Basic Sciences
This tutorial is intended as a very basic introduction to cardiovascular physiology with particular reference to anaesthesia. Once these basic principles have been mastered then it will be appropriate to move on to the more detailed tutorials that are available.
Every anaesthetic given to a patient will have an impact on their physiology and in particular on the cardiovascular system. Therefore, understanding the physiology of the cardiovascular system allows a better appreciation of the changes that occur when an anaesthetic is given and when and how best to treat any adverse events.
Before reading this tutorial, think about the following:
- What events occur in the heart each time it beats?
- How much blood does the heart pump out?
- What factors affect the amount of blood the heart pumps out?
- How is blood pressure related to blood flow from the heart?
- What are the normal cardiovascular responses to hypovolaemia?
- How do anaesthetics affect the cardiovascular system?
ANATOMY
The heart is composed of four chambers, left atrium and ventricle and right atrium and ventricle. The atria and ventricles are separated by the atrioventricular (AV) valves, mitral on the left and tricuspid on the right. Deoxygenated blood returns from the body via the great veins (superior and inferior vena cavae) to the right atrium and then passes through the tricuspid valve into the right ventricle. From here, blood is pumped through the pulmonary valve into the pulmonary artery (the only artery which carries deoxygenated blood in an adult) and on through the pulmonary capillaries in the lungs where it is oxygenated (and carbon dioxide removed). Blood returns to the left side of the heart via the pulmonary veins (the only veins to carry oxygenated blood in the adult) into the left atrium, then through the mitral valve into the left ventricle. From the left ventricle blood is pumped through the aortic valve into the aorta and then via the systemic vascular tree to the body’s organs.
The vascular tree is comprised of arteries, arterioles, capillaries, venules and veins, conventionally described in progressive order leaving from the left side of the heart and returning to the right. The arterial side of the circulation carries oxygenated blood. Both arteries and arterioles have thick, muscular walls as they carry blood under relatively high pressure. The average adult has a circulating volume of approximately 5000ml blood. In the normal resting state only about 15% (750ml) of the circulating volume is within the arterial system. As blood traverses capillary beds the pressure falls and the blood gives up oxygen and other nutrients to the tissues, while at the same time collecting carbon dioxide and other waste products of metabolism. The blood, now relatively deoxygenated starts its return journey to the heart in the venules and veins (thin-walled because of the low pressure), finally entering the vena cavae. The venous system contains approximately 60% (3000ml) of the blood volume and is often referred to as a capacitance system, the volume of which can be varied significantly by the sympathetic nervous system (see below). The remaining 25% (1250ml) of the blood volume is in the pulmonary circulation and heart.
THE CARDIAC CYCLE
The cardiac cycle refers to the mechanical events that occur during the contraction (systole) and relaxation (diastole) of the ventricular muscle. It must be remembered that this activity is initiated by the cardiac action potential that originates in the sino-atrial (SA) node, spreads through the atrial muscle, crosses the atrio-ventricular (AV) node, reaches the ventricles via the bundle of His and supplies the Purkinje fibres which innervate the ventricles. The sum of these action potentials is recorded as the ECG;
- P wave – atrial depolarisation
- PR interval – spread of excitation through the atria, AV node and bundle of His
- QRS complex – spread of excitation through the ventricles
- T wave – ventricular repolarisation
There are two important points to remember:
- Mechanical contraction occurs after depolarisation, therefore systole starts at the end of the QRS complex and ends during the T wave.
- A cardiac action potential or ECG signal does not mean that the heart is pumping blood, it only indicates electrical activity (remember the cardiac arrest patient with pulseless electrical activity (PEA).
Systole is the period of ventricular contraction. As contraction starts in both ventricles, the AV valves close to prevent back flow of blood into the atria. Ventricular contraction continues with a rapid increase in pressure but no change in volume; this is called isovolumetric contraction (meaning literally “same volume”). Eventually the pressure within the left and right ventricles exceeds the pressures in the aorta and pulmonary arteries respectively and at this point the aortic and pulmonary valves open and ejection of blood occurs. The amount of blood ejected in one cycle is referred to as the stroke volume (SV) and this is around 70ml in an average adult at rest. However, the ventricles do not completely empty, only sixty to eighty percent of the blood present in the ventricle is ejected (the ejection fraction). As the ventricles empty, the pressure within them starts to fall. When the pressure drops below that in the aorta and pulmonary artery, the aortic and pulmonary valves respectively close, signalling the end of systole.
Diastole is the period of ventricular relaxation. Initially there is a period of isovolumetric relaxation (again “same volume”) and all the valves are closed. As the atria fill with blood returning to the heart the pressure rises, when it exceeds that in the ventricles the AV valves open and as a period of passive filling occurs the volume of blood in the ventricles starts to increase. This passive filling is initially rapid, but slows as the pressure gradient across the AV valves decreases. Ventricular filling is completed by contraction of the atria, contributing twenty to thirty percent of ventricular volume, and signalling the end of diastole. The volume of blood in the ventricle at this point is often referred to as the end-diastolic volume (EDV) and is normally around 120ml.
It is interesting to consider how long each of these components of the cycle takes as heart rate varies. Under normal resting conditions, heart rate is approximately 70 beats/min and each cardiac cycle therefore takes approximately 0.85 sec. Systole lasts 0.3 sec and diastole lasts 0.55 sec, most of the time being taken up by ventricular filling. Now consider what happens when the heart rate is 180 beats/min; each cycle takes up 0.3 sec, with diastole and systole both lasting 0.15 sec. Diastole has been reduced more than systole (0.4sec compared to 0.15sec), reducing the time for ventricular filling. Systole cannot be reduced any more without affecting the stroke volume. An increase in heart rate beyond this rate reduces diastole further resulting in insufficient time for ventricular filling and a reduction in the volume of blood pumped out with each beat. Therefore for most adults, the maximum heart rate is around 180beats/min. In addition, perfusion of the muscles of the ventricles (via the coronary arteries) occurs predominantly during diastole. Due to this, at very high heart rates, the duration of coronary blood flow is reduced. This is the very time when the heart is working maximally and so has a high oxygen demand risking myocardial ischaemia (inadequate oxygen supply to cardiac muscle).
CARDIAC OUTPUT
So far we have defined the amount of blood ejected by each heart beat as the stroke volume. However, it is the constant flow of blood that is more important and this is referred to as the “cardiac output”. Cardiac output (CO) is defined as the volume of blood ejected by each ventricle per minute and is the product of the stroke volume (SV) and the heart rate (beats/min), it is expressed in litres/min. Clearly the output of both ventricles has to be the same, otherwise all the blood would end up in either the systemic or pulmonary circulation!
Cardiac Output = Stroke Volume x Heart Rate
Under normal resting conditions this is approximately 70ml x 70beats/min = 4900ml/min (around 5l/min). The stroke volume is conventionally only that of one ventricle and hence cardiac output is also conventionally that of one ventricle. The true volume of blood passing through the both sides of the heart is however double this figure. Due to fact that different sized patients will have different cardiac outputs, to allow meaningful comparisons to be made the cardiac index (CI) is often used. This relates CO to body surface area and is expressed in litres/min/m2. It is calculated by dividing CO by body surface area.
Cardiac Index = Cardiac Output/Body surface area
FACTORS AFFECTING CARDIAC OUTPUT
From the formula above it is obvious that only two factors affect cardiac output;
- Stroke volume
- Heart rate
1. Factors affecting stroke volume
There are three principle determinants of stroke volume:
- the degree of filling of the ventricle, or “preload”
- the contractility of the myocardium
- the resistance against which the ventricle has to work, or “afterload”.
Preload
The more a muscle fibre is stretched before being stimulated to contract, the greater its force of contraction. This property is however limited by the internal molecular structure of muscle such that above a critical (optimal) point further lengthening reduces force of contraction. This property is often referred to as the FrankStarling mechanism and applies equally to cardiac muscle. Blood returning to the heart during diastole stretches the ventricular muscle fibres. The greater the volume in the ventricle, the greater the force (or more correctly velocity) of contraction. Preload is therefore directly related to the end diastolic volume (or end diastolic pressure as the two vary together) and providing myocardial contractility and afterload remain constant, increasing preload will increase stroke volume. However, as stated above, beyond a certain point contractility falls and the clinical state of cardiac failure occurs (Fig 1.)
In clinical practice it is difficult to measure end diastolic volume and so we estimate this; central venous pressure (CVP) gives an estimate of the right ventricular EDV, pulmonary artery occlusion pressure (PAOP, sometimes called the “wedge pressure”) gives an estimate of the left ventricular EDV (clinically the side we are more often interested in).
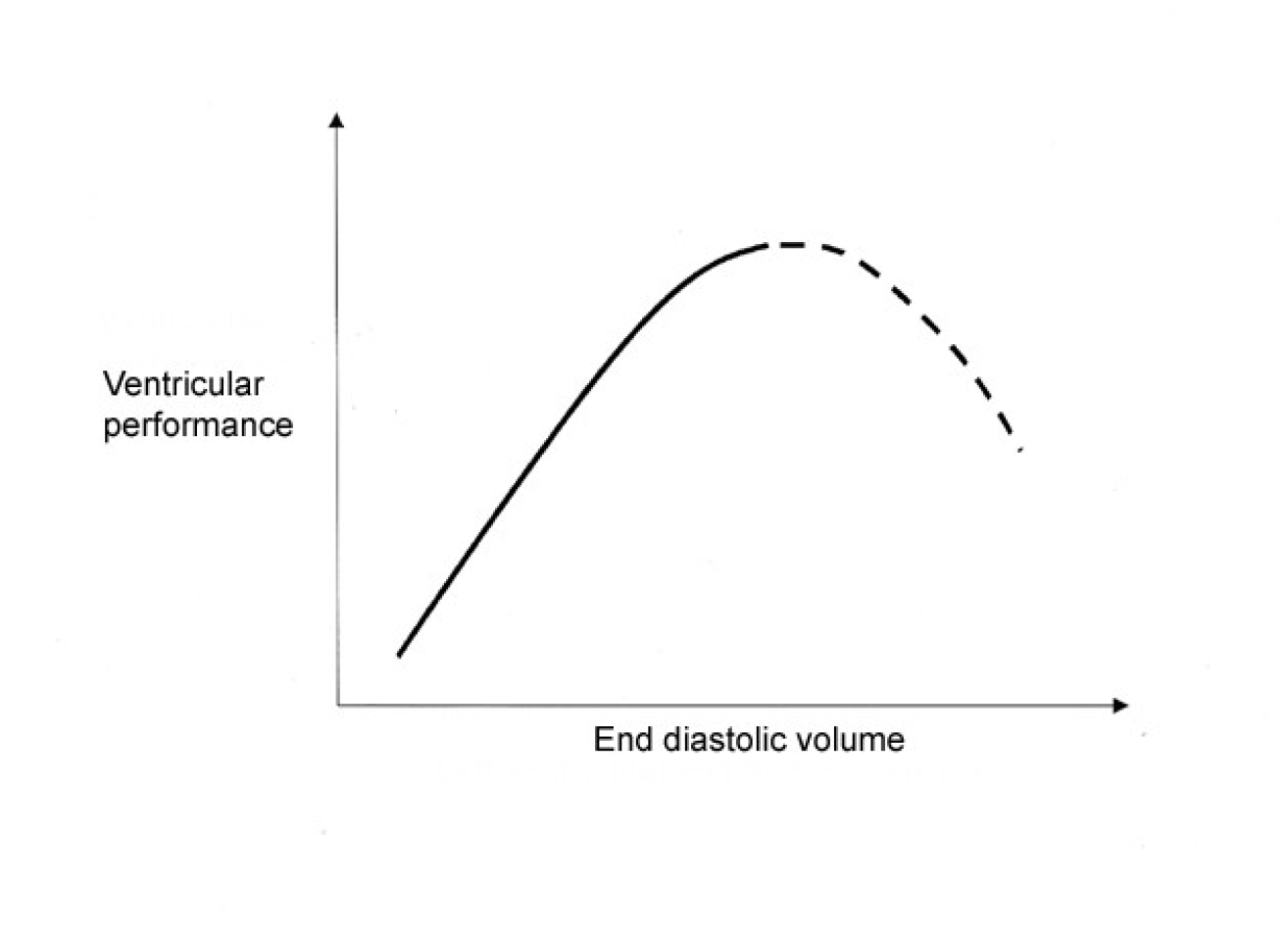
Figure. 1: Frank-Starling Curve
Myocardial contractility
This refers to the intrinsic ability of the cardiac muscle fibres to contract and is independent of the degree of preload and afterload. This is often referred to as the degree of inotropy and substances affecting this property are called inotropes; those increasing rate and force of contractility have a positive inotropic action, those that decrease contractility have a negative inotropic action. The most important determinant of contractility is the sympathetic nervous system. It acts directly, or via the release of catecholamines from the adrenal gland, stimulating adrenergic receptors (i.e. they respond to adrenaline, see below) which results in a positive inotropic effect (Fig 2.). Many factors cause a decrease in contractility i.e. are negatively inotropic; examples would be an acidosis, hypoxia, hypocalcaemia, and many drugs (particularly anaesthetics and antiarrhythmic drugs).
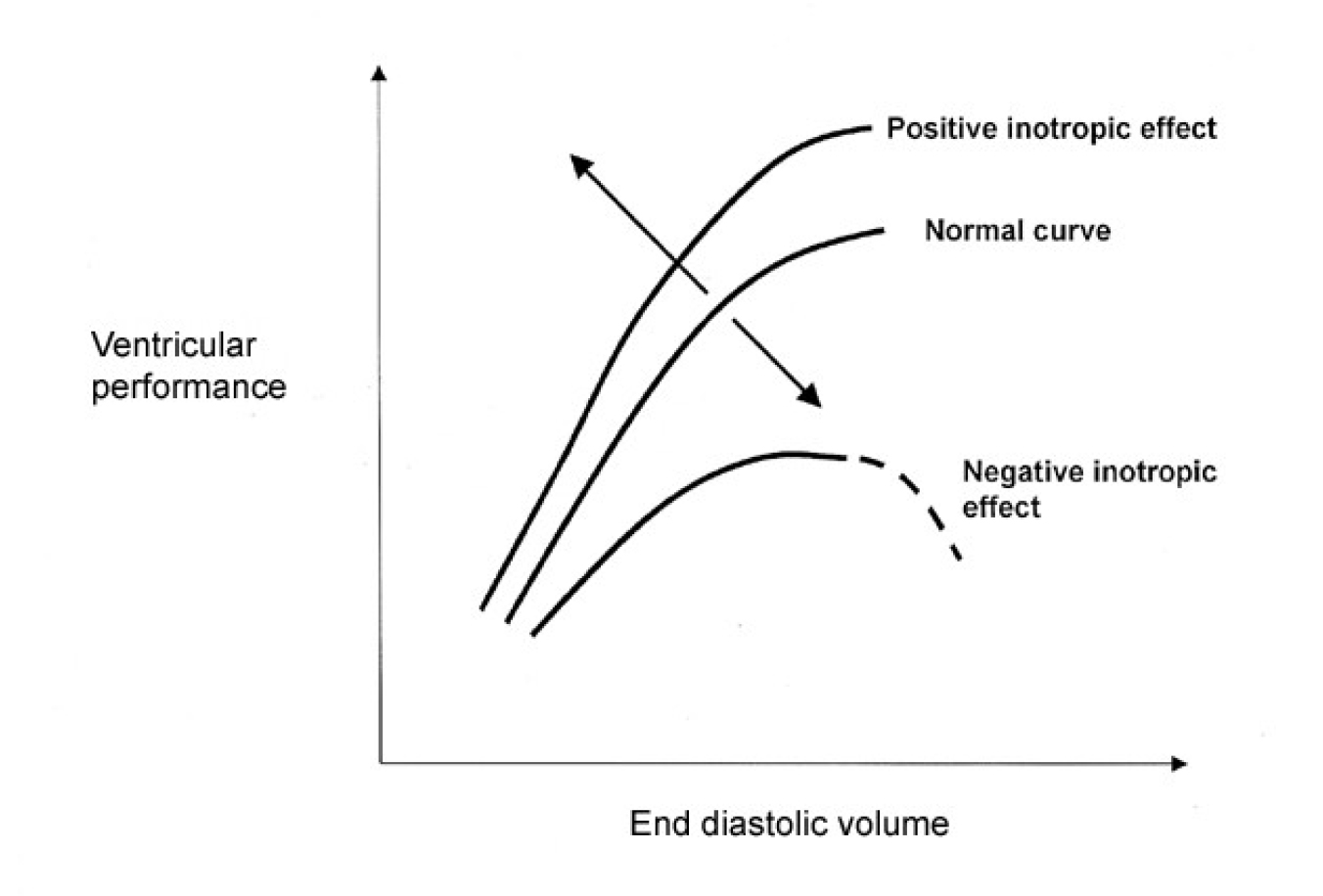
Figure 2.
Afterload
At the end of diastole, the ventricular muscle starts to contract. In order to achieve this it has to overcome those forces that are preventing it, namely the tension in the ventricular wall itself and the resistance offered to the ejection of blood from the ventricle. Clearly this will be very different for both ventricles. Measurement of true afterload is difficult and usually approximated as follows:
- Left ventricular afterload is the resistance offered by the systemic circulation and termed the systemic vascular resistance (SVR).
- Right ventricular afterload is the resistance offered by the pulmonary circulation and termed the pulmonary vascular resistance (PVR).
In health, afterload is determined predominantly by vascular tone, or the degree of vasoconstriction (or dilatation) of the arteries and arterioles. The greater the degree of constriction, the smaller the vascular lumen and the greater the resistance offered to ejection of blood from the ventricle. As a result, for a given preload and contractility, less blood will be ejected, the end systolic volume is increased and stroke volume falls. Vascular tone is controlled by the sympathetic nervous system and by the release of catecholamines, again acting on adrenergic receptors.
2. Factors affecting the heart rate
The heart has an intrinsic pacemaker, the sinoatrial node, which in the absence of any other influence discharges at around 100 beats/min. Changes in heart rate are brought about by the autonomic nervous system, either directly or via effects on the adrenal glands. Generally speaking, sympathetic stimulation will increase the heart rate via adrenergic receptors (a positive chronotropic effect) and parasympathetic stimulation, via the vagus nerve, will decrease the heart rate (a negative chronotropic effect). Under normal circumstances, resting heart rate is below 100 beats/min, and therefore there is dominance of vagal (parasympathetic) activity. Providing stroke volume is unchanged, an increase in heart rate will cause a rise in cardiac output and vice versa (CO=SV x HR). This is true in normal healthy individuals as the stroke volume is relatively unaffected between heart rates of 50-150/min. As explained above, at heart rates much greater than this, stroke volume eventually falls along with cardiac output. At lower heart rates, ventricular filling can increase to compensate, but the point is eventually reached where filling (EDV) is maximal and with a further reduction in rate once again cardiac output will fall. For example, if the maximal stroke volume is 120ml, cardiac output will fall once the heart rate drops below 40beats/min.
ADRENERGIC RECEPTORS AND THEIR ACTIONS
These receptors respond to stimulation by the sympathetic nervous system and catecholamines from the adrenal gland. Their primary function is to prepare the body for the primitive “fight or flight” response; ventilation increases along with bronchodilation, cardiac output is increased through an increase in rate and contractility and blood is diverted from non-vital organs (e.g. the gut) to vital organs (e.g. heart, kidneys and muscle). There are two main types of adrenergic receptors alpha (α) and beta (β), each of which is divided into two subtypes, α1, α-2, and β-1, β-2. Their location, and actions in the cardiovascular system when stimulated, is shown in table 1.

Table 1. Adrenergic receptors and their effects
BLOOD PRESSURE
So far we have concentrated on blood flow from the heart (the cardiac output), however, in day-to-day practice we rarely measure flow (it is technically difficult to measure flow accurately) but frequently measure blood pressure. It is important to understand how the two are related as pressure does not equate to flow; patients can have a “normal” blood pressure with much reduced flow and vice versa. When measuring systemic arterial pressure a number of different figures can be calculated.
- Systolic pressure – the maximum pressure generated, normally around 120mmHg
- Diastolic pressure – the minimum pressure generated, normally around 80mmHg
- Pulse pressure – the difference between systolic and diastolic, normally 40mmHg
- Mean pressure – the average over each complete cardiac cycle. This can be approximated to diastolic pressure plus one third of the pulse pressure and is normally around 90-100mmHg.
Blood pressure comes about as a result of the heart pumping blood into the arterial system, which is of relatively small capacity containing only about 15% of the circulating volume. If the volume of the arterial system changes for a given cardiac output, the pressure will change; a fall in volume of the arterial system results in an increased pressure and vice versa. It is not possible to measure the volume of the arterial system but it is related to the radius of the blood vessels; as they constrict, their radius and volume will decrease. As the radius decreases the resistance to flow increases such that if the radius is halved resistance to flow increases approximately 16-fold (strictly speaking this is only true of certain types of fluids flowing in a particular way, but it gives an indication of the magnitude of the effect). Therefore as the volume of the arterial system decreases the resistance to flow through it increases dramatically. As resistance can also be expressed in terms of pressure drop per unit flow (relating to Ohm’s law V=IR):
Resistance of the circulation = blood pressure/cardiac output
Therefore:
Blood pressure = cardiac output (CO) x systemic vascular resistance (SVR)
By looking at this equation we can immediately see that in a patient with a very low cardiac output, blood pressure can be maintained simply by the body increasing the SVR.
One might ask “why do we need a blood pressure?”. After all, it is flow of blood that is important and if the SVR was low, the heart could pump blood easily. The answer lies in the organs the blood perfuses. The kidneys require an adequate pressure to ensure that the glomeruli can filter and all organs require a pressure gradient between the arteries and veins to ensure adequate flow through their vast capillary beds. Low or no pressure would and does result in organ failure!
Under normal circumstances, blood pressure is tightly controlled by:
- Sympathetic and parasympathetic nerves
- Hormonal systems; adrenaline, noradrenaline, renin, angiotensin, antidiuretic hormone (ADH, vasopressin), aldosterone and atrial naturietic factor (ANF).
Other factors can also influence blood pressure, initiating either a direct response or a response via the above mechanisms, for example sepsis and metabolic acidosis.
These systems work to maintain an adequate perfusion pressure and blood flow to the vital organs and tissues of the body. Consider what happens in clinical practice – a typical case might be a patient who has lost 1.5-2l blood as a result of trauma and is continuing to bleed.
- The decrease in circulating volume will reduce venous return and preload. This causes a fall in EDV, SV, CO and finally blood pressure.
- The fall in blood pressure is sensed by receptors (baroreceptors) located in the carotid body, the arch of the aorta and within the heart that send impulses to the vasomotor centre in the medulla.
- In response, the vasomotor centre increases sympathetic outflow to the heart, blood vessels and adrenal gland, and decreases parasympathetic outflow to the heart.
- As a result, heart rate and force of contraction increase to try and restore CO, the arterioles constrict to increase the SVR and the large veins constrict to divert blood to the central circulation (remember 60% of blood volume is normally within the veins). The aim is to restore arterial blood pressure to a value that maintains adequate organ perfusion. Initially SVR will increase to a greater extent than CO, hence we may have a “normal” blood pressure but reduced CO.
- The sympathetic activity will also constrict vessels of non-essential organs e.g. the skin and gut to divert blood to vital organs e.g. brain and heart.
- ADH is released from the pituitary, which increases reabsorption of water and sodium in the kidneys to help maintain the circulating volume and induces further vasoconstriction.
- As a result of decreased renal blood flow and the sympathetic activity, the renin-angiotensin-aldosterone system is activated, further increasing retention of water and electrolytes with angiotensin II acting as a vasoconstrictor.
- If there is overshoot most of the above systems have a negative feedback loop and stretching of the atria will stimulate release of ANF causing salt and water loss via the kidneys.
HOW DOES THIS MANIFEST CLINICALLY?
Our hypovolaemic trauma patient will have a number of signs and symptoms as a result of the above. The first signs will be as a result of the increase in sympathetic activity; the patient will have a tachycardia, be vasoconstricted (cool, pale peripheries, increased capillary refill time, slightly increased perspiration) and the diastolic blood pressure may be elevated (increased SVR). They may have an increased respiratory rate (tachypnoea). Eventually if untreated, the tachycardia and tachypnoea will increase, and as venous return falls and the SVR cannot compensate, cardiac output falls the systolic blood pressure will decrease. The diastolic blood pressure may not be measurable. Urine output will decrease due to activation of the renin-angiotensinaldosterone system, stimulated by a reduced blood pressure across the renal capillary bed. Finally consciousness is impaired as blood flow to the brain falls. Not surprisingly, the key principle in managing these patients is resuscitation of the circulation to maintain an adequate blood pressure (not necessarily normal) for organ perfusion along with haemorrhage control.
HOW IS ALL THIS RELEVANT TO ANAESTHESIA?
General anaesthesia
- Almost all anaesthetic drugs have important actions on the cardiovascular system:
- Vasodilators – decreasing both preload and afterload
- Negatively inotropic – reducing cardiac contractility
Not surprisingly, induction and maintenance of anaesthesia is usually accompanied by a fall in the patient’s blood pressure; cardiac output falls due to decreased preload and myocardial contractility and the reduced afterload decreases systemic vascular resistance. The situation may be worsened by the fact that some drugs also depress the baroreceptor reflex to a varying degree and so there may not be a compensatory tachycardia. Finally, the anaesthetics also depress the medulla reducing the sympathetic response to the fall in blood pressure. Fortunately, most healthy adults will tolerate a fall in blood pressure of approximately 20% and once surgery starts this increases sympathetic activity and blood pressure is restored. If the blood pressure remains low, then treatment may be required. From the information above we can now formulate a logical method to restore the blood pressure and ensure an adequate blood flow to the various organs.
Increase preload. In an emergency, the patient’s legs can be elevated to augment venous return and preload. However, it is more usual to give a bolus of fluid intravenously, 10ml/kg initially and monitor the response. Increasing preload increases cardiac output and in turn, systemic blood pressure. Remember, continuing to give fluid will eventually cause over-stretching of the myocardial muscles fibres and reduce the force of contraction (Fig 1.) leading to cardiac failure (although in practice this is hard to achieve in young, fit adults).
Increase afterload. Once preload has been addressed, then vasopressor drugs can be used. These increase the systemic vascular resistance by acting as agonists (stimulating) the alpha adrenergic receptors (table 2). Drugs that act in this way include phenylephrine, metaraminol and noradrenaline (norepinephrine). Care must be taken as the baroreceptor reflex is preserved and profound bradycardia may occur. Ephedrine has weak alpha and beta-1 agonist effects. Adrenaline (epinephrine) is usually only used if other drugs have been ineffective or are unavailable. It is an agonist at both alpha and beta adrenergic receptors and the effects are dose-dependant; at low doses it is predominantly a beta (1 & 2) agonist and so whilst the increased heart rate and force of contraction (beta-1 effect) may improve cardiac output, SVR may fall due to the beta-2 effects. With increasing doses, the alpha agonist effect dominates peripherally and SVR is increased.
Remember, increasing SVR alone may increase blood pressure, but at the expense of a reduction in stroke volume and cardiac output.
Increase contractility. If myocardial depression with reduced contractility is contributing to hypotension, then it would be sensible to use a drug with an inotropic action. Dobutamine has beta-1 agonist actions increasing contractility and heart rate. Dopamine, the biological precursor of noradrenaline, has alpha and beta-1 agonist actions with the latter predominating at low doses and both effects at higher doses.
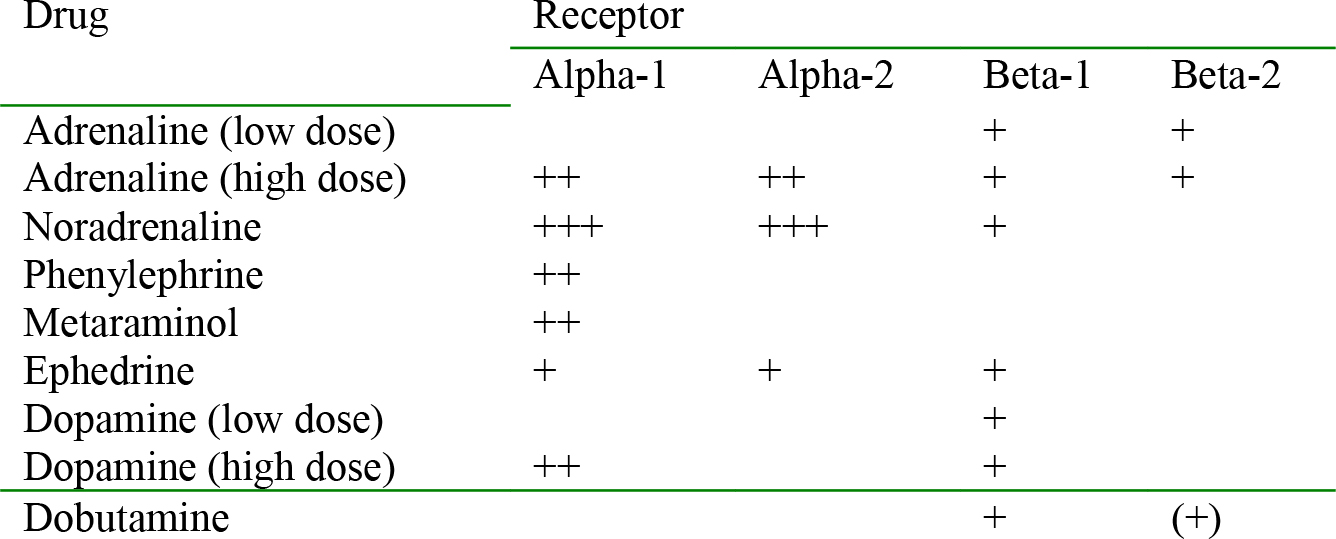
Table 2. Drugs and their actions at adrenergic receptors
Regional anaesthesia (spinal or epidural)
These techniques result in vasodilatation (reduced SVR) proportional to the height of the block. As the local anaesthetic drug spreads cranially, there is an increasing block of the sympathetic nerves leaving the spinal cord (the sympathetic chain) that supply the vascular beds. If cardiac output is maintained there will be a slight fall in blood pressure (remember, BP=CO x SVR). If however the cardiac output also falls, due to reduced preload (failure to maintain an adequate venous return), then blood pressure will fall significantly. This may be compounded further if the block extends to reach above the level of T5 (anaesthesia above the nipple) as the sympathetic supply to the heart will be reduced and result in a bradycardia (due to unopposed parasympathetic activity) and profound hypotension.
Mild hypotension. Give intravenous fluid bolus, 10-20ml/kg, to maintain preload and cardiac output. Alternatively, an alpha agonist could be used to counteract the vasodilatation eg phenylephrine or metaraminol. Care must be taken if these drugs are used as the baroreceptor reflex is preserved and the increase in blood pressure may be accompanied by a bradycardia. However, ephedrine is often used as the first line drug, despite the fact that its main effects are due to its beta-1 agonist action increasing heart rate.
Although theoretically a head-down position will increase venous return (preload), it may also encourage the spread of the block and cause further vasodilatation. Care must be taken if this technique is employed.
Moderate to severe hypotension. This is usually a result of a high block accompanied by a bradycardia. In addition to adequate fluid volume to maintain preload, atropine will be required (0.5mg iv). Although metaraminol or phenylephrine will cause vasoconstriction, the problem of a reflex bradycardia remains. Hypotension and bradycardia that does not respond to the above measures should be treated with small bolus doses (10micrograms) of adrenaline.
CONCLUSION
The key to treating the changes that occur in a patient’s cardiovascular status as a result of giving them an anaesthetic is understanding the underlying physiology and how this has been disturbed. This will then allow disturbances to be treated in the safest and most effective manner, with little potential for harm to the patient.



