large bore, low-pressure suction system to minimize cell damage. A second suction can be used prior to placental separation to reduce contamination with amniotic fluid and meconium. Bloody surgical swabs can be gently washed with isotonic saline in a sterile bowl and the fluid processed to optimize red-cell yield. Salvaged blood must be anticoagulated during collection, most commonly with heparinized saline (16).
The decision to reinfuse collected blood is based on the EBL, preoperative haemoglobin and the clinical scenario. If there is more than 500 mL of blood in the reservoir, this is usually a sufficient volume to allow processing and reinfusion (although this volume is device dependent). Reinfusion should occur within four hours of collection.
If reinfusion is considered, the salvaged blood is processed by differential centrifugation. Separation is based on particle mass; heavier erythrocytes are collected, and plasma, activated clotting factors, complement, and platelets are discarded. The blood is then washed, collected in a reinfusion bag and administered to the patient through an intravenous line (16).
Most cell salvage devices have a standard 170-200 micrometre filter. Microaggregate (40 micrometre) filters and leukocyte depletion filters are also available. They are placed between the reinfusion bag and the patient. The leukocyte depletion filter binds DNA containing cellular material to a polyethylene filter to improve removal of bacterial, malignant or amniotic contaminants. Leukocyte depletion filters theoretically decrease contamination of salvaged blood but clinical studies are lacking; they have been associated with higher rates of adverse events (5).
SUMMARY
Cell salvage is a safe and effective tool in the management of significant intraoperative blood loss at caesarean section. Its use is most cost effective and beneficial for pregnant women with risk factors for postpartum haemorrhage. The benefits are unclear in women who have lost less than 1000 mL of blood. There have been no serious adverse events reported due to ICS use in pregnant women. ICS avoids the risks associated with donor blood transfusion and helps preserve donated blood stocks.
REFERENCES AND FURTHER READING
- Knight M, Callaghan WM, Berg C et al. Trends in postpartum hemorrhage in high resource countries: a review and recommendations from the International Postpartum Hemorrhage Collaborative Group. BMC Pregnancy Childbirth 2009;9:55.
- Mehrabadi A, Liu S, Bartholomew S et al. Temporal Trends in Postpartum Hemorrhage and Severe Postpartum Hemorrhage in Canada From 2003 to 2010. J Obstet Gynaecol Can. 2014;36:21-33.
- The Association of Anaesthetists of Great Britain and Ireland, Obstetric Anaesthetists’ Association. Guideline for Obstetric Anaesthetic Services 2013. https://www.aagbi.org/sites/default/files/obstetric_anaesthetic_services_2013.pdf (accessed on 06/12/2017)
- National Institute for Health & Care Excellence. Guideline IPG144: Intraoperative blood cell salvage in obstetrics. http://www.nice.org.uk/guidance/ipg144 (accessed 26/11/2017)
- Khan, K, Moore P, Wilson M, et al. Cell Salvage and donor blood transfusion during caesarean section: A pragmatic, multicenter randomized controlled trail (SALVO). PLoS Med 14(12):e1002471. https://doi.org/10.1371/journal.pmed.1002471
- Rainaldi M, Tazzari P, Scagliarini G et al. Blood salvage during caesarean section. Br J Anaesth. 1998;80:195–8.
- Corfe J. Joint guideline on the management of intraoperative cell salvage in obstetrics. 2017. Norfolk and Norwich University Hospitals, Norwich, England. http://www.nnuh.nhs.uk/publication/download/cell-salvage-in-obstetrics-jcg0073-v2/ (accessed 10/11/2017).
- Albright C, Rouse D, Werner E. Cost savings of red cell salvage during caesarean delivery. Obstet Gynecol 2014;124:690-696.
- UK Cell Salvage Action Group. Technical factsheets and frequently asked questions (FAQ). https://www.transfusionguidelines.org/transfusion-practice/uk-cell-salvage-action-group/technical-factsheets-and-frequently-asked-questions-faq (accessed 10/11/2017)
- Kuppurao L, Wee M. Perioperative cell salvage. Cont Edu in Anaesth Crit Care Pain 2010;10:104-108.
- Brajtbord D, Johnson D, Ramsay M et al. Use of the cell saver in patients with sickle cell trait. Anesth 1989;70:878-879.
- Taylor RW, Manganaro L, O’Brien J et al. Impact of allogenic packed red blood cell transfusions on nosocomial infection rates in the critically ill patient. Crit Care Med. 2002;30:2249-2254.
- Goucher H, Wong C, Patel S et al. Cell salvage in obstetrics. Anesth Analg. 2015;121:465-468.
- Thomas D. Cell salvage in trauma. Trans Alt Trans Med. 2005;6:31-36.
- Allam J, Cox M, Yentis SM. Cell Salvage in obstetrics. Int J Obstet Anesth. 2008;17:37-45.
- Water J. Intraoperative blood recovery. 2013. ASAIO J. 2013;59:11-17.



