Intensive Care Medicine
QUESTIONS
Before continuing, try to answer the following questions (true or false). The answers can be found at the end of the article, together with an explanation.
- The global mortality rate in severe paediatric sepsis is estimated at;
a. 0.1%
b. 0.5%
c. 1%
d. 5%
e. 10% - Criteria included in the definition of systemic inflammatory response in children, are;
a. Pyrexia
b. Hypothermia
c. Leucopaenia
d. A confirmed bacterial infection
e. Tachycardia - Cold shock is:
a. The presenting clinical picture in 50% of children with septic shock
b. Characterized by a normal or low cardiac output and a low SVR
c. Associated with a prolonged capillary refill time
d. Associated with a wide pulse pressure
e. More likely to be associated with central venous catheter infections - The following statements about vasoactive drugs are true;
a. Noradrenaline is the first line agent in paediatric sepsis
b. Dopamine can be run as a peripheral infusion
c. Vasodilators have no role in the treatment of paediatric sepsis
d. Vasopressin is used in paediatric sepsis when Noradrenaline has failed
INTRODUCTION
The aim of this article is to provide the reader with an overview of the current guidelines and evidence for the management of sepsis in children. Sepsis is a major cause of morbidity and mortality in children and although mortality rates are lower in children than in adults, they are estimated at about 10% in severe sepsis.1 In 2003, the World Federation of Paediatric Intensive and Critical Care Societies (WFPICCS) launched an international paediatric sepsis initiative to reduce mortality and morbidity from sepsis in children2 by promoting early diagnosis and guiding effective treatment. The provision of dedicated neonatal and paediatric intensive care units, outreach teams, retrieval teams and the dissemination of guidelines to aid early recognition and treatment has contributed to falling mortality rates in paediatric sepsis.3
DEFINITIONS
Adult systemic inflammatory response syndrome (SIRS) criteria are modified to produce paediatric specific definitions.4
SIRS is a response to a stimulus, which results in two or more of the following:4
- Temperature > 38.5°C or < 36 °C
- Heart rate > two standard deviations above normal, or bradycardia in children < 1 year old (< 10th centile for age)
- Respiratory rate > two standard deviations above normal (or pCO2 < 32 mmHg)
- Leukocyte count > 12,000 cells/mm3, < 4,000 cells/mm3, or > 10% band forms
- Hyperglycaemia, altered mental status, hyperlactaemia, increased capillary refill time (CRT)
Sepsis is SIRS with a suspected or confirmed bacterial, viral, or fungal cause.
Severe sepsis is sepsis and organ hypoperfusion (raised lactate, oliguria, prolonged CRT, reduced mental status) or dysfunction (disseminated intravascular coagulopathy (DIC), acute respiratory distress syndrome (ARDS), acute renal failure (ARF)).
Septic shock is sepsis with fluid refractory hypotension and signs of hypoperfusion.
PRESENTATION
Adult and paediatric shock can be quite different. Adults tend to present with tachycardia, hypotension, low systemic vascular resistance (SVR) and a reduced ejection fraction, but with a relatively maintained cardiac output.5 In children, the sympathetic nervous system responds to sepsis by increasing heart rate and SVR to maintain mean arterial pressure (MAP). Loss of this compensatory mechanism leads to hypotension, usually a late sign. Two thirds of children present in ‘cold’ shock (normal/low cardiac output (CO) and high SVR), adults and the remaining one third of children present in ‘warm’ shock (normal/high CO and low SVR).6 Mortality in children with sepsis is associated with severe hypovolaemia and a low cardiac output (CO). It has been stated that for every extra hour a child remains in shock their mortality rate doubles.7

Table 1. Types of shock
Tissue oxygen delivery is the major limitation to oxygen consumption in children with sepsis and treatment should be targeted to improve this. The use of cardiac output measurements or surrogate measures, such as superior vena cava oxygen saturation (ScvO2) and lactate may act as guides to optimise treatment and improve oxygen delivery.8
Treatment
Consensus guidelines exist for the management of infants and children with septic shock.9 There is some evidence that adherence to these recommendations has improved survival.7,10 The treatment algorithm produced by the American College of Critical Care Medicine (ACCM) is shown in figure 1. Management can be broadly divided into two main phases:
- ABCs: During the first hour of resuscitation, fluid and inotropic drug therapy is directed towards maintaining goals of age appropriate heart rate, blood pressure, and a normal CRT ≤ 2 seconds (table 2). Oxygenation and ventilation should be supported as appropriate.
- Stabilization: beyond the first hour, management should move to an intensive care setting for further haemodynamic support and goal directed therapy. Treatment targets include normal perfusion pressure for age, ScvO2 > 70%, and Cardiac Index (CI) 3.3-6 L/min.
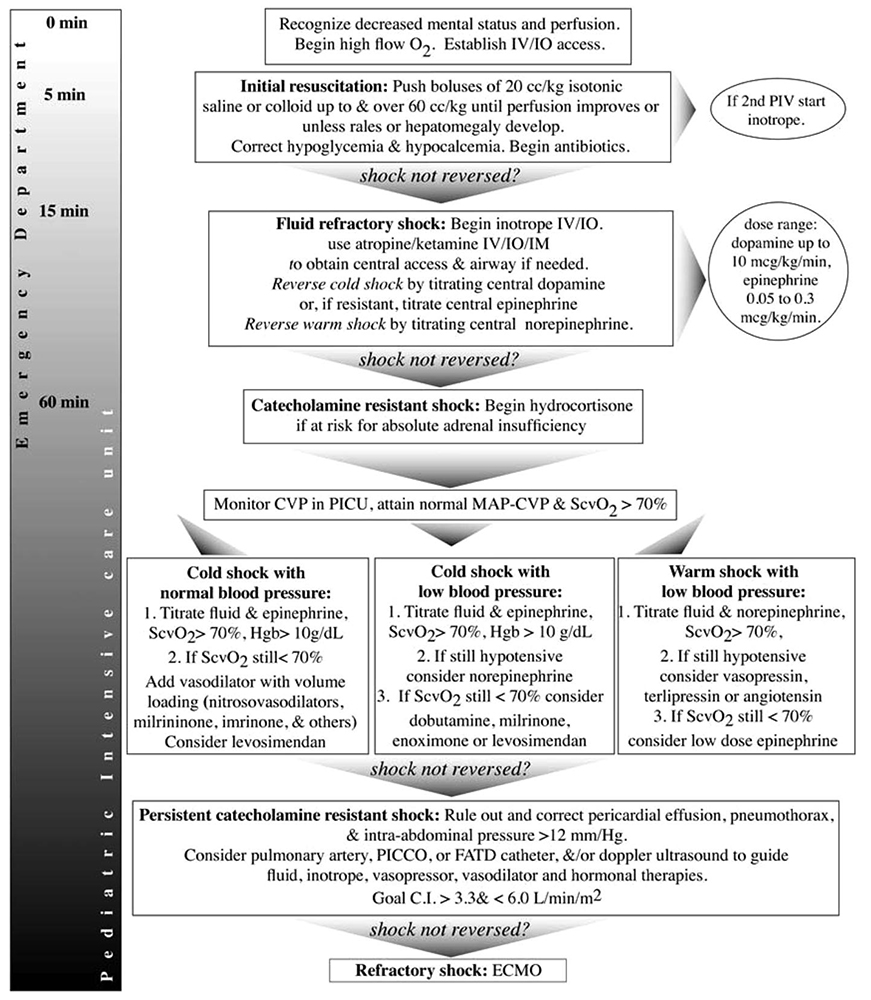
Figure 1. Algorithm for management of paediatric sepsis,9 reproduced with kind permission from the American College of Critical Care Medicine. Brierley J, Carcillo JA, Choong K, Cornell T, DeCaen A, Deymann A et al. Clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock: 2007 update from the American College of Critical Care Medicine. Crit Care Med 2009;37:666-688

Table 2. Age appropriate heart rates and perfusion pressures by age11
SPECIFIC RECOMMENDATIONS
Antibiotics
Antibiotics need to be administered within one hour of identification of severe sepsis, after appropriate cultures have been taken. Early antibiotic therapy and identification of the possible source of infection is critical. Broad-spectrum antibiotics should be commenced first; appropriate to the likely source of infection, the age of the child, and knowledge of local disease prevalence and drug resistant organisms. Antibiotic cover can then be rationalised as the clinical picture, culture results and local microbiology team advice dictates. Therapeutic drug monitoring should be used to ensure adequate target levels and avoid drug toxicity. Courses of antibiotics must be completed and intravenous conversion to oral drugs taken at appropriate stages. Source control strategies are important and include drainage or debridement of infected tissues and removal of infected devices or foreign bodies.
Neonatal sepsis
A distinction can be drawn between early (<72 hrs age) and late (>72 hrs age) phases of neonatal sepsis.12
In early onset neonatal sepsis causative agents are organisms commonly present in the maternal genital tract (e.g. group B streptococcus, Escherichia coli, klebsiella, enterobacter, and listeria monocytogenes).12 Typical broad-spectrum antibiotic cover is ampicillin and gentamicin (or amikacin), with therapeutic drug monitoring.
Late onset neonatal sepsis is due to pathogens in the post-natal environment (e.g. transmission from the care-giver, aspiration of feeds, and central line contamination). Initial broad-spectrum cover is often similar, ampicillin and either gentamicin or amikacin, but if meningitis is suspected then cefotaxime instead of gentamicin is used.12 Vancomycin is used for suspected central line sepsis instead of ampicillin.
Paediatric sepsis
Common infecting organisms include staphylococcus, streptococcus, pseudomonas and meningococcus. Antibiotic choice depends on the likely pathogens involved and should vary depending on clinical presentation, e.g. pneumonia, bloodstream infection, intra-abdominal sepsis or meningitis. Antibiotic regimens need to cover both gram positive and negative organisms.
Anaesthesia and Ventilation
Neonates and infants have a low functional residual capacity and a high work of breathing; early intubation and ventilation must be considered, especially in patients that show little response to aggressive fluid resuscitation and peripheral inotropes.
Induction drugs need to be carefully selected and administered to guard against excessive cardiovascular depression. Avoid large doses of thiopentone, propofol, midazolam and high inspired concentrations of volatile anaesthetic agents. Etomidate is associated with increased severity of illness in septic shock13 and is generally not recommended. Ketamine (1-2mg/kg) is a good alternative. Consider the need for a rapid sequence induction with cricoid pressure, and a nasogastric tube. Pre-oxygenation with 100% oxygen is desirable, but often practically difficult. There is potential for deterioration in cardiovascular parameters at this time and appropriate fluid boluses and inotropes should be prepared in advance.
Maintain sedation and paralysis post intubation and adopt a lung protective ventilator strategy, maintaining low lung volumes (6-7ml/kg tidal volume) with adequate PEEP and low mean airway pressure. Evidence for this is derived from adult practice.
High frequency oscillatory ventilation may be required where conventional ventilation alone proves inadequate.
Fluid resuscitation and intravenous access
Resuscitation should begin with boluses of 10-20ml/kg of crystalloid or 5 % albumin over 5-10 minutes with further aliquots titrated to clinical condition (e.g. heart rate, urine output, CRT and level of consciousness). Aggressive fluid resuscitation is a key stage to improved survival, provided there is also access to inotropic therapy and mechanical ventilation.14 Large fluid deficits are common and volumes of over 40-60ml/kg can often be required (but see below for resuscitation in special circumstances).
The optimal choice of fluid is not known, and a recent systematic review of resuscitation fluid in children was unable to find evidence to support the use of colloid over crystalloid.15 A large randomised study in adults, the SAFE trial, compared crystalloid and albumin fluid resuscitation, finding a trend towards improved outcomes in septic shock with albumin.16 The 2007 updated consensus guidelines 9 suggest a preference towards the use of colloid resuscitation and there are two particular studies that support this in children.17,18
Malnourished children are a special category of patients who do not tolerate aggressive fluid resuscitation, as they are at greater risk of congestive heart failure from over-hydration. Septic shock can be difficult to recognize and treat in these patients. Malnourished children require slow IV rehydration with careful and regular observation (every 5-10minutes). An infusion of 15ml/kg Ringer’s lactate 5% dextrose should be given over one hour; if there are signs of improvement, a repeat bolus can be given slowly, followed by oral or nasogastric rehydration. If the patient does not improve after one hour, a blood transfusion should be considered (10ml/kg slowly over three hours). If the child deteriorates during treatment (increased respiratory rate or heart rate) the infusion should be stopped.19
The practice of high-volume fluid resuscitation in sepsis has been challenged by the recent ‘Fluid Expansion As Supportive Therapy’ (FEAST) study, which investigated fluid resuscitation in children with a diagnosis of sepsis (but without hypotension), in a large cohort of children in Uganda, Kenya, and Tanzania.20 Resuscitation with a fluid bolus of 20-40 ml/kg saline or albumin was compared to the local practice of no fluid bolus resuscitation. The results of the study were surprising: the fluid bolus groups had increased mortality at 48 hours and a 4% higher risk of death and neurologic sequelae at four weeks compared to the children who did not receive a fluid bolus. Most deaths were early, 87% occurring in the first 24 hours. The study included many children with malaria (57%), severe anaemia (32%), hypoxia (25%) or coma (15%), and 6% had hypotension. This may represent a population in whom over hydration will not be well-tolerated, particularly if mechanical ventilation and inotropic support are not available. The implications of the FEAST study are not completely clear at present, but suggest that the traditional recommendation of aggressive bolus fluid resuscitation should not be used in children with severe anaemia or malaria, or other common febrile illness associated with a significant stress response but not hypotension (i.e. associated with ADH release and fluid retention). Particular caution should be used when using aggressive fluid resuscitation in patients in low-income countries in the absence of mechanical ventilation and inotropic support.21
Intravenous access is often difficult to achieve in critically ill children. Early intra-osseous access should be considered to avoid repeated or prolonged attempts at venepuncture and enable resuscitation to begin in a timely manner. In children with fluid refractory shock, central venous and arterial pressure monitoring can guide on-going resuscitation. Ultrasound guidance can be a useful tool to facilitate this.
Inotropic and vasoactive drug therapy
In fluid refractory shock, persistent hypotension is treated with either inotropes, vasopressors or a suitable combination of both. Regular re-assessment of the child with appropriate changes to the choice and rate of cardiovascular drug used is essential.
Dopamine is the first line agent. If central venous access will delay starting inotropes then the American College of Critical Care Medicine guidelines recommend the use of peripheral inotropes (not vasoconstrictors) with close monitoring of the IV access site to prevent extravasation injury.9
Subsequent inotropic support depends on the clinical presentation of the child; low cardiac output and high SVR (cold shock), high cardiac output and low SVR (warm shock), or low cardiac output and low SVR. Adrenaline may need to be added to dopamine refractory shock in cold shock and noradrenaline used with warm shock (table 3 gives guide infusion rates). Other agents to consider are the use of vasodilators (e.g. sodium nitroprusside or glyceride tri-nitrate) or phosphodiesterase inhibitors (e.g. milrinone) in the case of low CO and high SVR whilst on adrenaline infusion. Vasopressin is used in adult practice for the treatment of extremely low SVR despite high doses of noradrenaline, but there is currently no clear evidence for its use in paediatrics.22

Table 3. Recommended infusion rates
Therapeutic end points
In the first hour, the aim of resuscitation should be to achieve normalisation of heart rate, a capillary refill time ≤ 2 seconds, normal pulses with no differential between central and peripheral, warm extremities, urine output ≥ 1 ml/kg/hr, and normal mental status. Progress towards these targets can be used to monitor the progress of resuscitation.
Early goal directed therapy originated in the management of severe sepsis in adults and has been shown to have the largest mortality benefit of any sepsis study.23 Timely use of cardiac output monitoring and surrogate markers of organ perfusion is recommended in the management of paediatric sepsis, including lactate, improved base deficit, ScvO2 ≥ 70% or SvO2 ≥ 65%, CVP 8-12 mmHg or cardiac output monitoring (CO 3.3-6 L/min/m2).
An indirect measure of oxygen delivery can be made using ScvO2 and a study of children with sepsis compared the use of the ACCM guidelines with and without the goal of ScvO2 > 70%.8 When this goal directed approach was used, patients received more crystalloid, blood and inotropic support, resulting in a reduction in 28-day mortality from 39.2% to 11.8% in the ScvO2 monitored group. Normalising lactate clearance may be as effective as the use of ScvO2 as a resuscitation goal in the initial treatment of sepsis.24
Cardiac output monitoring in the form of echocardiography, trans-oesophageal Doppler, pulse contour analysis, or supra-sternal ultrasound cardiac output monitors can be helpful. Blood flow is difficult to determine clinically and blood pressure is a poor substitute, as it is affected by both cardiac output and by systemic vascular resistance. There is no good evidence for improved outcome with any of these monitoring tools, only observational data. A large multi-centre randomised controlled trial is needed.
Steroids
Evidence for the use of steroids in paediatric sepsis is lacking. A randomised controlled trial in children with septic shock is required and until then steroids should not be used routinely.25 Current retrospective studies of steroids in children with severe sepsis have shown their use to be an independent predictor of increased mortality.26
Hydrocortisone therapy is reserved for children with catecholamine resistance and suspected or proven adrenal insufficiency. Children at risk of adrenal insufficiency should be treated with steroids, but the recommended doses of hydrocortisone vary; the dose for stress cover is 1-2mg/kg/day whilst that for shock reversal is 50mg/m2/24hrs. Note the different units for these doses. Those at risk of adrenal insufficiency include children taking steroids for chronic disease, those with pituitary or adrenal abnormalities, and cases of catecholamine resistant severe septic shock. Adrenal insufficiency can be identified by random blood cortisol levels < 18 mcg/dl or a cortisol level increase of < 9 mcg/dl after an ACTH stimulation test.
DVT prophylaxis
Older post-pubertal children should have appropriate measures considered, unfractionated or low-molecular weight heparin or mechanical prophylactic devices such as compression stockings. In young children the majority of thrombotic events are associated with the use of central venous catheters, there is some evidence that heparin-bonded central venous lines may reduce thrombosis rates.27 A multicentre randomised controlled trial underway at present, is looking at catheter related infections in children and comparing the effectiveness of heparin bonded catheters and antibiotic impregnated catheters for the prevention of hospital acquired blood stream infections (CATCH trial28). A sidearm of this study will investigate the incidence of thrombosis.
Stress ulcer prophylaxis
Early enteral feeding or, where this is not possible, stress ulcer prophylaxis with H2 blockers or proton pump inhibitors should be used routinely in patients with severe sepsis. This is aimed at reducing the risk of GI bleeds and at the prevention of ventilator-associated pneumonia.
H2 blockers were previously used in nil-by-mouth neonates, but there is now evidence of increased risk of infections, necrotising enterocolitis and fatal outcome.29
Glycaemic control
Blood glucose should be kept within the normal range. Hypoglycaemia can cause neurological damage and should be treated promptly with 2ml/kg of 10% dextrose. Neonates will require a 10 % dextrose infusion (8 mg/kg/min) whilst children and adolescents have lower requirements, 5 mg/kg/min and 2mg/kg/min respectively. Requirements may be higher in children with metabolic disease or liver failure.
Hyperglycaemia is also a risk factor for increased mortality30 and should be treated with an insulin infusion and frequent glucose monitoring to avoid inadvertent hypoglycaemia.
Transfusion
Consider targeting transfusion to a haemoglobin goal of greater than 10 g/dl to achieve an ScvO2 > 70%, so enhancing oxygen delivery.9
After the initial resuscitation is complete, we do not know the best haemoglobin level for critically ill children. Common practice is to use conservative blood transfusion thresholds to reduce potential risks and complications. The transfusion strategies in paediatric intensive care units (TRIPICU) study looked at transfusion thresholds in stable, critically ill children.31 They found that a haemoglobin threshold of 7 g/dl decreased transfusion requirements without increasing adverse outcomes.
Renal replacement therapy
With large volumes of initial resuscitation fluid being given and often an on-going fluid requirement due to capillary leak, there can be significant tissue oedema and fluid overload. Diuretics, peritoneal dialysis or renal replacement therapy may be required once the child has been stabilised. Early implementation of continuous renal replacement therapy is associated with improved survival compared to late implementation.32
Protein C and Activated protein C (APC)
Use of APC in children is not recommended due to a lack of evidence of benefit and an increase in bleeding complications.33
Extra-corporeal membrane oxygenation (ECMO)
ECMO may be considered in those cases of severe septic shock, which have not responded to all conventional treatment strategies,34 where it may be associated with improved survival.
Intravenous Immunoglobulin
Intravenous immunoglobulin therapy may be associated with a reduction in mortality and should be considered in severe sepsis.34
CONCLUSION
Mortality rates in paediatric sepsis have fallen due to improvements in its management. Early recognition and aggressive treatment in line with protocol driven algorithms (such as the guidelines produced by the ACCM) form the mainstay of initial management. Subsequent early referral and transfer to intensive care units for on-going care, and the use of early goal directed therapy are essential to improve outcomes. For many therapeutic interventions there is a paucity of good supporting evidence. The guidelines provided by the ACCM provide an expert consensus approach to the management of septic children.
There is some emerging evidence that children in low-income countries represent a patient cohort in whom traditional recommendations of fluid resuscitation may not be applicable.
ANSWERS TO QUESTIONS
- FFFFT
- Although mortality rates are lower in children than in adults, they are estimated at about 10% in severe sepsis.
- TTTFT
- The presentation of SIRS can include a temperature of greater than 38.5 °C or < 36 °C, a tachycardia or bradycardia, tachypnoea and either a leucocytosis or leucopaenia. When these signs are associated with a confirmed bacterial, viral or fungal infection, this is termed sepsis.
- FFTFF
- Approximately two thirds of children present in cold shock characterised by a normal or lowered cardiac output and a high SVR. In this setting the CRT is prolonged and there is a narrow pulse pressure. Cold shock is normally associated with community-acquired sepsis, whilst central venous catheter infections tend to present with the features of warm shock.
- FTFF
- Dopamine is generally recommended as the first line inotropic agent in paediatric sepsis, and its commencement should not be delayed by a lack of central venous access. Where dopamine is run as a peripheral infusion extreme vigilance and monitoring of the iv infusion site is mandated. Vasodilators can be useful in the setting of a low cardiac output, coupled with a high SVR whilst on an adrenaline infusion. There is no evidence for the use of vasopressin in the treatment of paediatric sepsis.
REFERENCES and FURTHER READING
- Angus DC, Linde-Zwirble WT, Liddicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the US: Analysis of incidence, outcome and associated costs of care. Crit Care Med 2001; 29:1303-1310
- International Paediatric Sepsis Initiative. World Federation of Paediatric Intensive and Critical Care Societies. Accessed on 7/5/2012; http://www.wfpiccs.org/sepsis.php
- Carcillo JA. What’s new in pediatric intensive care. Crit Care Med 2006; 34: S183-S190
- Goldstein B, Giroir B, Randolph A. International pediatric sepsis consensus conference: Definitions for sepsis and organ dysfunction in paediatrics. Pediatr Crit Care Med 2005;6:2-8
- Aneja RK, Carcillo JA. Differences between adult and pediatric septic shock. Minerva Anestesiologica 2011; 77:986-92
- Brierley J, Peters MJ. Distinct hemodynamic patterns of septic shock at presentation to pediatric intensive care. Pediatrics 2008; 122:752-759
- Han YY, Carcillo JA, Dragotta MA, Bills DM, Watson RS, Westerman ME et al. Early reversal of pediatric-neonatal septic shock by community physicians is associated with improved outcome. Pediatrics 2003;112: 793-799
- de Oliveira CF, de Oliveira DSF, Gottschald AFC, Moura JDG, Costa GA, Ventura AC et al. ACCM/PALS haemodynamic support guidelines for paediatric septic shock: an outcomes comparison with and without monitoring central venous oxygen saturation. Intensive Care Med 2008;34:1065-1075
- Brierley J, Carcillo JA, Choong K, Cornell T, DeCaen A, Deymann A et al. Clinical practice parameters for hemodynamic support of pediatric and neonatal septic shock: 2007 update from the American College of Critical Care Medicine. Crit Care Med 2009;37:666-688
- Inwald DP, Tasker RC, Peters MJ, Nadel S. Emergency management of children with severe sepsis in the United Kingdom: the results of the paediatric intensive care society sepsis audit. Arch Dis Child 2009; 94: 348-353
- Report of the second task force on blood pressure control in children-1987. Task force on Blood Pressure Control in Children. National Heart, Lung, and Blood Institute, Bethesda, MD. Pediatrics 1987; 79:1-25
- Aneja RK, Varughese-Aneja R, Vetterly CG, Carcillo JA. Antibiotic therapy in neonatal and pediatric septic shock. Curr Infect Dis Rep 2011; 13: 433-441
- Annane D. ICU physicians should abandon the use of etomidate! Intensive Care Med 2005;31:325-326
- Carcillo JA, Tasker RC. Fluid resuscitation of hypovolemic shock: Acute medicine’s great triumph for children. Int Care Med 2006; 32: 958-961
- Akech S, Ledermann H, Maitland K. Choice of fluids for resuscitation in children with severe infection and shock: systematic review. BMJ 2010; 341: c4416
- Finfer S, Bellomo R, Boyce N, French J, Myburgh J, Norton R et al. SAFE study investigators: A comparison of albumin and saline for fluid resuscitation in the intensive care unit. NEJM 2004; 350:2247-2256
- Maitland K, Pamba A, English M, Peshu N, Marsh K, Newton C et al. Randomised trial of volume expansion with albumin or saline in children with severe malaria: Preliminary evidence of albumin benefit. Clin Infect Dis 2005; 40: 538-545
- Booy R, Habibi P, Nadel S, de Munter C, Britto J, Morrison A et al. Meningococcal research group: Reduction in case fatality rate from meningococcal disease associated with improved healthcare delivery. Arch Dis Child 2001; 85: 386-390
- WHO. Chart 8. How to give IV fluids for shock in a child with severe malnutrition. In: Pocket Book of Hospital care for children. Guidelines for the management of common illnesses with limited resources. p13 2005 World Health Organization
- Maitland K, Kiguli S, Opoka RO, Engoru C, Olupot-Olupot P, Akech SO et al. Mortality after fluid bolus in African children with severe infection. NEJM 2011; 364: 2483-95
- Duke T. What the African fluid bolus trial means. Lancet 2011 378; 1685-7
- Meyer S, McGuire W, Gottschling S, Shamdeen GM, Gortner L. The role of vasopressin and terlipressin in catecholamine-resistant shock and cardio-circulatory arrest in children: Review of the literature. Wein Med Wochenschr 2011; 161: 192-203
- Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. NEJM 2001; 345: 1368-77
- Jones AE, Shapiro NI, Trzeciak S, Arnold RC, Claremont HA, Kline JA et al. Lactate clearance vs. central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA 2010; 303: 739-46
- Markovitz BP, Goodman DM, Watson S, Bertoch D, Zimmerman J. A retrospective cohort study of prognostic factors associated with outcome in pediatric severe sepsis: What is the role of steroids. Pediatr Crit Care Med 2005;6:270-274
- De Kleijn ED, Joosten KF, Van Rijn B, Westerterp M, De Groot R, Hokken-Koelega A et al. Low serum cortisol in combination with high adrenocorticotrophic hormone concentrations is associated with poor outcome in children with severe meningococcal disease. Pediatr Infect Dis J 2002;21:330-336
- Pierce CM, Wade A, Mok Q. Heparin-bonded central venous lines reduce thrombotic and infective complications in critically ill children. Intensive Care Med 2000;26:967-972
- Catheter infections in children; CATCH trial. Accessed on 24/06/12: http://www.catchtrial.org.uk/index.html
- Terrin G, Passariello A, De Curtis M, Manguso F, Salvia G, Lega L et al. Ranitidine is associated with infections, necrotizing enterocolitis and fatal outcome in newborns. Pediatrics 2012; 129: e40-45
- Lin JC, Carcillo JA. Increased glucose/glucose infusion rate ratio predicts anion gap acidosis in pediatric sepsis. Crit Care Med 2004;32(Suppl 20):A5
- Lacroix J, Hebert PC. Transfusion strategies for patients in paediatric intensive care units. NEJM 2007;356:1609-1619
- Foland JA, Fortenberry JD, Warsaw BL, Pettignano R, Merritt RK, Heard ML et al. Fluid overload before continuous hemofiltration and survival in critically ill children: A retrospective analysis. CritCare Med 2004; 32: 1771-1776
- Nadel S, Goldstein B, Williams MD, Dalton H, Peters M, Macias WL et al. Drotrecogin alfa (activated) in children with severe sepsis: A multicentre phase III randomized controlled trial. Lancet 2007; 369: 836 – 843
- Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R et al. Surviving Sepsis Campaign: International guidelines for management of severe sepsis and septic shock. Int Care Med 2008; 34: 17-61



