Basic Sciences
Introduction
Local anaesthetic drugs are used widely for the provision of anaesthesia and analgesia both intra- and post-operatively. Understanding the pharmacology of these agents as a group, as well as the differences between specific drugs, enables the anaesthetist to use them safely to maximum effect. This tutorial focuses on the basic structure and function of local anaesthetics. Learning will be improved by trying to answer the questions posed in the text before moving on.
More detail can be found in the “Further reading” section at the end.
Definition of a local anaesthetic
A local anaesthetic can be defined as a drug which reversibly prevents transmission of the nerve impulse in the region to which it is applied, without affecting consciousness. There are many drugs which exert local anaesthetic activity in addition to their main clinical uses, but this tutorial will focus on those drugs which are principally used for their local anaesthetic properties.
The structural classification of local anaesthetics
Local anaesthetics generally have a lipid-soluble hydrophobic aromatic group and a charged, hydrophilic amide group. The bond between these two groups determines the class of the drug, and may be amide or ester. Examples of amides include lignocaine, bupivacaine and prilocaine. Examples of esters include cocaine and amethocaine.
The clinically significant differences between esters and amides
The ester linkage is more easily broken than the amide bond so the ester drugs are less stable in solution and cannot be stored for as long as amides. Amide anaesthetics are also heat-stable and can therefore be autoclaved; esters cannot.
The metabolism of most esters results in the production of para-aminobenzoate (PABA) which is associated with allergic reaction. Amides, in contrast, very rarely cause allergic phenomena. For these reasons amides are now more commonly used than esters.
Local anaesthetics as isomers
Local anaesthetics may also be considered in terms of their stereoisomerism. This term describes the existence of molecules with the same molecular and structural formula, but different spatial orientation around a particular atom, the chiral centre. This is like the right and left foot being mirror images of each other. Stereoisomerism occurs in the case of bupivacaine which has two stereoisomers, known as R and S forms, and also in the case of prilocaine. The combination of equal amounts of the two stereoisomers of a particular drug is known as a racemic mixture.
Why might this isomerism be important?
The different arrangements of the R and S forms of bupivacaine are thought to be associated with differences in potency and side-effect profile. This is easy to understand if you were to try and put your right foot in your left shoe – it doesn’t work as well and causes side effects (pain)! This is the reason why more drugs are being prepared as a single stereoisomer such as levobupivacaine. Another familiar example of this is ketamine.
In contrast amethocaine (an ester) and lignocaine are achiral, ie they have no stereoisomers.
The mechanism of action of local anaesthetics
Local anaesthetics disrupt ion channel function within the neurone cell membrane preventing the transmission of the neuronal action potential. This is thought to occur via specific binding of the local anaesthetic molecules (in their ionised form) to sodium channels, holding them in an inactive state so that no further depolarisation can occur. This effect is mediated from within the cell; therefore the local anaesthetic must cross the cell membrane before it can exert its effect. A second mechanism is also thought to operate, involving the disruption of ion channel function by the incorporation of local anaesthetic molecules into the cell membrane (the membrane expansion theory). This is thought to be mediated mainly by the unionised form acting from outside the neuron. Nerve fibres differ in their sensitivity to local anaesthetics. Small nerve fibres are more sensitive than large nerve fibres while myelinated fibres are blocked before non-myelinated fibres of the same diameter. Thus the loss of nerve function proceeds as loss of pain, temperature, touch, proprioception, and then skeletal muscle tone. This is why people may still feel touch but not pain when using local anaesthesia.
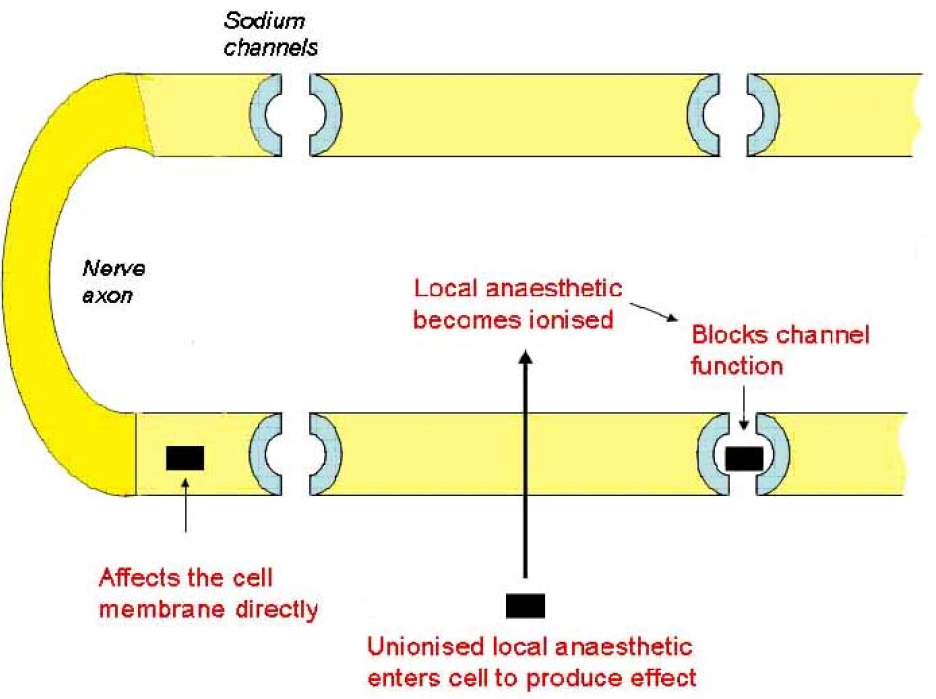
The importance of the pKa of a local anaesthetic drug.
All local anaesthetic agents are weak bases, meaning that they exist in two forms: unionised (B) and ionised (BH+). The pKa of a weak base defines the pH at which both forms exist in equal amounts. As the pH of the tissues differs from the pKa of the specific drug, more of the drug exists either in its charged or uncharged form. This is expressed in the Henderson-Hasselbalch equation:
- pKa – pH = log [BH+] / [B]
where [B] is the concentration of unionised and [BH+] the concentration of ionised drug.
How may the pKa of a local anaesthetic influence its speed of onset?
The pKa of a local anaesthetic determines the amount which exists in an ionised form at any given pH. At physiological pH (7.4) all local anaesthetics are more ionised than unionised (as all the pKa values are greater than 7.4). However the proportions vary between the drugs: lignocaine has a pKa of 7.9 and is approximately 25% unionised at pH 7.4 . Bupivacaine has a pKa of 8.1 and hence less of the drug is unionised at pH 7.4 (about 15%).
As the drug must enter the cell in order to have its effect it must pass through the lipid cell membrane. Unionised drug will do this more readily than ionised drug. Therefore the drug which is more unionised at physiological pH will reach its target site more quickly than the drug which is less so. This explains why lignocaine has a faster onset of action than bupivacaine.
Can this theory explain why local anaesthetics often don’t work in infected tissue?
The relevant feature of infected tissue is that it tends to be a more acidic environment than usual. As the pH is reduced the fraction of unionised local anaesthetic is reduced and consequently the effect is delayed and reduced. Infected tissue may also have an increased blood supply and hence more anaesthetic may be removed from the area before it can affect the neurone.
How else may the physicochemical characteristics of a local anaesthetic affect its function?
Physicochemical features such as the aromatic ring structure and hydrocarbon chain length of a particular local anaesthetic determine the lipid solubility of the drug and hence its potency. This makes sense since the more lipid soluble drug penetrates the cell membrane more easily to exert its effect. The more potent the drug, the smaller the amount required to produce a given effect. Thus bupivacaine – which is highly lipid soluble – is approximately four times more potent than lignocaine. This is reflected in the different preparations available of these two drugs; bupivacaine being more potent is prepared as a 0.1 – 0.5% solution. Lignocaine conversely is commonly presented as a 1% or 2% solution.
The duration of action of the drug is also related to its structure, primarily to the length of the intermediate chain joining the aromatic and amine groups. However it should be noted that protein binding is probably at least as important a determinant of duration of action. Clearly the molecular structure of the drug also affects protein binding ability and therefore all local anaesthetics differ in the extent to which they are protein-bound. So, for example, lignocaine is approximately 65% protein bound whereas bupivacaine is 95% protein bound. Therefore one can predict that bupivacaine will have a longer duration of action than lignocaine – which is in fact the case. Procaine (an ester), in contrast, is only 6% protein bound and has a very short duration of action. Differences in protein binding also result in differing duration of unwanted side effects and is one of the reasons that bupivacaine is considered more toxic than lignocaine.
Pharmacokinetics of local anaesthetics
Absorption and distribution
Local anaesthetic drugs are administered to the areas around the nerves to be blocked – which include skin, subcutaneous tissues, intrathecal and epidural spaces. Some of the drug will be absorbed into the systemic circulation: how much will depend on the vascularity of the area to which the drug has been applied and intrinsic effects of the drug or its additives on vessel diameter. Some local anaesthetics have vasodilatory effects at low concentrations, increasing their systemic absorption. This is countered in some preparations which include a vasoconstrictor such as adrenaline or felypressin. Cocaine, in contrast, has a vasoconstrictive effect.
The distribution of the drug is influenced by the degree of tissue and plasma protein binding of the drug. As discussed above, the more protein bound the agent, the longer the duration of action as free drug is more slowly made available for metabolism.
Metabolism and excretion
Ester and amide anaesthetics differ in their metabolism. Esters (except cocaine) are broken down rapidly by plasma esterases to inactive compounds and consequently have a short half life. Cocaine is hydrolysed in the liver. Ester metabolite excretion is renal.
Amides are metabolised hepatically by amidases. This is a slower process, hence their half-life is longer and they can accumulate if given in repeated doses or by infusion. Prilocaine is also metabolised extra-hepatically.
Which local anaesthetic drugs are more likely to affect the foetus when given in pregnancy and why? How does the situation change if the foetus is compromised?
The esters are metabolised sufficiently rapidly to have minimal effects on the foetus so little remains in the maternal circulation to cross the placenta. Amide local anaesthetics are more likely to cross the placenta. Of these, placental transfer is greater in those which are less protein-bound (such as lignocaine).
If the foetus is compromised it may become acidotic. In this situation more of the foetal local anaesthetic will be ionised and hence unable to return to the maternal circulation. This phenomenon is known as ion trapping and can result in foetal toxicity.
These effects are not likely to be important when small amounts of drug are used during spinal anaesthesia, but may become so when larger amount are used for epidural anaesthesia or other nerve blocks around the time of delivery.
Clinical uses of local anaesthetics
Preparations
Local anaesthetics are available as solutions for injection, sprays, creams and gels. They are prepared as the hydrochloride salt to enable them to be dissolved in water (resulting in an acidic solution). Of note, due to new legislation, some of the newer local anaesthetics are described in terms of the quantity of free base present alone, in contrast to the older drugs which are described in terms of the quantity of total hydrochloride salt present. This is why, for example, 10ml of 0.5% bupivacaine (a racemic mixture) contains fewer local anaesthetic molecules than 10ml of 0.5% levobupivacaine. Most local anaesthetic preparations contain a preservative agent such as 0.1% sodium metabisulphite, with or without a fungicide. Multidose vials contain 1mg/ml of the preservative methyl parahydroxybenzoate. The drug may also be combined (by the manufacturer or in some cases the clinician) with other local anaesthetics (e.g. EMLA cream – eutectic mixture of local anaesthetics) or additives designed to enhance their effects. These include adrenaline 1/200,000, bicarbonate (eg 0.15ml of 8.4% solution added to 10ml 0.5% bupivacaine) or glucose (usually 80mg/ml).
How might adrenaline, bicarbonate and glucose variously affect the action of local anaesthetics?
Adrenaline acts as a vasoconstrictor. The result is to minimise the vasodilator effect of (for example) lignocaine and decrease the rate at which drug is removed from the site of action by absorption into the systemic circulation. It also reduces traumatic (surgical) blood loss from the site by the same mechanism.
Bicarbonate added to a local anaesthetic increases the pH of the environment when administered. Consequently more drug is present in its unionised form and speed of onset of anaesthesia is increased. Too much bicarbonate however may result in precipitation of the local anaesthetic as the unionised form is much less soluble in water than the hydrochloride salt.
Glucose is added to bupivacaine in order to increase the baricity of the solution to greater than that of CSF. When administered as a spinal anaesthetic this results in more controlled spread of solution within the intrathecal space.
What harmful effects of local anaesthetics do you know?
Potential problems
Local anaesthetics may be toxic if sufficient amounts are absorbed into the systemic circulation. Of these bupivacaine appears to be the most dangerous although all can be harmful. Clinical toxicity appears to relate to the effects of the drug on other excitable membranes in the CNS and cardiovascular systems. CNS effects may include tingling of the lips, slurred speech, reduced level of consciousness and seizures. Cardiac effects on a variety of ion channels may cause arrhythmias and reduced myocardial contractility. In the case of bupivacaine the cardiac effects are particularly difficult to treat since its strong protein binding makes it difficult to displace from the myocardium. In contrast lignocaine may be used clinically for its cardiac effects as an antiarrhythmic.
Unexpected local anaesthetic toxicity can occur where the pharmacokinetics of the drug are altered by co-morbidity such as cardiac or hepatic failure (reducing metabolism of the drug), alterations in plasma protein binding, or interactions with other drugs.
Other clinical problems are more specific to particular drugs. The incidence of allergy to PABA, a metabolite of many esters has been mentioned. Prilocaine is metabolised to Otoluidine which can cause methaemoglobinaemia in susceptible individuals. Cocaine is a potent vasoconstrictor and may cause problems in patients already on vasoconstricting drugs such as monoamine oxidase inhibitors.
Summary
Understanding the pharmacology of local anaesthetics enables the anaesthetist to predict the potency, speed of onset, duration of action and safety of a specific drug in a given clinical situation. This maximises the opportunity for safe and effective use of local anaesthesia in a wide variety of contexts.
Further reading
- Tuckley JM. Pharmacology of local anaesthetic agents. Update in Anaesthesia 1994; 4: 19-24 Available at: www.world-anaesthesia.org
- Principles and Practice of Pharmacology for Anaesthetists: Calvey and Williams Pharmacology for Anaesthesia and Intensive Care: Peck, Hill and Williams
- Lagan G, McClure HA. Review of local anaesthetic agents. Current Anaesthesia & Critical Care 2004 15: 247-254



