Intensive Care Medicine
Self assessment questions
Scenario: A twenty-year-old female is brought into the Emergency Department having been found unconscious in her bedsit. There is no other recent history. She did not respond to a bolus of 50% dextrose in the ambulance, despite having an unrecordable blood glucose when tested by the paramedics. While she is being intubated on account of reduced level of consciousness, an arterial blood gas sample reveals profound lactic acidosis (pH 7.05, pCO2 2.5 kPa, base deficit – 10, lactate 13 mg/L). Blood pressure is 95/50 mmHg.
- What are the possible explanations for her presentation?
- Laboratory tests demonstrate hepatocellular necrosis (AST 21,000 U/L) and coagulopathy (INR 9.1) with thrombocytopenia (platelet count 26 x 109 /L). Acute liver failure appears the most likely diagnosis.
- What are the most likely causes of acute liver failure (ALF) in this previously well patient?
- Her mean arterial blood pressure remains low (50mmHg) after 3 litres of colloid and crystalloid. The casualty nurse, who is doing half-hourly neurological observations, reports reduced pupillary response to light.
- What severe complications of ALF may result in death within hours, and what are the immediate management priorities for this patient?
Introduction
Successful management of this rare but potentially devastating disorder relies on early recognition. The hallmark of acute liver failure (ALF) is encephalopathy (ranging from a subtle alterations in consciousness level to coma) in the context of an acute, severe liver injury. The presence of a liver injury is suggested by raised transaminase levels (in the thousands indicating hepatocellular necrosis), impairment of synthetic function manifested by coagulopathy (INR>1.5), and metabolic derangements such as hypoglycaemia and lactic acidosis. Sometimes the history contains clues to the cause of the liver injury (eg. paracetamol ingestion), but frequently the cause is not apparent.
Types of liver failure
The speed of onset of encephalopathy after the onset of symptoms (usually jaundice) is important in both categorisation and prognosis. If this is less than seven days the term ‘hyperacute’ liver failure is used; 8-28 days indicates ‘acute’ liver failure and 5 to 26 weeks, is termed ‘subacute’ liver failure. However, ALF can be used to describe all these categories. The more slowly progressive types of ALF can result in an equally severe illness, but tend to be caused by idiosyncratic drug reactions, autoimmune hepatitis and ‘seronegative’ hepatitis (in which no specific cause is determined). Hyperacute types are caused by paracetamol and the viral hepatitides, and, because of the speed of onset, there is often not enough time for frank jaundice to develop before encephalopathy occurs. Table 1 summarizes the main causes of ALF in the United Kingdom. There is significant geographical variation; for example paracetamol accounts for only 2% in France, and viral hepatitis causes 55% of cases in Japan. In India Hepatitis E is responsible for 33% of cases.
This article will concentrate on the management of ‘hyperacute’ ALF, which is associated with the most dramatic presentations.

Table 1.
ALF causes multi-system failure. The most dangerous consequences of ALF are cerebral oedema and the risk of tentorial herniation, severe vasodilatory shock, lactic acidosis and hypoglycaemia. Acute renal failure will almost always follow. Coagulopathy, although often profound, rarely results in catastrophic bleeding.
Assessment of encephalopathy
Grading of encephalopathy in ALF borrows from features seen in cirrhosis. However, encephalopathy in ALF differs greatly from that seen in cirrhosis because cerebral oedema is a frequent complication in grade 3 and 4 (see section 2, Cerebral Protection).

Table 2: Grading of encephalopathy
Investigations to determine the cause of ALF, although of paramount importance, are rarely informative in the early stages of care. For example, viral and autoimmune serology (where available) will not generally yield results within 12-24 hours. An ideal investigative screen is summarised in Table 2, but delay in receiving results should not hold up discussion with a liver unit. An ultrasound, if available, is always useful to exclude underlying cirrhosis (which will lead to different management), massive malignant infiltration (which may save the patient a disruptive transfer out of area) and an acute vascular pathology (eg. portal or hepatic vein thrombosis).
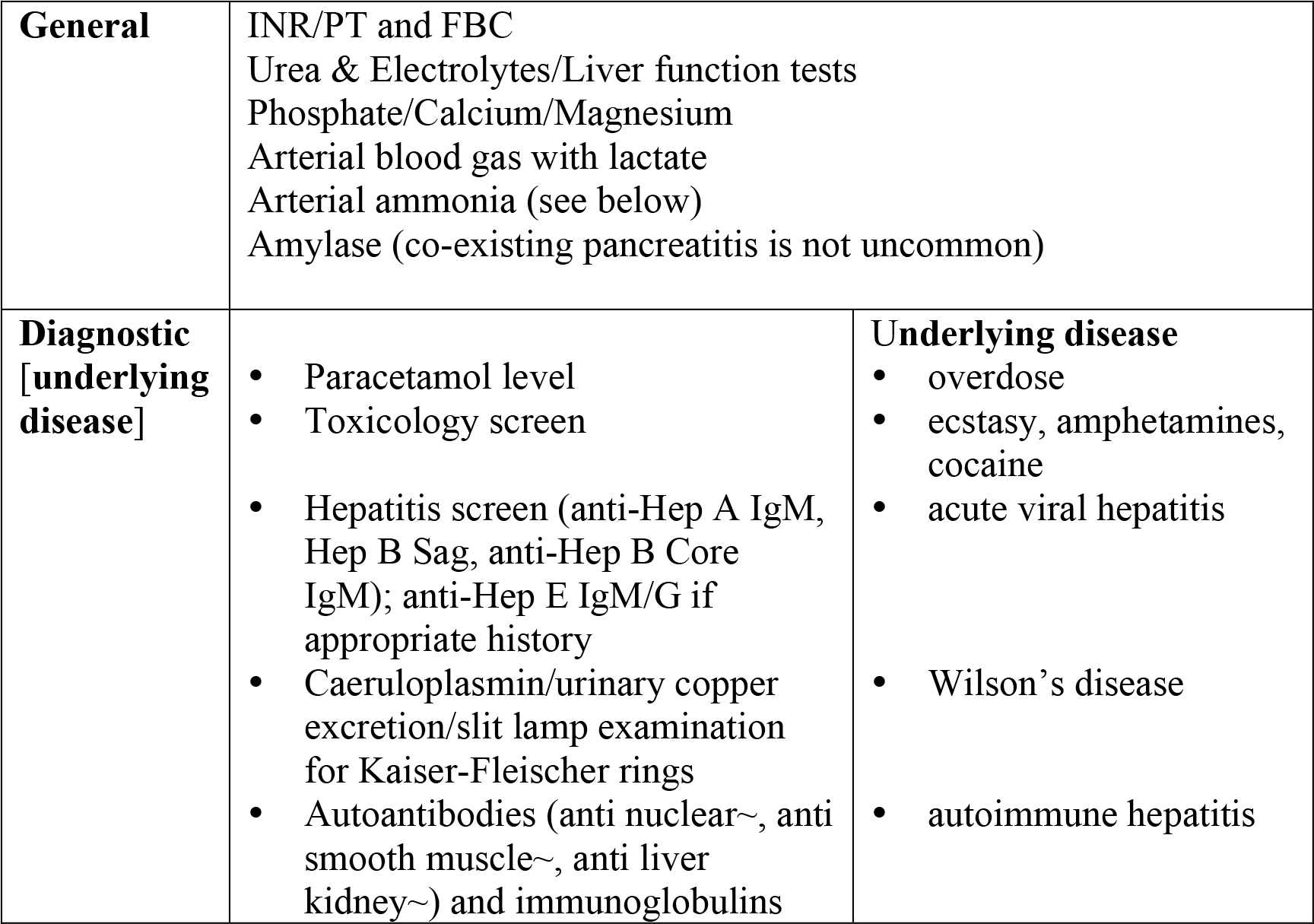
Table 3. Investigations in ALF.
Management
Acute management is focussed on –
- Global organ support
- Cerebral protection
- Identification of patients likely to benefit from liver transplantation (LT), and liason with a specialist liver unit
- Ensuring safe inter-hospital transfer
The role of specific antidotes to reverse the effect of the aetiological agent or pathogen (eg. antiviral therapy, copper chelation in Wilson’s disease, steroids in autoimmune disease) are very limited, however N-acetylcysteine should be administered if paracetamol toxicity is suspected, even if the conventional therapeutic window has passed. It may have a beneficial role in other types of ALF, but this remains unproven.
1. Global organ support
Circulation
Vasodilatory shock is common. Invasive monitoring (eg. with ‘pulse-induced contour cardiac output’ – PiCCO) will aid assessment of volume and identify co-existing cardiac dysfunction. Aggressive fluid resuscitation is almost always required, with the fluid volume required often exceeding 3 litres. As in head injury, salt-containing fluids are appropriate, since hyponatraemia may exacerbate cerebral oedema. Vasopressor therapy should be instituted if mean arterial blood pressure does not respond to filling. Patients requiring inotropes may have relative adrenal deficiency and should have a short synacthen (ACTH stimulation) test and hydrocortisone 50mg 6 hourly commenced.
Renal/Acid-Base balance
Continous veno-venous haemofiltration (CVVHF) will be required if the patient is anuric or acidotic. CVVHF is favoured since it is better tolerated by patients with haemodynamic compromise. A bicarbonate buffered replacement solution should be used (lactate will not be handled well by the liver), and epoprostanol (Flolan!) used to anticoagulate the circuit. High volume (90mls/kg/hr) can be used if the acid-base status is extremely deranged.
Respiratory function
Although ARDS can develop in ALF, specific ventilatory strategies are not usually recommended in the early phase. CO2 targets are covered below.
Coagulation
The extent of coagulopathy is critical in deciding if the patient will benefit from LT, and administration of FFP is not recommended unless there is clinically significant bleeding. Platelets can be supported freely. Coagulopathy should not necessarily preclude central line insertion (especially femoral) by an experienced operator. Dissemninated intravascular coagulation and fibrinogen defects can further complicate the coagulopathy. Gastric ulcer protection with iv or enteral proton pump inhibitors reduces the risk of gastrointestinal bleeding.
Protection against sepsis
Bacterial and fungal sepsis complicated a high proportion of ALF patients before antibacterial prophylaxis became standard. Recommended agents are TazobactamPiperacillin (Tazocin®) 4.5g/iv/tds and Fluconazole 200mg/iv daily.
Gastrointestinal tract and nutrition
Gastric ulcer protection with IV or enteral proton pump inhibitors reduces the risk of gastrointestinal bleeding. There are no contraindications to early enteral feeding, and standard amounts of nitrogen and carbohydrate can be administered.
2. Cerebral protection
Cerebral oedema complicates severe encephalopathy, and may occur in 80% of patients in grade IV. Those most at risk are the young, the septic, the hyponatraemic and patients with significant elevations in blood ammonia (which appears to have an important role in both encephalopathy and oedema). Arterial ammonia concentration correlates with the degree of encephalopathy, and is useful in assessing the risk of developing intracranial hypertension. Experience at Kings College Hospital has shown that over 30% of patients with a level 100-200µmol/L developintracranial hypertension, and over 50% with levels exceeding 200µmol/L. Venous ammonia levels are significantly lower than arterial levels due to the ability of skeletal muscle to metabolise ammonia.
Clinical signs of rising intracranial pressure (ICP) are hypertonia, clonus, pupillary abnormalities (dilatation, reduced responsiveness to light) and in the latter stages hypertension (bursts to > 200 mmHg, or sustained above 150 mmHg), bradycardia and cerebral posturing.
Such patients will have been intubated for airway protection. Recommended sedation is propofol (possible beneficial cerebral metabolic effects) and fentanyl. Additionally the following prophylactic measures are advised.
Posture
Positioning the head in the midline, with the angle of the body 20° from horizontal will aid cerebral venous outflow.
Normocapnia (PaCO2 4.5-5 kPa)
Hypercapnia should be avoided, however the ‘traditional’ approach of hyperventilation and low PaCO2, although effective in short term reduction of cerebral blood flow and ICP, can lead to cerebral vasospasm and increased risk of brain injury.
Body temperature
Fever should be managed with cooling blankets, or if on CVVHF reduced thermal compensation.
Avoidance of stimulation
Surges in ICP are seen in monitored patients who are exposed to stimuli such as loud noise, suctioning and excessive movement.
Specific treatments if raised ICP is suspected include the following. These would normally be undertaken only after discussion with a specialist centre.
Mannitol
0.5g/kg boluses over 10 minutes may be administered and repeated, but serum osmolarity should be monitored so that 320 mosm/L is not exceeded. To prevent fluid overload a 500ml diuresis (or negative balance if being haemofilitered) should be obtained after each bolus.
Hypothermia
Small case series have demonstrated that reducing the core temperature to 32°-34°C (‘moderate hypothermia’) can safely bring about significant and prolonged reductions in ICP in patients with raised ICP that has not responded to medical therapy. In Jalan’s uncontrolled study (2004), 13 of 14 patients were successfully bridged to transplant (for between 10 and 118 hours) with this approach. ICP fell from a median of 36.5 mmHg to 16.3 mmHg (p<0.001) over 4 hours. However, there are concerns about increased susceptibility to infection, cardiovascular instability and bleeding with hypothermia of this degree, and multi-centre randomised controlled trials are under way in the hope that a mortality benefit will be detected. The pathophysiological rationale behind this treatment appears strong, and in practical terms reducing core temperature to around 35°C is a reasonable target.
Hypernatraemia
Serum sodium in the range 145-155 mmol/L has been shown to have a beneficial effect on ICP. This target can be achieved with continuous central infusion of 30% NaCl, with regular monitoring. A slow bolus of 20ml 30% sodium chloride may also be administered for possible surges in ICP.
Indomethacin and thiopentone are also used in cases resistant to these measures, but will usually only be administered after insertion of an intra-cranial pressure monitor.
3. Identification of patients likely to benefit from liver transplantation (LT), and liason with a specialist liver unit.
Studies have shown that patients meeting certain criteria (based on aetiology of liver failure, degree of encephalopathy, severity of coagulopathy and presence of extrahepatic organ dysfunction) who at a higher risk of death, obtain a survival advantage from LT. The widely quoted King’s College Hospital criteria are now integrated into a 10 category scheme used by the United Kingdom Transplant (UKT) service. The categories covering the scenarios most likely to be seen (paracetamol toxicity, viral hepatitis) are listed in BOX 3. It should be noted that patients do not need to meet these criteria to benefit from specialist care; it is preferable for the patient to be in a liver unit if criteria are being approached.
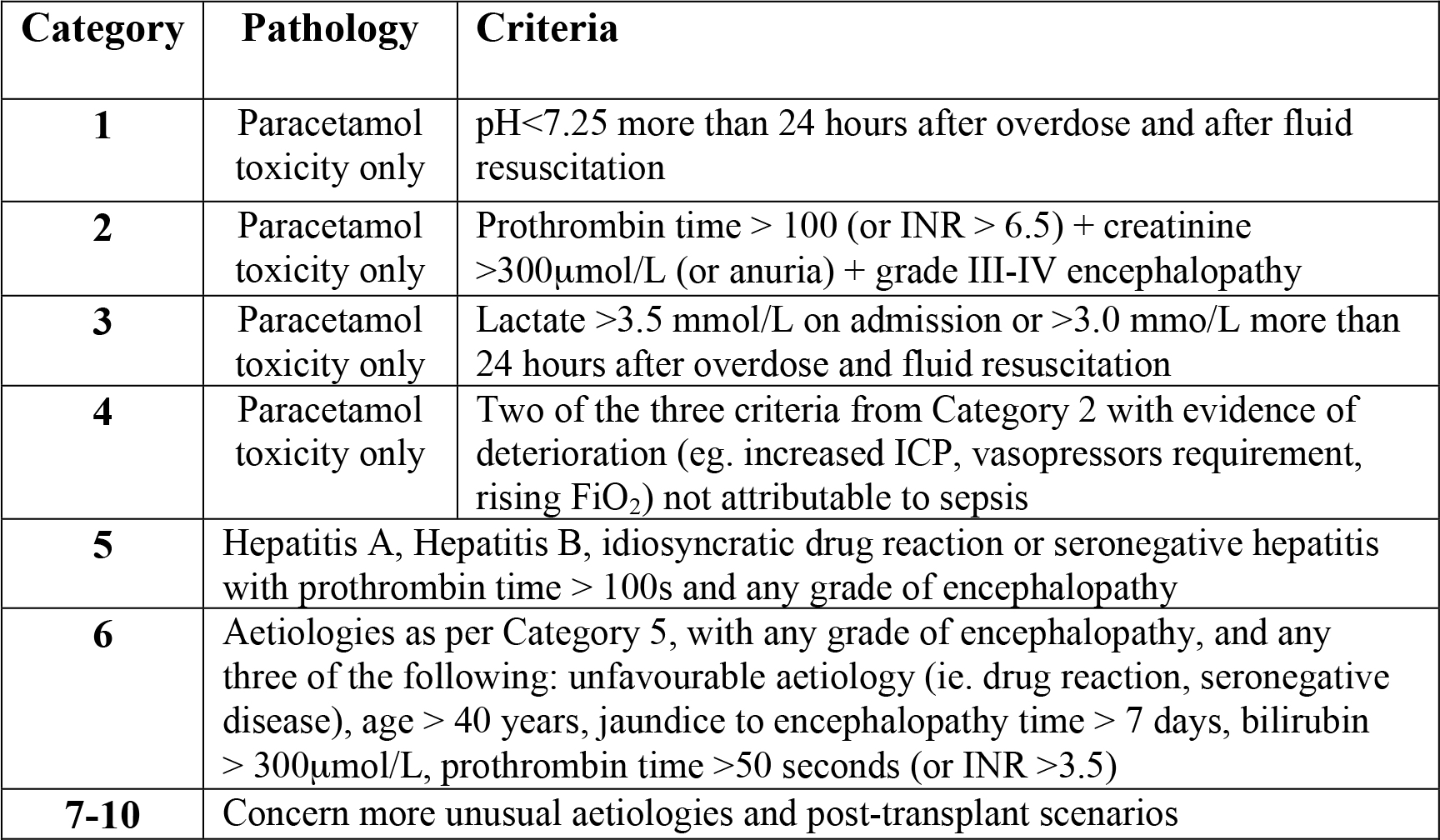
Table 4: UKT acute liver failure (super urgent) transplant criteria
Specific information that will be requested by the liver unit will include:
- Known historical factors – timing of hepatoxic drug ingestion (eg. paracetamol), psychiatric history, major comordities,
- Current status – level of consciousness, cardiovascular status and vasopressor requirement, FiO2/PaO2 ratio, urine output, U&E’s/creatinine, INR/PT (and blood products given), pH/blood lactate, monitoring and vascular access in place,
- Geography – distance from tertiary centre, estimated duration of transfer.
4. Ensuring safe inter-hospital transfer
If the patient requires transfer to a liver unit, remember that deterioration can occur swiftly, especially in terms of conscious level, cerebral perfusion and cardiovascular stability. Where available, air transfer is often advised if the disease process appears to be advanced. The following summary covers recommendations for inter-hospital transfer.
Ventilation
Elective intubation is almost always required, even if a good conscious level has been maintained in the referring hospital, to avoid the risks of emergency intubation during transfer. Aim to achieve normocapnia.
Cardiovascular
An arterial catheter with portable blood pressure monitoring is preferred. Wide bore peripheral venous access and central access, through the femoral vein if necessary, are usually advised in case of the need for large volume infusions and vasopressors.
Ample reserves of colloid/crystalloid should be taken, as should norepinephrine (made up prior to departure, eg. 5mg/50mls). Adequate cerebral perfusion will depend on maintaining a mean arterial pressure of over 70mmHg.
Neurological
Monitor pupil size and response. If there is a suspicion of rising ICP mannitol 0.5g/kg should be given. For a 70kg man give 175ml of 20% mannitol or 350ml of 10% mannitol over 10 minutes.
Metabolism
Frequent blood sugars, with hypoglycaemia treated with 50% glucose. Sodium bicarbonate (50-100mls of 8.4%) can be given prior to departure if acidosis is profound.
Key Points
- ALF should be considered in the context of unexplained loss of consciousness, hypoglyaemia or coagulopathy.
- ALF causes death by cerebral oedema, vasodilatory shock and hypoglycaemia.
- Aggressive volume resuscitation is required. Saline and artificial colloids are safe, but 5% glucose may exacerbate cerebral oedema and should not be used.
- Vasopressors should be used if blood pressure remains low after fluid resuscitation – adequate cerebral perfusion pressure must be maintained.
- Encephalopathy in ALF is progressive – early elective intubation is preferable to emergent intubation
Answers to self assessment questions
- ALF mimics other conditions that can present with obtundation or coma, shock, hypotension and acidosis, eg. septicaemia, hypoperfusion secondary to cardiac failure or volume depletion. Derangements in haematological and coagulation indices typical of ALF can also be seen in acute presentations of haematological malignancy or disseminated intravascular coagulation. Severe hyperlactataemia without liver failure or hypoperfusion may be induced by toxic drug effects (metformin, theophylline, ethylene glycol, methanol, cocaine, cyanide). A marked transaminitis (>40x upper limit of normal) will usually lead the clinician to suspect hepatocellular necrosis, although in non-hyperacute types AST/ALT may be ‘burning out’ at presentation due to the progressive loss of hepatocytes. Bilirubin may not be particularly high in rapid onset disease.
- The most likely causes of ALF in a patient of this age are: paracetamol poisoning, acute hepatitis A or B infection, idiosyncratic drug reaction, seronegative hepatitis, Wilson’s disease and autoimmune hepatitis. Hepatitis E should be suspected if there is a history of recent travel to an area where the virus is endemic. Hepatitis E infection in pregnancy is associated with a 20% risk of death in some reports.
- This patient may die due to raised intracranial pressure and tentorial herniation, progressive vasodilatory shock, unrecognised hypoglycaemia or severe acidosis and cardiac arrest. If ALF is strongly suspected the priorities are: airway protection, avoidance of hypoglycaemia, maintenance of cerebral perfusion pressure (fluid resuscitation +/- vasopressors), avoidance of hyponatraemia and, where available, liason with a liver transplant unit. If the patient is too unstable to be transferred (hypotension, profound acidosis resulting in suppression of cardiac function) further optimisation with guided volume resuscitation and continuous veno-venous haemofiltration may be required. See text for strategies to minimise the risk of raised intracranial hypertension.
Recommended reading and important papers in anaesthetic/intensive care management
- O’Grady JG, Alexander GJM, Hayllar KM, Williams R. Early indicators of prognosis in fulminant hepatic failure. Gastroenterology 189;97:439-45
- Bernal W, Donaldson N, Wyncoll D, Wendon J. Blood lactate as an early predictor of outcome in paracetamol-induced acute liver failure: a cohort study. Lancet. 2002; 359: 558-63.
- Wijkicks EFM, Nyberg SL. Propofol to control intracranial pressure in fulminant hepatic failure. Transplant Proc 2002; 34: 1220-1222
- Jalan R, Olde Daminsk SWM, Deutz NEP, et al. Moderate hypothermia in patients with acute liver failure and uncontrolled intracranial hypertension. Gastroenterology 2004; 127: 1338-1346
- Murphy N, Auzinger G, Bernal W, Wendon J. The effect of hypertonic sodium chloride on intracranial in patients with acute liver failure. Hepatology 2002; 39: 464-470
- Davenport A, Will EJ, Davidson AM. Improved cardiovascular stability during continuous modes of renal replacement therapy in crticially ill patients with acute hepatic and renal failure. Crit Care Med 1993; 21: 328-338
- Harrison PM, Keays R, Bray GP, et al. Improved outcome of paracetamol-induced fulminant hepatic failure by late administration of acetylcysteine. Lancet. 1990; 30: 1572–1573



