General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding the pharmacodynamics and mechanism of action of intrathecal (IT) opioids:
a. Opioids increase calcium entry which hyperpolarises the nerve cell.
b. Nerve cells are more likely to transmit a signal after an opioid has bound to it.
c. Fentanyl has a fast onset of action because it is highly spinal selective.
d. Morphine has a slow onset of action because it is lipophilic.
e. Fentanyl has a short duration of action because it remains fixed in the CSF. - Regarding clinical uses of intrathecal opioids
a. Fentanyl has been shown to increase the duration of motor block.
b. Intrathecal morphine is a safe and effective analgesic option for day case surgery.
c. Intrathecal morphine at doses of 100–200mcg are safe and effective in major joint replacement surgeries.
d. In thoracic surgery, intrathecal morphine should be considered a 2nd or 3rd line analgesic option.
e. High dose intrathecal morphine improves outcome in major abdominal surgery. - Adverse events with intrathecal opioids
a. Intrathecal fentanyl causes delayed respiratory depression at 6-10 hours after administration.
b. Severe respiratory depression caused by intrathecal morphine can be managed with naloxone infusion.
c. Pruritus can be treated with antihistamines.
d. 5HT3 antagonists are beneficial for treatment of IT opioid induced nausea and vomiting and has the added effect of reducing pruritus.
e. Urinary retention is a dose dependent side effect of intrathecal morphine.
Key Points
- Intrathecal opioids are useful agents in acute pain management
- Lipophilic opioids such as fentanyl have rapid onset but short duration of action
- Hydrophilic drugs like morphine have a slow onset, longer duration of action but greater risk of delayed respiratory depression
- Other common side effects include pruritus, nausea and vomiting and urinary retention
- Doses of intrathecal morphine >300mcg pose greater risk of respiratory depression
- Patients receiving intrathecal morphine should be closely monitored in the first 24 hours postoperatively
INTRODUCTION
Spinal anaesthesia is when local anaesthetic is injected intrathecally (i.e. into the cerebrospinal fluid in the subarachnoid space) to establish a spinal block and facilitate a surgical procedure. Equally, opioids can be injected intrathecally either as single agents (e.g. major abdominal surgery under general anaesthesia) or as an adjunct to local anaesthetic, producing a better quality block and prolonged analgesia post operatively. This tutorial aims to explore the benefits and potential adverse effects when opioids are administered intrathecally.
Historical Background
During the mid to late 19th century, as the world of anaesthesia was evolving, interest developed in the spinal cord as a potential analgesic target. James Leonard Corning and August Bier led the way in neuraxial blockade with the use of cocaine, while Bier successfully performed surgery under neuraxial block in Kiel in 18981.
In Paris in 1901 Nicolae Racoviceanu-Piteşti first described of the use of opioids injected intrathecally for the purpose of analgesia. However it wasn’t until the 1970s that the field really expanded. Opioid receptors were identified in the dorsal horn of the spinal cord and were subsequently proven to modulate nociceptive input. Notably in 1979 Wang described the successful use of intrathecal morphine in a cohort of eight patients with genitourinary malignancies2.
Since this time the number of reports, studies and review articles on this subject has grown steadily, reflecting the sum of our clinical experience and our ever improving neuro-pharmacological understanding of the spinal cord as an analgesic target.
PHARMACOLOGY OF INTRATHECAL OPIOIDS
Mechanism of action
The fundamental effect of an opioid binding to its receptor in the spinal cord is to decrease, or turn off, a passing nociceptive signal. While they also modulate the pain pathway in the midbrain by influencing the descending pathways, it is this signal blocking aspect that is integral to their effect. Of note there are many more opioid receptors located presynaptically than post-synaptically.
The opioid receptors (Mu, Delta and Kappa) are all G-Protein linked and they achieve their signal-reducing effect in the following ways:
a) Decrease presynaptic Ca++ entry → inhibits transmitter release
b) Increase postsynaptic efflux of K+ → hyperpolarises cell
c) Inhibit adenylate cyclase → inhibits transmitter release
Thus opioids essentially decrease release of excitatory transmitters (e.g. glutamate and substance P) and create conditions where the nerve cell is less likely to depolarise and thus transmit the nociceptive signal. Inhibitory transmitters such as glycine and gamma amino butyric acid (GABA), however, also play a role in opioid mediated analgesia, specifically by activating descending inhibitory pathways. A detailed description of the complex system of receptors and transmitters involved in pain transmission are beyond the scope of this article.
Pharmacodynamics
A thorough understanding of the pharmacodynamic properties of the various opioids, and the differences between them, informs us of their efficacy and most troublesome adverse effects. The most clinically relevant property of the drug is the degree of lipophilicity2. The table below compares the properties of fentanyl and morphine, the most commonly used agents.
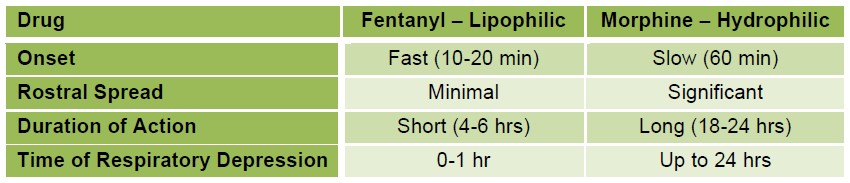
Table 1. Comparison of lipophilic and hydrophilic opioids
Once injected into the cerebrospinal fluid, lipophilic fentanyl rapidly diffuses into neuronal tissues, binding with high affinity to receptors and producing a rapid onset of action. This lipophilic agent, however, also diffuses rapidly into nonneuronal tissues such as myelin and epidural fat, causing CSF concentrations to fall quickly, shortening duration and limiting spread towards the patients head. This is in contrast to morphine, which, being hydrophilic, maintains its concentration in the CSF for longer giving a longer duration of action and more analgesic spread above the injection point. This greater spread superiorly, can be a benefit clinically as it provides a wider area of analgesic coverage. However the slower onset and prolonged action are potentially associated with delayed respiratory depression2.
Ideally, opioids injected intrathecally would display spinal selectivity i.e. provide the benefit of analgesia at the spinal level without the problems of respiratory depression at the systemic level. However, Barnards demonstrated that a large part of the analgesic effect of intrathecal opioids arises from their systemic effect, especially for the lipophilic agents3.

Table 2. Spinal Selectivity of different opioids3.
CLINICAL USES OF INTRATHECAL OPIOIDS
There are a number of indications for intrathecal opioids. We will discuss its use during the peri-operative period for acute pain management and also briefly its use in the treatment of chronic cancer pain. Of note, only preservative free opioids should be used for injection into the CSF.
Day case surgery
The most commonly used intrathecal opioid in the day case setting is fentanyl, which has a synergistic effect with local anaesthetic agents, and has been shown to improve the quality of the block and have some post-operative analgesic effects. It does not prolong the duration of block and there is no association with delayed respiratory depression. Intrathecal morphine is contraindicated in day case surgery due to the risk of delayed onset respiratory depression.
Major joint replacement
Intrathecal morphine (ITM) has been shown to be safe and effective when used as an adjunct to local anaesthetic in spinal anaesthesia for lower limb joint replacement. Studies have demonstrated that doses of 100-200 mcg improve patient satisfaction and decrease morphine PCA usage in patients undergoing total hip replacements (THR). The main clinically relevant side effect is pruritus. Doses should be adjusted for individual cases and a dose at the lower end of the range (i.e. 100mcg) should be considered in older patients. Frassanito et al have shown ITM to be better than single shot femoral nerve block at reducing pain scores and supplemental morphine usage for patients having total knee replacements (TKR). In general, the analgesic requirements after TKR are higher than THR. However many authors have advised against an ITM dose greater than 300mcg in this patient population.4, 5 At doses above 300mcg, the risk of nausea, pruritus, urinary retention and respiratory depression exceed the analgesic benefit.
Obstetric practice
The use of intrathecal morphine and fentanyl is very common in caesarean section patients. Generally the dose used is 100mcg of morphine as a single dose added to the local anaesthetic in spinal anaesthesia. Doses higher than this have resulted in higher incidence of side effects with minimal benefit in terms of quality of analgesia. Some centres utilise fentanyl as a co-adjuvant. Fentanyl has been shown to decrease the dose of local anaesthetic required and improve the quality of the block.
Urology
In urology surgery, ITM has been shown to be effective at doses of 50-300mcg, depending on the procedure. In patients undergoing transurethral resection of prostate (TURP), as little as 50mcg of ITM added to local anaesthetic for spinal anaesthesia has been shown to be effective in decreasing detrusor muscle spasm post operatively. Multiple studies have shown a decrease in supplemental analgesic requirements in the first 24 hours post operatively in patients undergoing radical retropubic prostatectomy. In nephrectomy, doses of 300 – 500mcg of intrathecal morphine have been shown to be provide effective analgesia. However, as mentioned previously the side effect profile at doses >300mcg needs to be considered.4,5 These studies have also shown an increased time to first use of supplemental analgesia.
Major laparotomy
ITM has shown a definite reduction in pain scores in patients undergoing major abdominal surgery in the first 24 hours. However, postoperative pain tends to last greater than 24 hours and the analgesic effect, and opioid sparing effect seems to be limited to the first 24 hours.6 ITM alone has not been shown to decrease hospital length of stay, but may have benefit as part of a multimodal rehabilitation regime Those that advocate the use of ITM in laparotomy generally agree that higher doses are required than in pelvic and orthopaedic lower limb surgery. Reported doses have ranged from 300-400mcg up to as high as 7-10mcg/kg body weight. However, dose finding studies have failed to definitively establish the optimal analgesic dose with the lowest serious side effect profile e.g. respiratory depression. Some recommend never exceeding 300mcg ITM for avoidance of respiratory depression but this dose is unlikely to provide effective analgesia for upper GI, hepatobiliary or major bowel surgery.
Thoracotomy
Patients undergoing thoracotomy have been shown to achieve better analgesia with the use of ITM along with IV PCA compared to PCA alone. However, thoracic epidural analgesia or paravertebral catheter seem to provide more effective analgesia over a more prolonged period of time with lower risk of respiratory depression. The decrease in the pain visual analogue scale (VAS) scores with ITM in this population does not appear to be greater than with simple analgesics such as paracetemol and NSAIDs. Therefore in thoracic surgery, ITM is recommended as a co-analgesic with paravertebral catheters or as a second line technique where an epidural is contraindicated or not technically possible.
Spinal surgery
Studies have shown ITM to be very effective in control of pain post major spinal surgery. 3-5mcg/kg as recommended by Raw et al, alongside IV PCA, has been shown to provide effective analgesia for this often painful procedure while minimising the risk of respiratory depression.7
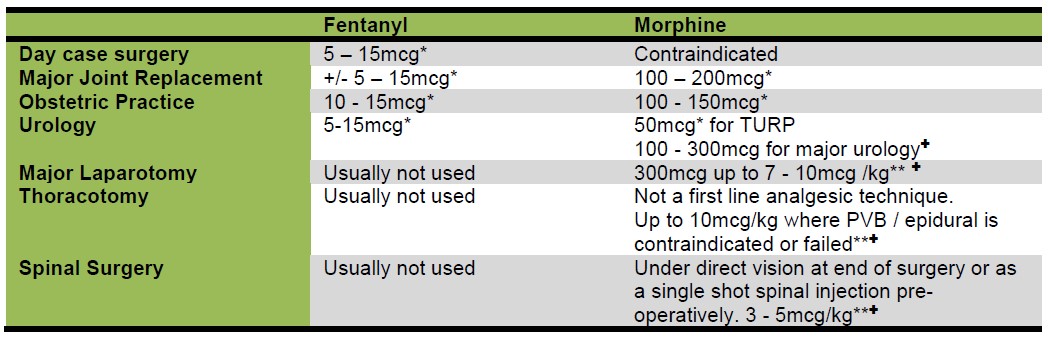
Table 3. Suggested doses of intrathecal opioids for different types of surgeries. *concomitant use of local anaesthetic for a spinal block (TURP= Transurethral resection of prostate, PVB= paravetebral block) ** Any patient receiving doses > 300mg ITM should be monitored in a high dependency setting for respiratory depression for at least 24 hours. ✚for perioperative analgesia in patients undergoing GA
Chronic Cancer Pain
According to the American Cancer Society, up to 75% of patients diagnosed with cancer suffer from pain at some stage over the course of their disease.8 This pain tends to increase over time in patients where the disease is progressive and not amenable to curative treatment. This progressive debilitating pain can lead to depression, and is often inadequately treated. Morphine is the most commonly used opioid in this setting, with hydromorphone as an alternative if morphine has been found to be ineffective or isn’t tolerated. In the setting of refractory pain, or in patients with intolerable side effects from systemic administration, intrathecal delivery can be an alternative.8 The mode of delivery is variable. Many institutions will begin treatment with a one-off intrathecal injection, or a disposable short-term intrathecal catheter. This is usually followed by a long-term tunnelled intrathecal catheter, or, if duration of treatment is expected to be prolonged, an implantable pump device which can be refilled and programmed to deliver the infusion at different rates.8 The opioid of choice is titrated to effect and individualised to the patient so guideline doses are not practical. Opioid can also be combined with a low dose of local anaesthetic agent, alpha agonists (Clonidine), GABA agonists (pregabalin) or other agents (ketamine). This however is beyond the scope of this article. Similar side effects to perioperative use can occur and are discussed in the next section. The additional risks of haematoma and or infection at the site of the pump or worse, in the subarachnoid space, are rare but need to be considered.
COMPLICATIONS OF INTRATHECAL OPIOIDS
As with all routes of administration, opioids exert beneficial analgesic effects but at the cost of potentially serious side effects. Respiratory depression and, more specifically with ITM, delayed respiratory depression are the most feared complication of intrathecal opioids. A large meta-analysis of trials of intrathecal morphine showed a high risk of respiratory depression, with an odds ratio of 7.86 and a NNH of 15.6 However, while some patients required naloxone to maintain adequate ventilation, no patients required intubation. It is worth noting that the trials assessed for respiratory depression were heterogenous, and the overall risk at doses over 300mcg is not known. Jacobsen et al demonstrated a respiratory depression incidence of 60-100% in patients receiving 1-2.5mg IT morphine. The definition of respiratory depression is not uniform across published literature, making it difficult to ascertain the true incidence.
Table 1 shows the timing of onset and duration of respiratory depression with the main IT opioids in common clinical use. This explains the reasoning behind avoiding intrathecal morphine in day case surgery. Detection of respiratory depression can be difficult as both the respiratory rate and tidal volumes can be affected to differing degrees. Some patients may have a normal respiratory rate but a rising carbon dioxide level while others may have low respiratory rate with normocarbia due to increased tidal volumes.2 Pulse oximeter monitoring will detect a desaturating patient but will not show hypercarbia. However, this effect seems to be most pronounced with intrathecal doses higher than 300mcg. It therefore seems reasonable to not exceed this dose in patients who would not otherwise have blood gas analysis carried out in the 24 hours postoperatively. Risk factors for respiratory depression include increasing age (> 65 years), use of ITM dose exceeding 300mcg, and concomitant opioid use via another route. The ASA guidelines for detection and treatment of respiratory depression due to neuraxial opioid recommend hourly monitoring for 12 hours post administration of ITM and 2 hourly for the following 12 hours.9 Following the first 24 hours the duration and frequency of monitoring should be dictated by the patient’s clinical condition.9,10 Generally, the use of routine supplemental oxygen is not recommended in the absence of hypoxia as it may mask respiratory depression and a rising carbon dioxide level.9
Pruritus is an extremely common side effect with intrathecal opioids and as with other side effects, can be prolonged with ITM. Studies have shown an incidence ranging from 5.1% to as high as 85%. Conclusive evidence for the most effective treatment is lacking. However, ondansetron has been shown to be effective, and should be considered first line. Pruritus is frequently treated with antihistamines although their usefulness for this particular cause of pruritus is questionable. Sedating anti-histamines may interrupt the itch-scratch cycle, but don’t decrease the sensation of itch. With intrathecal morphine, doses of naloxone under 2mcg/kg/hr as an infusion have been shown to be effective in treating pruritus in some studies and not affect the quality of analgesia. Sub hypnotic doses of propofol have also been shown to be effective in treating pruritus in these patients.
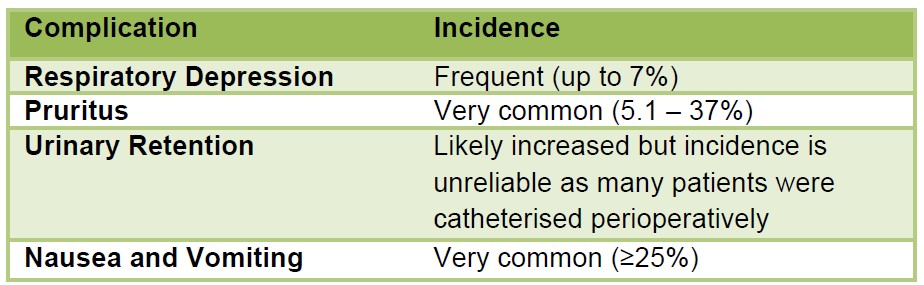
Table 4: Complications of intrathecal opioids
Urinary retention is thought to be common with intrathecal morphine but not with lipophilic opioids. A meta-analysis in the British Journal of Anaesthesia showed a slight increase in incidence with intrathecal opioid used for major surgery without local anaesthetic.6 Given the risk of detrusor muscle damage from an undiagnosed distended bladder, patients should be monitored for urinary retention and catheterised if it occurs.
Nausea and vomiting is associated with opioid use regardless of the route of administration. Studies have been equivocal about whether intrathecal opioids specifically cause an increase in incidence compared to other routes of administration.6 Lipophilic opioids such as fentanyl and sufentanil seem to be non-causative. Incidence of nausea and vomiting in ITM, on the other hand, is equivalent to that seen in systemic administration. Therefore standard treatment and prophylaxis for opioid induced nausea and vomiting are advised. The 5HT3 antagonist ondansetron may be particularly useful due to its added benefit of potentially reducing IT morphine induced pruritus.
The other side effects of sedation, delayed gastric emptying and sweating are probably no more an issue than with systemic opioids.
Some studies have advocated the use of naloxone infusion as a routine measure to reduce the incidence of respiratory depression and also of other adverse events. Further studies are required to assess if naloxone can be administered in a dose which results in a decrease in adverse events, specifically respiratory depression, without compromising the quality of analgesia. Further study may reveal this to be an effective method for allowing ward level care in patients receiving high dose intrathecal morphine. However, that evidence is not yet clear and patients who have received >300mcg ITM should be nursed in a high dependency setting, with a high staffing ratio and frequent or continuous monitoring for the first 24 hours post administration.
ANSWERS TO QUESTIONS
- Regarding the pharmacodynamics and mechanism of action of intrathecal opioids?
a. False – Binding of opioid receptors lead to a decrease in calcium influx and an increase in potassium efflux. It is the latter mechanism that causes hyperpolarisation of the cell.
b. False – Binding of opioid causes hyperpolarsation of the cell, thereby decreasing signal transmission.
c. False – Fentanyl has moderate spinal selectivity
d. False – Morphine is a hydrophilic opioid, fentanyl is lipophilic.
e. False – Fentanyl is lipophilic, allowing binding to neuronal and vascular tissue and thereby rapidly decreasing the CSF concentration. - Regarding the clinical uses of intrathecal opioids
a. False – Fentanyl improves quality of analgesia without prolonged motor block
b. False – IT morphine is considered contraindicated in day case surgery due to the risk of delayed onset respiratory depression.
c. True – 100mcg has been shown to be effective in hip replacement and doses of 150 – 200mcg in knee replacement.
d. True – Epidural or paravertebral catheters have been shown to be more effective than IT morphine in this population. Intrathecal morphine in conjunction with paravertebral catheters can be considered second line.
e. False – Improves pain scores and decreases analgesic requirements but no change in patient outcome. - Adverse events with intrathecal opioids.
a. False – Fentanyl causes early, short duration respiratory depression.
b. True – Naloxone as a bolus is short acting, so an intravenous infusion should be commenced to prevent recurrence after initial treatment with naloxone bolus in severe respiratory depression.
c. False – Anti histamines do not appear to reduce the sensation of pruritus, but cause drowsiness, thereby reducing scratching.
d. True – 5 HT3 antagonists have been shown to reduce nausea and vomiting with IT opioids, and may also help reduce pruritus.
e. False – Increases risk of urinary retention but this appears to be dose independent.
REFERENCES AND FURTHER READING
- Gorelick, P.B. and D. Zych, James Leonard Corning and the early history of spinal puncture. Neurology, 1987. 37(4): p. 672-4.
- Hindle, A., Intrathecal opioids in the management of acute postoperative pain. Continuing Education in Anaesthesia, Critical Care & Pain, 2008. 8(3): p. 81-85.
- Mugabure Bujedo, B., A clinical approach to neuraxial morphine for the treatment of postoperative pain. Pain Res Treat, 2012. 2012: p. 612145.
- Sultan, P., M.C. Gutierrez, and B. Carvalho, Neuraxial morphine and respiratory depression: finding the right balance. Drugs, 2011. 71(14): p. 1807-19.
- Gehling, M. and M. Tryba, Risks and side-effects of intrathecal morphine combined with spinal anaesthesia: a meta-analysis. Anaesthesia, 2009. 64(6): p. 643-651.
- Meylan, N., et al., Benefit and risk of intrathecal morphine without local anaesthetic in patients undergoing major surgery: meta-analysis of randomized trials. Br J Anaesth, 2009. 102(2): p. 156-67.
- Raw, D.A., J.K. Beattie, and J.M. Hunter, Anaesthesia for spinal surgery in adults. Br J Anaesth, 2003. 91(6): p. 886-904.
- Paice, J.A. and B. Ferrell, The management of cancer pain. CA Cancer J Clin, 2011. 61(3): p. 157-82.
- Practice Guidelines for the Prevention, Detection, and Management of Respiratory Depression Associated with Neuraxial Opioid Administration: An Updated Report by the American Society of Anesthesiologists Task Force on Neuraxial Opioids and the American Society of Regional Anesthesia and Pain Medicine. Anesthesiology, 2016. 124(3): p. 535-52.
- Naulty, J.S., The role of intrathecal opiates in the management of acute pain. Clin J Pain, 1989. 5 Suppl 1: p. S16-27.



