General Topics
KEYPOINTS
- Patients are living longer with metastatic malignancy and are presenting with metastatic spinal disease (MSD) in greater numbers
- Metastatic spinal disease surgery (MSDS) aims to improve pain, preserve neurological function, prevent pathological fractures and correct any spinal instability. Surgery does not improve survival.
- Anaesthesia for patients with MSD is challenging. Anaesthetic considerations include management of potentially difficult airway, haemorrhage, venous thromboembolism (VTE), hypercalcaemia and chemotherapy toxicities.
- MSDS is commonly associated with significant haemorrhage with an average blood loss of 2 litres. Haemostatic adjuncts, blood conservation strategies, preoperative embolisation and intraoperative cell salvage should be considered.
- MSDS requires multimodal perioperative analgaesia, and this could involve the use of gabapentinoids, selective COX inhibitors, clonidine, ketamine and regional catheters.
INTRODUCTION
Advances in cancer management have improved the life expectancy of patients with metastatic disease. Osseous metastases are the third most common site of metastasis after lung and liver with the majority of bony metastases affecting the vertebral column. Some studies estimate that up to 40% of all patients with cancer will have spinal metastases during the course of their disease1. As a result of this, an increased number of patients with spinal metastases are being considered for spinal surgery.
Treatment of MSD aims to improve pain, preserve neurological function, prevent pathological fractures and correct any spinal instability to improve quality of life. In this article we aim to give an outline of spinal surgery for spinal metastases and discuss in detail the specific challenges this poses to the anaesthetist.
Pathophysiology of metastatic spinal disease
Approximately 70% of osseous spinal metastases involve the thoracic spine, 20% the lumbar spine and 10% the cervical spine. The most common cancers resulting in osseous metastases are breast, lung, gastrointestinal tract, prostate and myeloma2. The major mechanism for metastasis is thought to be via the haematogenous route where direct invasion of blood vessels by the primary tumour results in cancer cells being transported to other parts of the body. It is postulated that retrograde venous spread via the Batson plexus during Valsalva manoeuvres may also play a role. Although rarer, direct invasion of the primary tumour into the spine is also a possibility.
The median survival of patients with spinal metastases is 10 months although this figure is continually improving and there is variation depending upon the primary tumour type. Spinal metastasis is one of the leading causes of morbidity in cancer patients, with complications including hypercalcaemia, pain, spinal instability and pathological fractures. Neurological demise can affect bladder and bowel continence and significantly reduce mobility. These morbidities can result in a significantly reduced quality of life in addition to the limited life expectancy.
Management of metastatic spinal disease
Management options for MSD include non-surgical treatments such as braces, steroids, bisphosphonates, radiotherapy and chemotherapy. Surgical approaches range from minimally invasive verterbroplasty or screw fixation to major open debulking and spinal reconstruction. The optimal management of patients with MSD involves a multi-disciplinary approach involving medical and radiation oncologists, radiologists, spinal surgeons and anaesthetists.
Surgery for metastatic spinal deposits has not been shown to improve survival and the intended benefit is to improve quality of life by improving pain, preventing pathological fractures, preserving neurological function and improving spinal stability. When deciding whether to undertake surgery for MSD, the improvement in quality of life must be weighed against the risks and potential morbidity of surgery. Complications from major spinal surgery can be as high as 25%, with the most common being wound infection2. In patients with a limited life expectancy, any complication from surgery and the extra time needed to overcome these complications can negate any potential benefit.
Various scoring systems can be used to guide whether surgery should be undertaken and if so, the extent of the surgery to undertake2. The fittest patients with good overall prognosis from their underlying malignancy and a greater potential for spinal instability are considered for the most extensive surgery, whereas patients in whom the overall prognosis is poor and the metastases are extensive are only considered for limited if any surgery2.
ANAESTHETIC CONSIDERATIONS
Patients undergoing surgery for MSD can have multiple organ impairment occurring through the sequelae of serial cancer progression: this includes metastases, metabolic derangements and immunosuppression due to chemoradiotherapy. In addition to these patient factors, surgery can be lengthy, extensive, involve special positioning and have the potential for major haemorrhage. Consequently, providing anaesthesia for metastatic spinal surgery has special considerations which are discussed below.
Airway management
Airway management may be difficult, particularly with cervical spine instability secondary to metastases. Radiationinduced fibrosis may make airway manipulation more difficult. Reinforced endotracheal tubes (ETT) are commonly used and in our practice, we routinely perform asleep fibreoptic intubations to minimise neck movement and confirm ETT position to avoid bronchial intubation when in prone position. Tape should be considered over ties when securing the ETT, as ties may impair the venous drainage of the head and neck potentially worsening facial and airway oedema.
Airway oedema can be a significant problem on extubation. Factors that make airway oedema more likely are cervical spine surgery and prone surgery lasting greater than 5 hours3.
Prone positioning
Prolonged prone positioning is often required for MSDS. Prone positioning can lead to important complications which are outlined in table 1.

Table 1 Complications of prolonged prone positioning3, 4
Risk of massive haemorrhage
MSDS has a significant risk of massive haemorrhage. A recent meta-analysis found an average estimate of 2180 mls of blood loss during MSDS5. The extent of haemorrhage is dependent on many factors including the extent of surgery, the vascularity of the tumours, the site and size of the tumours and prone positioning.
Preoperative selective angiography is useful in identifying the vascularity of the tumours and selective embolisation is important in reducing the perioperative blood loss. As the mechanism for metastasis is via haematogenous spread, metastases commonly occur in areas with good vascular supply, increasing the propensity for haemorrhage.
In addition to embolisation, other strategies for control of haemorrhage include preoperative optimisation of haemoglobin, acute normovolaemic haemodilution, intraoperative cell salvage, tranexamic acid and topical haemostatic agents (figure 1). In a recent meta-analysis, tranexamic acid has been found to reduce bleeding without increasing the risk of VTE during spinal surgery6 and this is thought to be the case even in the setting of malignancy.
Despite haemostatic measures, significant blood loss commonly occurs during MSDS. Studies have shown an increased risk of postoperative infection and progression of tumour growth associated with allogeneic blood transfusion, thought to be secondary to the associated immunosuppression7. As a result, there has been significant interest in the use of cell salvage in MSDS. The use of cell salvage has been variable and the subject of significant debate due to the concerns of transfusing salvaged malignant cells and the possibility of contributing to metastatic spread. A recent systematic review found numerous studies describing the use of cell salvage with a leucodepletion filter as a safe technique in oncological surgeries7. We have not found studies specifically pertaining to MSDS however we postulate that similar concepts will likely apply.
COMPLICATIONS ASSOCIATED WITH METASTATIC MALIGNANCY
Venous Thromboembolism (VTE)
20% of all newly diagnosed VTEs are in patients with malignancy and carry a significant associated mortality. Prophylactic VTE measures include low molecular weight heparin (LMWH), compression stockings and automatic calf compression devices. Prophylactic LMWH must not be given in the 12 hours preceding surgery and should be withheld until at least 6 hours following surgery when the risk of VTE versus post-operative bleeding must be weighed up. In contrast, compression stockings and calf compression devices can continue throughout the intraoperative and perioperative period. In patients who have had a VTE within 1 month of planned surgery, perioperative removable IVC filters are recommended8. After surgery, IVC filters should be removed and traditional anticoagulation restarted as soon as possible.
Hypercalcaemia
All patients with metastatic malignancy should have calcium levels checked preoperatively. Hypercalcaemia should be corrected prior to surgery with infusions of 0.9% NaCl, furosemide and bisphosphonates. If left untreated, hypercalcaemia can cause shortened PR interval, shortened QTc, prolonged QRS and cardiac conduction abnormalities (Figure 2). It can also cause muscle weakness necessitating lower doses of non depolarising muscle relaxants.
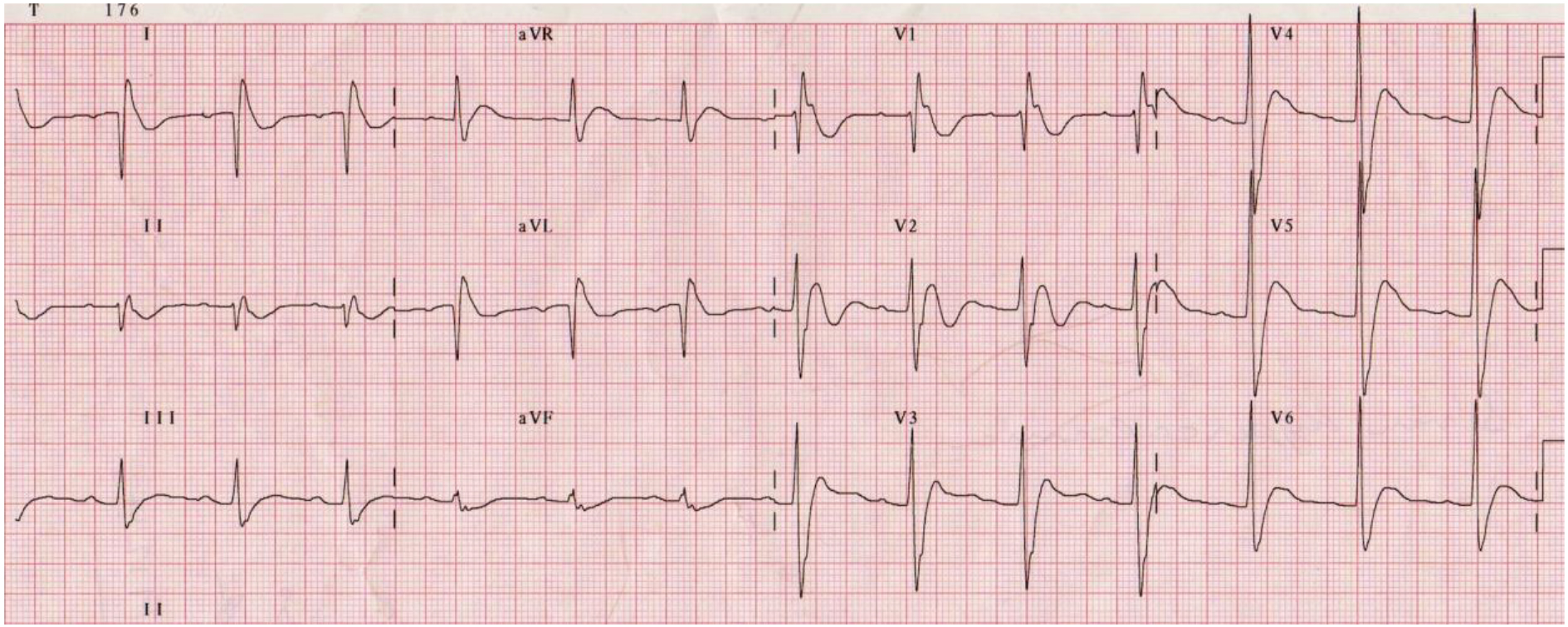
Figure 2. ECG changes in severe hypercalcaemia: Broadened QRS, Short QT interval and J waves- best seen in lead V1 (Taken with permission from ECG library www.lifeinthefastlane.com)
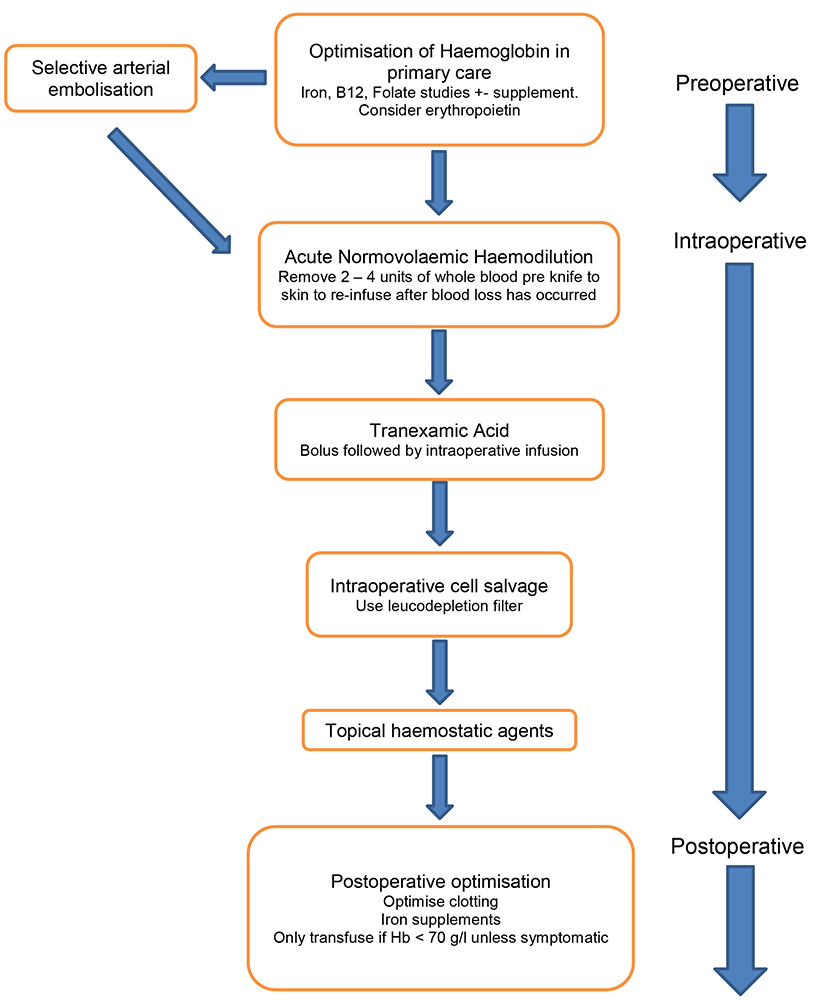
Figure 1. Blood Conservation Strategies
Chemotherapy
Chemotherapy can affect any organ system and in-depth discussion is beyond the scope of this article. However, knowledge of chemotherapy agents and the potential adverse effects is an important perioperative consideration. See Table 2 for potential chemotherapy toxicities which could affect perioperative management. It is important to note that most chemotherapy agents can lead to the mentioned toxicities, only the common offenders are listed below.

Table 2 Important perioperative considerations of complications associated with chemotherapy9
Minimising further metastatic spread
Impaired immunity has been shown to result in faster tumour growth and metastatic spread. It is becoming clearer that perioperative immunosuppression from the surgical stress response is an important factor in disease progression and anaesthesia may play a part in modulating perioperative immunosuppression10, 11. The relationship between anaesthesia, perioperative immunosuppression and cancer progression is the focus of much research at present but there remain many unanswered questions.
Data from retrospective studies suggest regional anaesthesia is associated with reduced disease progression rates when compared with general anaesthesia11. This effect is postulated to be due to regional techniques not impairing cellular immunity, particularly the function of Natural Killer (NK) cells that normally act to protect against metastatic dissemination. Regional techniques also reduce opiate requirements and opiates are known to impair NK cells. However, well-conducted randomised controlled trials are lacking to prove these findings. Currently there is little evidence that induction agents used in standard doses are potential culprits for disease progression. In-vitro and invivo animal studies on volatile agents have also produced mixed results10.
Opiates have long been known to impair NK cell function but in-vitro and animal studies have yielded mixed results10. A retrospective cohort study, in patients with breast cancer, undergoing mastectomy and axillary clearance, compared general anaesthesia with morphine PCA to general anaesthesia with a paravertebral block catheter with a paravertebral infusion12. This small study found improved survival with regional techniques. From this, a causal relationship was hypothesised that morphine-based analgesia may be detrimental to survival and a randomised controlled trial is currently underway to investigate this hypothesis.
Animal and human studies have shown a benefit of NSAIDs in the prevention and spread of cancer. A retrospective study of patients undergoing mastectomy for breast cancer showed a lower rate of recurrence with ketorolac administration10. However, randomised controlled trials are still lacking.
In summary it is clear that impaired immunity in the perioperative period plays a role in disease progression in metastatic cancer. It is less clear how anaesthetic techniques modulate perioperative immunity and disease progression.
PAIN MANAGEMENT
Good quality effective perioperative analgesia is always important for any surgery. Extensive surgical wounds on a background of chronic cancer pain can make perioperative analgesia in MSDS challenging. Effective perioperative analgesia in MSDS can reduce immobility and hence reduce the risk of respiratory infections and venous thromboembolism. Good quality perioperative analgesia may also reduce chronic post-surgical pain13 and as the intended benefit of MSDS is to improve quality of life, this is an important consideration.
Preoperatively patients are often on complex analgesic regimes for chronic cancer pain. These regimens often include high dose opioids. Patients’ narcotics and analgesics should be continued perioperatively unless contraindicated. Patient controlled opiate analgesia is a valuable option for postoperative pain. However decreasing opiate consumption using a multimodal approach is theoretically important as previously discussed.
Perioperative NSAIDs, gabapentinoids, paracetamol, regional blockade and ketamine can all be used. Bleeding risk and renal impairment need to be evaluated before NSAID administration. Perioperative gabapentinoids have been shown to reduce early postoperative pain, opiate consumption and chronic post surgical pain13. Regional blockade including neuraxial techniques, can be considered but the evidence is currently lacking. A recent systematic review of analgesia following major spinal surgery found no benefit for most regional anaesthetic techniques14. Trials using ketamine after major spinal surgery have demonstrated decreased overall opioid consumption14. In conclusion, analgesic regimes need to be tailored to the patient but should include a multimodal approach as detailed here to achieve good quality pain relief whilst at the same time minimising opiate consumption.
CONCLUSION
We have presented the main anaesthetic considerations when undertaking surgery for spinal metastases. As detailed in this article, MSDS can be complex and confer significant perioperative risk. Multidisciplinary approach can help weigh intended benefits versus potential complications of surgical interventions.
REFERENCES AND FURTHER READING
- Joaquim AF, Powers A, Laufer I et al An update in the management of spinal metastases. Arq Neuropsiquiatr. 2015 Sep;73(9):795-802
- Choi D, Crockard A, Bunger C et al Review of metastatic spine tumour classification and indications for surgery: the consensus statement of the Global Spine Tumour Study Group Eur Spine J (2010) 19:215–222
- Nowicki R, Anaesthesia for major spinal surgery Contin Educ Anaesth Crit Care Pain (2014) 14 (4): 147-152
- DePasse J M, Palumbo M A, Haque M et al Complications associated with prone positioning in elective spinal surgery World J Orthop. 2015 Apr 18; 6(3): 351–359.
- Chen Y, Tai BC, Nayak D et al Blood loss in spinal tumour surgery and surgery for metastatic spinal disease: a meta-analysis. Bone Joint J. 2013 May;95-B(5):683-8.
- Li ZJ, Fu X, Xing D et al Is tranexamic acid effective and safe in spinal surgery? A meta-analysis of randomized controlled trials. Eur Spine J. 2013 Sep;22(9):1950-7.
- Kumar N, Chen Y, Zaw AS et al Use of intraoperative cell-salvage for autologous blood transfusions in metastatic spine tumour surgery: a systematic review Lancet Oncol 2014; 15: e33–41
- British Committee for Standards in Haematology: Writing group: Baglin TP, Brush J, Streiff M, Guidelines on use of vena cava filters British Journal of Haematology, 2006 134, 590–59
- Livshits Z, Rao RB and Smith SW An Approach to Chemotherapy-Associated Toxicity Emergency Medicine Clinics of North America 2013 1, 32, 167 – 203
- Bharati SJ, Chowdhury T, Bergese SD et al Anesthetics impact on cancer recurrence: What do we know? J Can Res Ther 2016;12:464-8
- Tedore T, Regional anaesthesia and analgesia: relationship to cancer recurrence and survival. Br J Anaesth 2015; 115 (suppl_2): ii34-ii45.
- Exadaktylos AK, Buggy DJ, Moriarty DC et al Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology. 2006 Oct; 105(4): 660–664.
- Schmidt PC, Ruchelli G, Mackey SC et al Perioperative gabapentinoids: choice of agent, dose, timing, and effects on chronic postsurgical pain. Anesthesiology. 2013 Nov;119(5):1215-21
- Sharma S, Balireddy RK, Vorenkamp KE et al Beyond opioid patient-controlled analgesia: a systematic review of analgesia after major spine surgery. Regional Anesthesia and Pain Medicine 2012; 37(1): 79-98



