Patient Safety
KEY POINTS
- Although a rare event in hospital practice, intra-arterial drug injection (IADI) may result in significant morbidity.
- Whilst the exact pathophysiology is unclear (and likely varies by drug) thrombosis is believed to represent a final common pathway. This may then lead to tissue ischaemia and necrosis.
- Particularly high-risk drugs when injected intra-arterially include benzodiazepines, penicillins, clindamycin, thiopental, phenytoin, and diclofenac.
- Management of IADI includes symptomatic treatments, assessing the risk of potential sequalae, and considering further interventions in high-risk cases (eg heparin and vascular/interventional radiology involvement).
- Prevention of IADI includes clear labelling of arterial lines, colour-coded tubing and attachments and the use of noninjectable ports on arterial lines.
INTRODUCTION
Accidental intra-arterial drug injection (IADI) represents a critical patient safety incident that may result in significant harm. Incidence of accidental IADI is often quoted as being between 1:3400 and 1:56 000; however this is based on studies from the 1940s and 1950s.1,2 A more recent UK study showed 28.5% of intensive care unit clinical directors surveyed were aware of an IADI occurring within their hospital during the preceding 5 years.3
Intra-arterial injection may occur via peripheral or central arteries and may occur iatrogenically or from illicit drug misuse. This tutorial will focus on peripheral iatrogenic IADIs, covering the aetiology, pathophysiology and management along with considering how such an event can be prevented.
TYPICAL SITES AND RISK FACTORS
IADIs may occur through accidental injection into an arterial line, or via a peripheral venous cannula (PVC) that has been mistakenly placed intra-arterially. Those at higher risk of accidental intra-arterial injection include the following4,5:
- Patients with arterial cannulae in situ
- Morbidly obese patients
- Patients with darkly pigmented skin
- Patients with PVCs in high-risk anatomical areas (ie, where arteries run near to veins, such as in the medial antecubital fossa)
- Patients with thoracic outlet syndrome (where radial artery pulsation may be lost with abduction and internal rotation of the arm
- Those with aberrant arterial anatomy (most commonly of the radial artery)
Regarding aberrant anatomy, normally the brachial artery divides into the radial and ulnar arteries within the cubital fossa. The radial artery then runs down the lateral aspect of the forearm, under the cover of brachioradialis, until the distal forearm, where it lies superficially over the ventral aspect of the wrist.6 Superficial radial arteries in the forearm are found in around 1% of all patients and may occur in association with a high-rising radial artery (ie, one that originates proximal to the cubital fossa), or with the antebrachialis superficialis dorsalis.4,5 Here, the radial artery divides in the forearm, producing a superficial branch that runs over the radial styloid process, close to the cephalic vein (a common site of venous cannulation near the lateral aspect of the wrist).5 Other abnormalities in the ulnar artery and palmar arches may also lead to superficial arteries, which may be inadvertently cannulated.4 Published case reports demonstrate the most common sites for accidental arterial cannulation within the arm are the lateral forearm, antecubital fossa, and dorsum of the hand.7
Features suggesting arterial rather than venous cannulation are shown below4,5:
- Marked pain upon cannulation
- Backflow of bright red blood into tubing
- Pulsatile blood within tubing
- Palpation of a pulse immediately proximal to cannula insertion point
- Arterial trace on pressure transduction of PVC
- Increase in ipsilaterally measured invasive blood pressure when flushing the PVC
- High PO2 of aspirated samples on blood gas analysis
Note, no single feature is completely reliable. Where there is doubt regarding whether the nature of the vessel is arterial or venous, the cannula should be removed and resited prior to use.8
PATHOPHYSIOLOGY OF INJURY FOLLOWING INJECTION
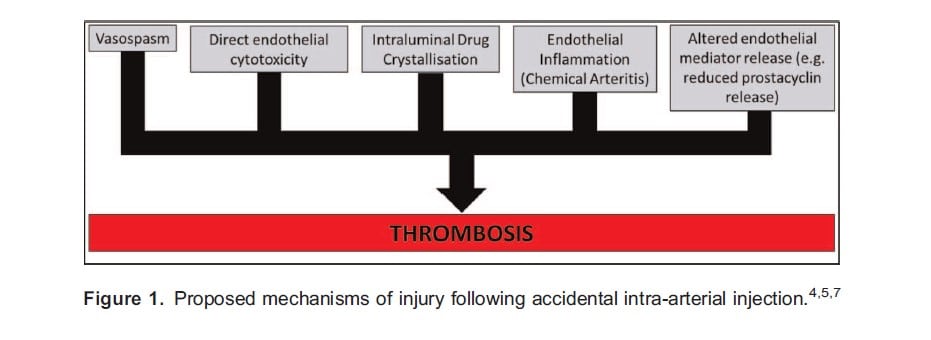
The mechanism of injury caused by inadvertent IADI has not been fully elucidated and it is likely that different drugs cause damage in different ways. Proposed mechanisms of injury are as shown in Figure 1, which also highlights the role of thrombosis as a potential final common pathway.
Perhaps the drug most well known to be damaging when injected intra-arterially is thiopental. Thiopental demonstrates tautomerism, being more water-soluble at increasingly alkaline pH and more lipid-soluble at increasingly acidic pH. Consequently, injection of a solution of thiopental (pH 10.5) into the bloodstream (pH 7.4) induces a shift to the less water soluble form.9 Within the venous system this is insignificant due to the continual dilution of thiopental as it ascends towards the heart; however, in the arterial circulation the continual narrowing of distal vessels presents an opportunity for crystals to form.7 Thiopental crystals may then obstruct the arterial lumen, be directly cytotoxic to the endothelium, and/or induce chemical arteritis.4,7 Vasodilatation may then be impaired through reduced release of vasodilatory (and antithrombotic) substances by the damaged endothelium.4 All these potential mechanisms will promote thrombus formation, leading to ischaemia, inflammation, oedema, and tissue necrosis.4
CLINICAL COURSE
Conscious patients commonly complain of pain or discomfort upon injection. This occurs distal to the site of injection and varies in intensity from mild to severe.5 Although often settling initially, pain can recur in subsequent hours and this may indicate that a significant injury is developing.7 Paraesthesia may also develop over the first 24 hours, with motor deficits described by some.5
Clearly, those with a decreased level of consciousness (eg due to general anaesthesia) at the time of injection will not describe any symptoms and are, therefore, at greater risk of receiving high-volume intra-arterial injections.
Signs of distal ischaemia (eg mottling) may take up to 72 hours to develop and their initial absence does not predict outcome. Similarly, peripheral pulses may remain palpable, since vascular occlusion often occurs distal to sites of palpation.7 Increasing pain, pallor, pulselessness, paraesthesia, and paralysis may indicate the development of compartment syndrome and requires urgent surgical review.5 Tissue necrosis tends to develop over up to 2 weeks and may require amputation. In the long term, patients may go on to develop chronic pain syndromes in the affected limb.5
Importantly, not all patients will develop sequalae and the time frame to development of signs and symptoms is highly variable. Published evidence suggests that certain drugs commonly used in anaesthetic practice are higher risk for the development of complications, these include the following7,10:
- Benzodiazepines
- Penicillins
- Clindamycin
- Thiopental
- Phenytoin
- Diclofenac
MANAGEMENT
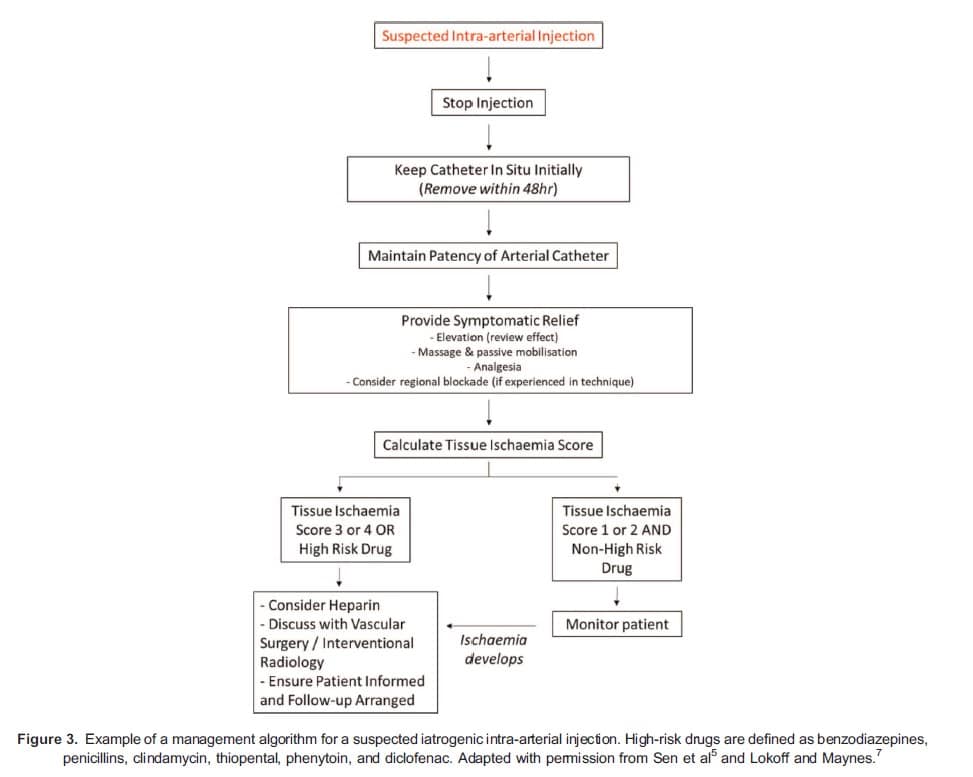
Published literature on IADI is limited to heterogeneous case reports and case series, often in patients who have self-injected drugs of abuse in the community. Optimal management of iatrogenic events is, therefore, uncertain and subject to ongoing debate. Common management approaches can be subdivided into initial actions, symptomatic treatments, assessment of potential severity, and further interventions.
Initial Actions
Should accidental IADI be recognised whilst drugs are still being injected this should cease immediately. Cannulae should initially be left in situ as they may be useful for angiography and for delivering intra-arterial therapy. Cannulae should, however, be removed within the first 48 hours so as to reduce the risk of clot formation upon the PVC.11 In order to maintain patency, an isotonic infusion may be commenced (eg 0.9% sodium chloride running at a rate of 1 mg/kg/hr12). Whilst seeming logical, there are no outcome data on the value of this intervention.5
Symptomatic Treatments
Secure intravenous access should be obtained as a priority and analgesia (eg opiates 6nonsteroidal anti-inflammatory drugs) administered.5 Oedema is common following IADI and one simple intervention may be to elevate the affected limb (for up to 72 hours) in order to improve venous and lymphatic drainage and reduce compartment pressures.5 Concern exists, however, about impairing arterial inflow in those with near-complete arterial occlusion and the effects of elevation should be continually reviewed and removed if circulatory flow is worsened.4 Regular massage of the affected limb and passive mobilisation of the joints (for 20-30 minutes 3 times a day) has also been used as a component of care in some cases where positive outcomes have been observed.13
Local anaesthetic blockade has been described, both for analgesia and to achieve vaso- or veno-dilatation. For IADIs, the classically described upper arm block is the stellate ganglion block. This, however, is likely an unfamiliar technique to many anaesthetists and one that carries significant risks (including pneumothorax, carotid artery or internal jugular vein puncture, and vagus nerve or brachial plexus injury14). Axillary brachial plexus blockade (without or without catheter insertion) has been used by some authors and has the advantage of being a more practised technique for many.5 Regardless, the risk of regional anaesthesia in patients who may also be anticoagulated and/or thrombolysed should be carefully considered, especially given that patient outcomes following regional blockade have not been found to be significantly improved.7,15
Of note, intra-arterial lidocaine (up to 2 mg/kg, without adrenaline) has been used by some to prevent arterial vasospasm. In one case report, its use 12 hours after intra-arterial injection of thiopental resulted in rapid clinical improvement; however, concern exists that a strategy that involves further IADI may increase the risk arterial damage.4,5 Certainly, its use should be avoided if other regional anaesthetic techniques are utilised.
Assessment of Potential Severity
Not all IADIs will result in damage and treatments are not without risk. One scoring system that may allow for better targeting of treatment is the Tissue Ischaemia Score, which was developed in 1990 following experience with 48 patients who had self-injected illicit drugs intra-arterially.16 This simple score is calculated from the presence/absence of 4 clinical features, with a score of 3 to 4 being associated with a worse clinical outcome. The components of the score are shown in Figure 2.
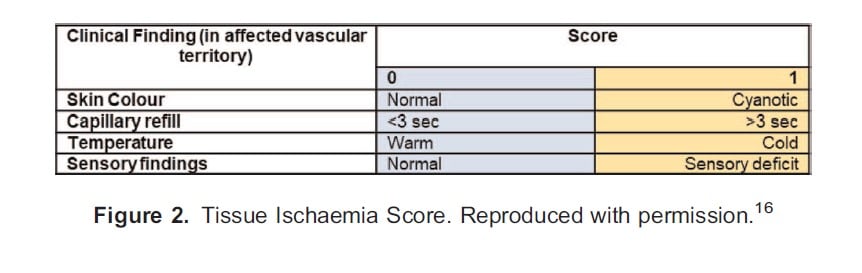
It is important to recognise this score was developed from experience with patients whose IADI occurred in the community, with the time to presentation variable (up to 99 hours) and the commonly injected drugs different from in-hospital practice (the most common drug injected in this study was methylphenidate).16 Consequently, for iatrogenic IADIs it has been suggested that either those exposed to high-risk drugs or those with a tissue ischaemia score of 3 to 4 should be commenced on further treatment. Patients injected with lower-risk drugs and with a low tissue ischaemia score (1-2) can be monitored, with further treatment instigated only if signs of ischaemia develop.7
Further Interventions
Given the proposed pathophysiology, anticoagulant therapy appears logical and has been widely used.5,7,8,10,12,16 A recent meta-analysis of 209 patients from 25 studies did not demonstrate a significant reduction in amputation rates with anticoagulation; however, in all studies the patients had self-injected drugs (often crushed oral preparations), there was variation in the time to hospital presentation, and varying regimes for anticoagulation were used.15 The generalisability to iatrogenic IADIs is, therefore, unclear. If anticoagulation is being considered one suggested protocol would be an intravenous bolus of 5000 units of unfractionated heparin, followed by an intravenous infusion (for at least 72 hours) to maintain an activated partial thromboplastin time ratio between 2.0 and 2.5. If signs of distal ischaemia do not develop within the initial 72-hour period then heparin may be discontinued.7
Corticosteroids have been used to reduce inflammatory damage in some case reports as part of a number of treatment interventions. Meta-analysis of IADIs amongst drug abusers did show statistically significant reductions in amputation rates on initial analysis; however, significance was lost on adjustment for potential confounders.15 In some studies, antibiotics have also been routinely administered; however, current recommendations are that these only be used in patients with clinical evidence of infection.5 In the presence of compartment syndrome, urgent surgical review should be sought and fasciotomies may be indicated.
Early referral to vascular surgery and/or interventional radiology should also be undertaken. Angiography may be considered, especially if ischaemia develops despite initial treatment.7,15 In the presence of positive angiography, and in the absence of contraindications, intra-arterial thrombolysis has been used by some authors and may offer benefit.7,12 Heparin may be continued intravenously following thrombolysis or may be replaced by an intra-arterial infusion.17 Should major bleeding or compartment syndrome develop, both the thrombolytic and heparin should be immediately stopped.12 In some cases where crushed tablets have been self-injected intra-arterially, a 4-hourly alternating intra-arterial infusion of thrombolysis and prostaglandin E1 has been used, with generally positive outcomes.13
Intra-arterial papaverine has also been used in the treatment of IADIs, where it induces vasodilatation by directly relaxing vascular smooth muscle.11 Intravenous iloprost (a prostaglandin I2 analogue) induces vasodilatation as well as having antiplatelet properties, and it may also have a role in treatment.5,8
Follow-Up
Patients should, at the earliest opportunity, be informed of the event in an open and honest manner and, in the UK, professional guidance exists regarding this duty of candour.18 Local critical incident paperwork should be completed and events discussed at local morbidity and mortality meetings. This allows for incidents to be reflected upon, root cause analysis undertaken, and local policies reviewed to reduce the risk of similar events occurring in the future. Patients should be referred for regular physiotherapy and should be actively followed up to ensure complete symptom resolution. Chronic pain will require referral to specialist colleagues (Figure 3).5
PREVENTION
Given the potentially serious morbidity associated with IADI, the aim should be to prevent these events before they happen. Within anaesthetic and intensive care unit practice, the most likely source of IADI is through the administration of drugs through an indwelling arterial catheter. The following steps can reduce the risk these events occurring19:
- Labelling lines (eg with red “ARTERIAL” stickers), especially around injection ports
- Using colour-coded tubing and injection ports (red for arterial, blue for venous)
- Minimising injection ports within lines (where used, keep these close to the cannula site)
- Considering the use of noninjectable connectors on arterial lines
- Tracing tubing to the cannula prior to injection
- Providing regular education for all those involved in setting up and using arterial lines
SUMMARY
Accidental intra-arterial drug injection is a rare but potentially serious adverse event. It may occur either within hospital as an iatrogenic incident, or within the community as a complication of intravenous drug abuse. The exact mechanism of injury likely varies according to the exact circumstances and drugs involved; however, thrombosis is believed to represent a final common pathway through which tissue ischaemia and necrosis develops. No randomised control trials exist to guide patient management and many interventions have been utilised in case studies (often simultaneously). In general, cannulas should be kept in situ initially, symptomatic relief provided, and an assessment of the likelihood of serious injury undertaken. Expert advice should be sought at an early stage, patients should be informed of the event as soon as practicable, and close follow-up undertaken.
REFERENCES
1. Cohen SM. Accidental intra-arterial injection of drugs. Lancet. 1948;255:409-417.
2. Dundee JW. Thiopental and other barbiturates. Int Anesthesiol Clin. 1956;197-243.
3. Mariyaselvam M, Hutton A, Young P. Accidental intra-arterial injection: an under-reported preventable never event. Crit Care. 2015;19 (S1):P166.
4. Ghouri AF, Mading W, Prabaker K. Accidental intraarterial drug injections via intravascular catheters placed on the dorsum of the hand. Anesth Analg. 2002;95:487-491.
5. Sen S, Chini EN, Brown MJ. Complications after unintentional intra-arterial injection of drugs: risks, outcomes & management strategies. Mayo Clin Proc. 2005;80(6):783-795.
6. McMinn RMH, Gaddum-Rosse P, Hutchings RT, Logan BM. McMinn’s Functional and Clinical Anatomy. 1st ed. London, UK: Mosby; 1995.
7. Lokoff A, Maynes JT. The incidence, significance, and management of accidental intra-arterial injection—a narrative review. Can J Anaesth. 2019;66:576-592.
8. Lake C, Beecroft CL. Extravasation injuries and accidental intra-arterial Injection. Contin Educ Anaesth Crit Care Pain. 2010;10:109-113.
9. Peck TE, Hill SA, Williams M. Pharmacology for Anaesthesia and Intensive Care. 3rd ed. Cambridge, UK: Cambridge University Press; 2008.
10. Patel J, Shah P, Gandhi F. Evaluation and management of accidental intra-arterial injection in the ante-cubital fossa. J Vasc Med Surg. 2020;8(2):387.
11. Arquilla B, Gupta R, Gernshiemer J, Fischer M. Acute arterial spasm in an extremity caused by inadvertent intra-arterial injection successfully treated in the emergency department. J Emerg Med. 2000;19(2):139-143.
12. Breguet R, Terraz S, Righini M, Didier D. Acute hand ischemia after unintentional intraarterial injection of drugs: is catheterdirected thrombolysis useful? J Vasc Interv Radiol. 2014;25:963-968.
13. Rohm S, Staab H, Schulz H, Richtr O, Aust G. Good clinical outcome after accidental intra-arterial injection of flunitrazepam tablets in 16 drug abusers with critical limb ischemia. Eur J Vasc Endovasc Surg. 2014;47:61-67.
14. Thanawala V, Dedhia J. Stellate ganglion block. Anaesth Tutorial Week 2012;256. https://resources.wfsahq.org/atotw/stellate-ganglion-block/
15. Devulapalli C, Han KD, Bello RJ, et al. Inadvertent intra-arterial drug injections in the upper extremity: systematic review. J Hand Surg Am. 2015;40(11):2262-2268.
16. Treiman GS, Yellin AE, Weaver FA, et al. An effective treatment protocol for intraarterial drug injection. J Vasc Surg. 1990;12(4):456-466.
17. Morrison HL. Catheter-directed thrombolysis for acute limb ischemia. Semin Intervent Radiol. 2006;23(3):258-269.
18. General Medical Council. Openness and honesty when things go wrong: The professional duty of candour. Published 29th June 2015 (Updated 15th March 2022). https://www.gmc-uk.org/-/media/documents/openness-and-honesty-when-thingsgo-
wrong–the-professional-duty-of-cand____pdf-61540594.pdf
19. Mariyaselvam MZ, Heij RE, Laba D, et al. Description of a new non-injectable connector to reduce the complications of arterial blood sampling. Anaesthesia. 2015;70:51-55.



