Intensive Care Medicine
CLINICAL CASE
A 26 year old female presents to the emergency department (ED) with sudden onset severe abdominal pain.
She is also feeling generally unwell and dizzy.
The patient states she is 8 weeks pregnant.
On arrival in ED her blood pressure (BP) is 80/60 mmHg and heart rate (HR) is 85 bpm. Fluid resuscitation with 4 litres of crystalloid solution begins immediately.
‘FAST’ ultrasound scan (focused abdominal sonography in trauma) shows a large amount of free fluid in the peritoneal cavity. Bedside pelvic ultrasound performed by the obstetrician is suggestive of ectopic pregnancy although an intrauterine pregnancy is also seen (Heterotropic ectopic pregnancy).
The patient is booked for an urgent laparotomy, and arrives in theatre 15 minutes later.
On arrival in the OT her BP is 65/45 mmHg and her HR is 125 bpm. SaO2 99% 2L O2. The patient is pale, drowsy and difficult to rouse.
- Anaesthetic given: RSI Induction (40mg propofol, 80mg suxamethonium), 3mg midazolam, metaraminol infusion to maintain mean arterial pressure.
- Routine monitoring (pulse oximetry, non-invasive blood pressure, capnography, ECG and gas analysis).
- Forced air warmer, blood warmer and constant temperature monitoring are used.
- An arterial line is placed for continuous invasive BP monitoring.
- Massive transfusion: 8 units (cross matched) RBC, 4 FFP, 8 cryoprecipitate. Transfusion begun within 5 minutes of anaesthesia induction.
Surgical findings are: a gestational sac in right fallopian tube (eroded through a blood vessel) with 3 litres of blood in the pelvis. The ectopic pregnancy is removed and surgery is complete within 40 minutes.
The patient is transferred to Intensive Care and is ventilated for 12 hours. She is extubated, then transferred to the ward the next day and discharged home two weeks later with a viable intrauterine pregnancy.
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article.
- What is hypovolaemic shock?
- What is the normal blood volume for an 80kg man? What about for a 60kg woman?
- What clinical signs are used to assess the degree of blood loss?
- What are the main goals of initial management of massive haemorrhage?
- What blood products can be used in severe haemorrhage?
- What are the main complications from massive haemorrhage?
ASSESSING DEGREE OF BLOOD LOSS: PHYSIOLOGICAL RESPONSE
- Shock: inadequate tissue perfusion for metabolic requirements and the associated physiological response.
- Hypovolaemic shock: inadequate tissue perfusion due to reduced circulating blood volume.
- Normal blood volume is 70mls/kg. That is 4-5L total blood volume (TBV) for an average sized adult female and 5-6L for an average sized adult male.
- Loss of over 40% of blood volume is immediately life threatening1.
- Massive transfusion (commonly used definitions include): 1. An entire blood volume within a 24 hour period. 2. >50% of blood volume within 3 hours.
- Assessment is a combination of clinical signs and measured blood loss.
- Tachycardia and pallor are key early signs of significant haemorrhage. Severe haemorrhage is indicated by hypotension, altered consciousness and oliguria. Clinical signs are a very important indicator of volume status.
- Blood loss can be observed and measured. Visible blood loss is often overestimated and non-visible blood loss such as retroperitoneal or long bone bleeding can be difficult to quantify2. In the operating theatre blood loss is monitored by close observation of blood in suction equipment and on drapes, and is measured by collection and weighing of blood soaked swabs.
- Keep in mind that medications can cause alteration from normal response patterns. For example β-adrenoceptor antagonists can make heart rate an unreliable sign, while vasodilator drugs including ACE inhibitors and calcium channel antagonists can lead to an exaggerated hypotensive response.
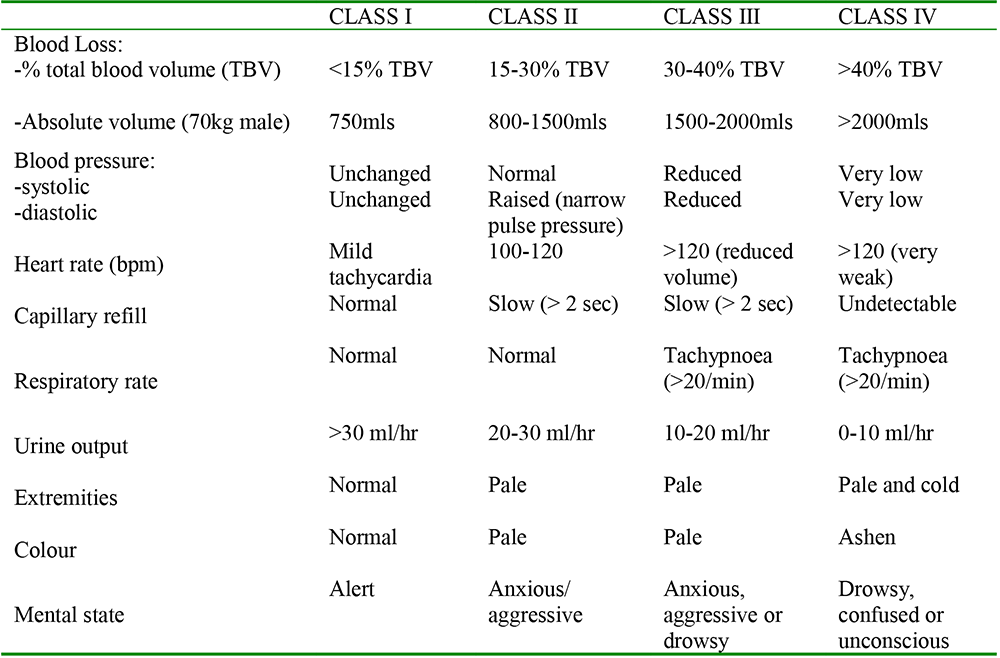
Table 1: Baskett’s classification of hypovolaemic shock3.
ANAESTHETIC CONSIDERATIONS
- High concentration oxygen therapy ensures maximum oxygen saturation of haemoglobin and increased plasma dissolved oxygen. Oxygen may also cause some constriction of peripheral vessels, which may help maintain blood pressure.
- Fluid therapy is used to restore adequate circulating volume. It is important to understand the properties of each fluid option and it’s relevant distribution after administration. For example 0.9% saline is a common choice of crystalloid in resuscitation, however it can contribute to acidosis if large volumes are given (hyperchloraemic acidosis). Start with 20mls/kg of crystalloid while the degree of blood loss is assessed. Consider the use of further crystalloids, colloids, red blood cells and coagulation factors based on clinical situation and potential complications.
- Hb/Hct levels do not fall for several hours after acute major blood loss.
- In haemorrhage, Hb concentration is strongly related to plasma volume. Care should be taken when interpreting the Hb concentration, as it can be misleading if used to judge blood loss or the need for blood replacement. For example, a healthy euvolaemic patient may have a normal Hb concentration of 13g/dl. After 1 litre of blood loss (thus decreasing total volume of both haemoglobin and plasma) this patient may maintain a Hb concentration of 13g/dl. After replacement of the 1 litre blood loss with crystalloid solution this patient can have a restored circulating intravascular volume but their Hb concentration may drop to 9g/dl. Hb concentrations can be misleadingly high prior to resuscitation and fall rapidly during fluid therapy2.
- Evidence suggests that a Hb concentration of 7-9g/dl is acceptable for non-bleeding critically ill patients. Transfusing to a Hb concentration of 8-10g/dl provides a safety buffer for further bleeding2. Patients with heart disease should be kept at the upper end of this range.
- Drugs which cause vasodilation or reduce cardiac output (eg. induction agents, neuraxial anaesthesia) may worsen hypotension and further impair tissue oxygenation.
- Smaller doses of anaesthetic agents are required to produce the usual clinical effect due to a higher proportion of blood flow going to vital organs.
- Hypothermia is avoided through use of warmed IV fluids/blood, forced air warmers, minimising patient exposure, a warm theatre environment and warm surgical irrigation.
- Risk management needs to consider both the direct risk of hypovolaemia and the secondary risks of massive transfusion.
- Source control is often important in minimising the blood loss. If haemorrhage is ongoing, there should be no delay in achieving operative assessment and operative control of bleeding.
- Current evidence suggests the optimal blood pressure to achieve prior to source control is a mean arterial pressure (MAP) of 60-70mmHg. This will maintain organ perfusion while avoiding unnecessary blood loss from damaged vessels. More aggressive restoration of normal blood pressure is achieved after source control is established2.
INITIAL MANAGEMENT OF MASSIVE HAEMORRHAGE
- Good management requires cooperation and communication between all team members (anaesthetist, surgeon, haematologist, blood bank staff, and theatre assistants).
- Intraoperative management of hypovolaemia is coordinated by the anaesthetist.
Good clinical management involves understanding of the physiology of blood loss and appropriate resuscitation, as well as good logistical organisation in a potentially stressful and chaotic environment. Advance warning where possible is very valuable, as this allows for adequate preparation. It is important to maximise staffing levels and to delegate roles. Good communication is essential.
If there is more that one anaesthetist and two or more anaesthetist assistants, the role delegation might include the following:
- Principal anaesthetist to coordinate resuscitation
- Second anaesthetist to focus on the anaesthetic (specifically airway, anaesthesia drug administration, and monitor progress of vital signs).
- First anaesthetic assistant to aid airway management, provision of required drugs, provision of equipment for venous/arterial access, double checking blood products prior to transfusion.
- Second anaesthetic assistant to supervise delivery of blood products (including warming devices, transfusion machines, and documentation of blood products administered).
- A hospital orderly (where available) is very useful for collecting blood products and delivering them to the theatre as required.
- Goals of management are: haemostasis, restoration of circulating blood volume, and blood component replacement.
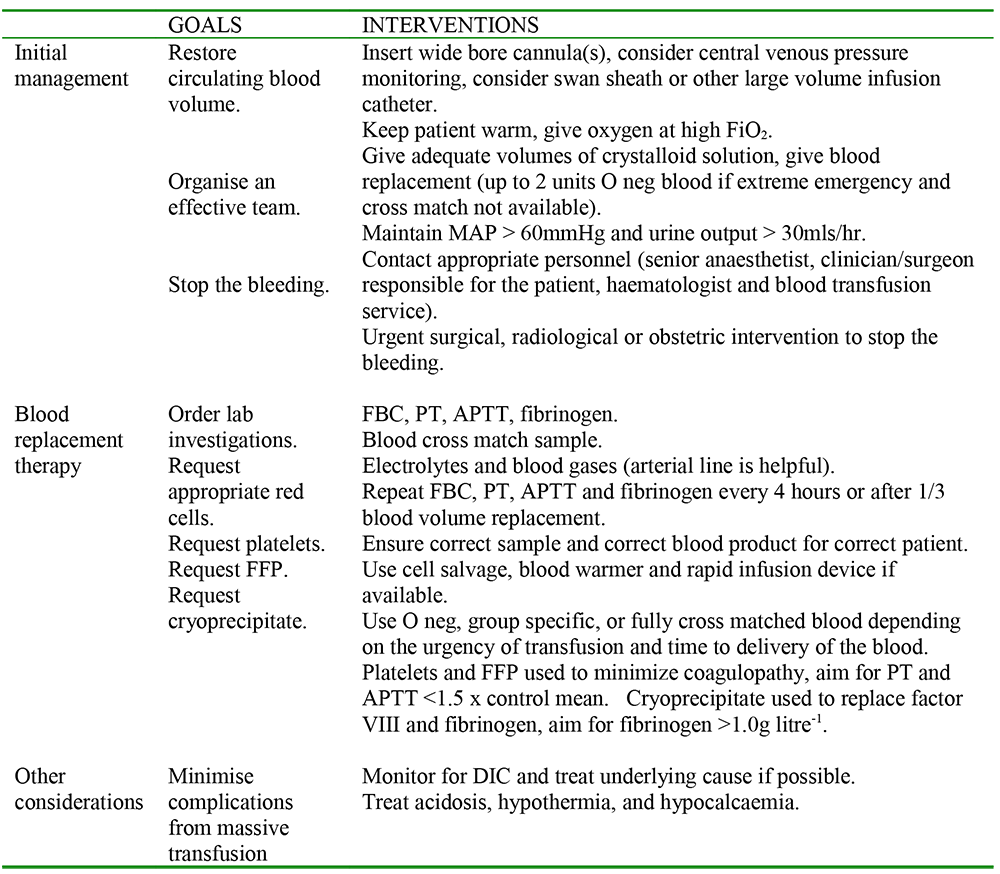
Table 2: Guideline for management of massive blood loss (adapted from reference 1).
BLOOD PRODUCTS
Best practice will consider use of RBC, platelets, FFP and cryoprecipitate to achieve Hct above 24%, platelets above 50 x109 litre-1 and fibrinogen above 0.5-1.0g litre-1.
Packed Red Blood Cells
Packed red cells are produced by removing 150-200ml citrated plasma from a whole unit of blood, and have a shelf life of approximately 35 days. In extreme emergencies O Rh negative blood may be used while awaiting a cross matched blood supply (in males and post menopausal females O Rh positive or negative blood may be used as risk of sensitisation and haemolytic disease of the newborn does not apply). Blood group specific transfusion should be given at the earliest opportunity.
Whole Blood
This is blood prior to fractionation into individual components and it has a shelf life of 35 days. 70ml of citrate preservative is added to 420ml of blood. Whole blood for transfusion is generally only used in centres where blood fractionation into component products is not possible.
Platelets
Platelets are removed from the plasma component of whole blood and they have a shelf life of 3-5 days. Platelets above 50 x109 litre-1 are required to prevent excessive bleeding risk and a higher level of 100 x109 litre-1 has been recommended in patients with high energy trauma or central nervous system injury. Platelet count should be measured at least every 4 hours or after 1/3 blood volume replaced. Initial dose is 4-8 platelet concentrates. Six units usually increases the platelet count by 20-30 x109 litre-1. Anticipating platelet requirement and advance platelet request may be necessary to ensure availability when required.
Fresh Frozen Plasma
FFP is produced by freezing plasma and it lasts for one year. It requires thawing prior to use. FFP contains all the coagulation factors. Coagulation factor depletion is the primary cause of coagulopathy in the setting of major haemorrhage. Fibrinogen falls first (reaching the critical level of 1.0 g litre-1 after 150% blood loss) and later there is a fall in other clotting factors to 25% activity after 200% blood loss1. Use of FFP should be considered after approximately one blood volume has been lost. Aim for PT and APTT <1.5 x control mean. The recommended initial dose is 10-15mls/kg. Sufficient quantity of FFP will correct fibrinogen and most coagulation factor deficiencies but large volumes are often required. If fibrinogen levels remain critically low (below 1.0 g litre-1) after FFP has been given, cryoprecipitate infusion should be considered.
Cryoprecipitate
Cryoprecipitate is obtained by rapidly thawing FFP and separation of cryoprecipitate from albumin/factor IX/immunoglobulins. It is then stored frozen and thawed immediately prior to use. Cryoprecipitate is rich in factor VIII and fibrinogen. In acute haemorrhage, aim for fibrinogen >1.0g litre-1.
Recombinant activated factor VII (rFVIIa)
rFVIIa was originally developed to treat bleeding in haemophiliacs with antibodies to factor VIII and IX. Factor VIIa enhances thrombin generation on the surfaces of platelets. It is a very expensive product and is not licensed for use in acute traumatic haemorrhage. There have been several case reports demonstrating dramatic improvements in the setting of acute haemorrhage, however no randomized controlled trial has yet been published to investigate this application5. While clinical use may be promising, the dosing and clinical indications for use are currently under examination4.
Antifibrinolytics
A large amount of evidence supports the use of antifibrinolytics to treat bleeding in elective general and cardiac surgery4 and they may also be useful in trauma patients. This includes tranexamic acid, ε- aminocaproic acid and aprotinin. The risk of precipitated thrombosis and other complications must be considered.
CONCLUSION
The outcome from massive transfusion has improved over time. This is likely due to active warming measures, aggressive correction of coagulopathy, and decreased operative times for initial surgery. New drugs such as recombinant factor VIIa have increased pharmacologic control over ongoing bleeding, although data is conflicting regarding the long term effect on outcome.
Prevention and management of later complications of massive haemorrhage such as coagulopathy, acidosis, hypothermia and death are important considerations, and will be discussed in detail in a later tutorial of the week.
IMPORTANT POINTS
- Loss of over 40% of total blood volume is immediately life threatening.
- Clinical signs can give a good indication of degree of blood loss.
- The main management goals in major haemorrhage are: haemostasis, restoration of circulating volume, and blood volume replacement.
ANSWERS TO QUESTIONS
- Hypovolaemic shock is inadequate tissue perfusion due to reduced circulating blood volume.
- Normal blood volume is 70mls/kg. That is 5600ml total blood volume for an 80kg person and 4200ml for a 60kg person.
- Clinical signs to assess extent of blood loss include; blood pressure, heart rate, capillary refill, respiratory rate, urine output, mental state, colour of extremities and complexion.
- The main goals of initial management of massive haemorrhage are haemostasis, restoration of circulating blood volume, and blood component replacement.
- Blood products used in severe haemorrhage include red blood cells, fresh frozen plasma, and cryoprecipitate. Albumin, antifibrinolytics and recombinant Factor VIIa are sometimes used.
- The main complications from massive haemorrhage include coagulopathy, acidosis, hypothermia and death.
REFERENCES and FURTHER READING
- Stainsby D, MacLennan S, Hamilton PJ. Management of massive blood loss: a template guideline. Br. J. Anaesth. 2000;85;487-491.
- Marcela C. ABC of transfusion. 4th ed. Wiley-Blackwell; 2009.
- Baskett PJ. ABC of major trauma. Management of hypovolaemic shock. BMJ. 1990;300;1453-1457.
- Spahn DR, Cerny V, Coats TJ, et al. Management of bleeding following major trauma: a Europeanguideline. Crit Care. . 2007;11;R17.
- Spahn DR, Tucci MA, Makris M. Editorial: Is recombinant FVIIa the magic bullet in the treatment of major bleeding? Br. J. Anaesth. 2005;94;553-555.



