Basic Sciences
QUESTIONS:
Before continuing try to answer the following questions. The answers can be found at the end of the article.
- Prophylactic antibiotics
- Should be continued for 5 days post operatively
- Should be used in addition to on-going antibiotic therapy
- Should be administered at least one hour of the beginning of the procedure
- Should target the anticipated organisms.
- Can be used to treat existing infections
- Surgical Site Infections (SSI’s)
- account for 50% of nosocomial infections
- are caused by endogenous flora
- are less common when implanting prosthetic materials
- the incidence can be reduced by giving broad spectrum prophylaxis
- are influenced by the type and duration of surgery.
- Suitable Antibiotics for prophylaxis
- can be used for all types of surgery
- should be used only in high risk patients
- can only be given intravenously
- should be given once at the beginning of the operation only
- will eradicate Methicillin Resistant Staphylococcus Aureus (MRSA)
INTRODUCTION
Antibiotic drugs may be given prophylactically to prevent the development of infection. This use is common in some forms of surgery and anaesthetists are often responsible for the administration of prophylactic antibiotics. Therefore it is important to understand the rationale behind this particular use and how suitable combinations of drugs may be chosen.
SURGICAL SITE INFECTION
Surgical site infection (SSI) accounts for approximately 15% of all hospital acquired, or nosocomial infections in the UK and their occurrence doubles the average length of stay of the patient in hospital. Infection in the wound produces pain and carries the risk of generalised sepsis and multi organ failure. In the UK there is a detailed protocol for the surveillance of SSIs, and hospitals are obliged to report the rates of SSIs for different types of surgery. Clear definition of surgical site infection and active surveillance both in hospital and, with increasingly early discharge, in the community, are set out in the protocol.
ANTIBIOTIC SURGICAL PROHYLAXIS
The principles of good antibiotic prophylaxis include;
- Reducing the incidence of surgical site infection
- Using appropriate antibiotics in a manner supported by evidence
- minimising the effect on the patient’s normal bacterial flora
- minimising adverse effects
- causing a minimal change to the patient’s host defence systems.
Surgical site prophylaxis accounts for one third of all antibiotic prescribing.
Prophylaxis prescribing must be distinguished from empiric therapy. The concept of prophylaxis is that antibiotics given pre-operatively will prevent infection occurring and therefore there is no need to continue the administration of the antibiotic into the post-operative period
Choice of Antibiotic
The choice of antibiotic for surgical site infection prophylaxis will depend on the following factors;
- The anticipated infecting organisms
- Local information regarding drug resistance
- Penicillin sensitivity of the individual
- Narrow spectrum low cost drugs should be the first choice.
Cephalosporins particularly Cefazolin are appropriate first line agents for most surgical procedures, where the patient is not sensitive to cephalosporins and there is no known pattern of resistance to these drugs.
Dose and duration of Antibiotic prophylaxis
Evidence suggests that one single dose of antibiotics should be given for prophylactic purposes. The standard dose used in therapy is used for prophylaxis. The commonest route of administration is intravenous and the drug should be given 30 minutes before the commencement of surgery.
In prolonged surgery, especially cardiac surgery re-dosing at 4 hour intervals is indicated. In the majority of other surgical procedures there is no evidence of increased effect by repeat dosing. The one exception to this rule is in hip arthroplasty, where continuing antibiotic therapy for 24 hours reduces infection rates. In massive blood loss and fluid replacement where losses exceed 1500mls, there is evidence that the serum concentration of antibiotic is at risk of falling below the minimum inhibitory concentration, and repeat dosing should be considered when fluid replacement has been achieved. There is very little evidence that prophylactic antibiotics should be continued beyond 24 hours.
Alternative Routes of Administration
Oral administration of prophylactic antibiotics has significant disadvantages. Effective serum concentrations are achievable with many antibiotics, but rates of absorption are variable from individual to individual. In SSI prophylaxis it is important to have effective drug concentrations when surgery commences, therefore intravenous administration is preferred, but should be carefully timed to 30 minutes before the commencement of surgery. For this reason it falls to the Anaesthetist to administer the drugs.
Other routes are used in specific circumstances; In ophthalmology, during cataract surgery, intra-cameral cephalosporin instillation reduces post-operative infection and in penetrating eye injury instillation intra-viterally or intra-camerally of cephalosporin reduces the incidence of endophthalmitis. In joint replacement surgery the combined use of intravenous antibiotics plus the use of antibiotic impregnated cement reduces both would infection and the incidence of osteomyelitis and infection of the prosthetic joint. In neurosurgery the instillation of antibiotics into ventriculo-peritoneal shunts at the time of insertion reduced infection rates, but there is no good evidence for the insertion of shunts that are themselves impregnated with antibiotics. A similar lack of evidence for the effectiveness of antibiotic impregnated central venous catheters prevents recommendation of their use.
NORMAL BACTERIAL SKIN FLORA
The internal organs of mammals, including humans are normally sterile. The external surfaces, including skin, mucus membranes and bowel wall are colonised with bacteria and fungi from the environment. This includes the lower colon, anterior urethra and vagina.
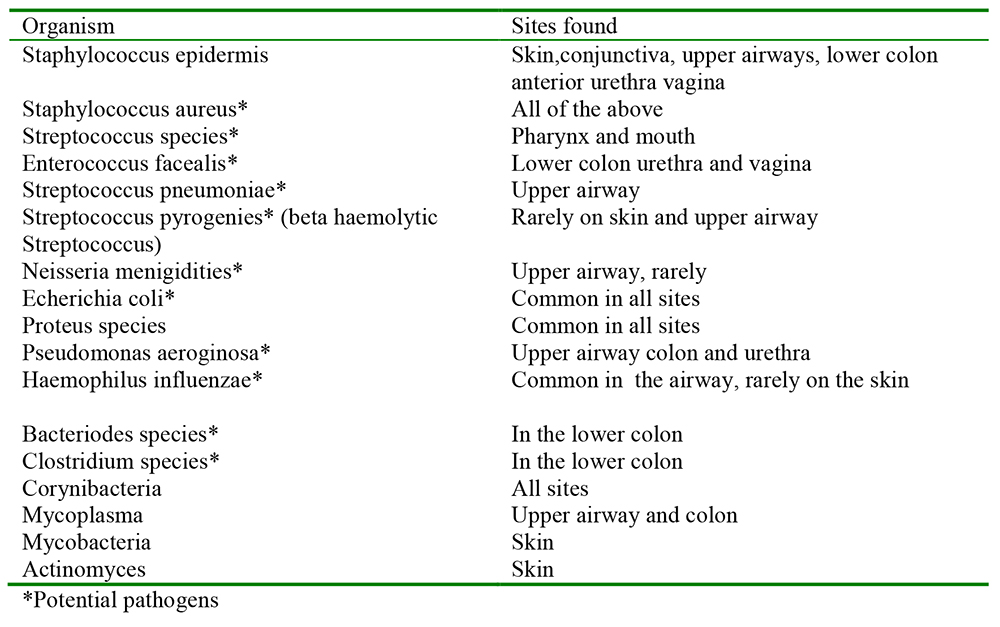
Table 1. The organisms that are commonly found at different anatomical sites
Up to 200 different species have been found on Humans, the colonisation starts at birth, the foetus in-utero is sterile. The exact composition can vary with age, stress, nutritional state and hormonal state. Variations are small and the composition is remarkably stable. Approximately 1012 on the skin, 1010 in the mouth and 1014 in the intestinal tract. This is more than the number of cells in a human!
The association between these bacteria and humans is mostly mutually beneficial; the bacteria derive nutrients and warmth, and in return provide assistance in digestion, synthesise vitamins, stimulate the immune system (particularly in the gut), and provide protection from colonisation by pathogens. The normal flora can however opportunistically become pathogenic to the host human in certain circumstances.
The average human has two square meters of skin. Bacterial colonisation is not uniformly distributed, for example the axilla and the space between the toes are heavily colonised while the rest of the skin is sparsely colonised. The common skin flora are gram positive staphylococcus. They are not normally pathogenic and produce fatty acids that inhibit fungal growth.
Some bacteria produce glycoproteins on their walls that allow them to adhere to specific surfaces. These glycoproteins are called adhesins and explain the specificity of certain species to the upper respiratory tract. Some species produce biofilms, a collection of bacterial cells and glycoproteins on a surface, the common example of this is the formation of dental plaque.
The lower colon flora can be influenced by diet, age and the use of antibiotics. The predominant species is Bacteroidies, which out-number E.Coli by 1000 to 10,000 to 1. The bacterial flora of the lower GI tract synthesis and secrete vitamins, and has a significant role in preventing colonisation with pathogens. Using antibiotics alters this flora and can allow overgrowth of resistant species. Colitis caused by Clostridium Difficille can result from antibiotic use and carries a significant morbidity and mortality. The toxins produced by these organisms cause colitis resulting in diarrhoea and pain. This is the commonest cause of diarrhoea in hospitals. It is associated with the use of broad spectrum antibiotics, and is the major reason for recommending narrow spectrum antibiotics for surgical prophylaxis.
Methicillin Resistant Staphylococcus Aureus (MRSA)
Since the introduction of the antibiotic Methicillin in 1959, increasing numbers of Staphylococcus Aureus (which is part of the normal bacterial skin flora) have been detected which carry a gene encoding resistance to this and other antibiotics in the penicillin family. Many of these isolates are multiply resistant.
Staphylococcus species of bacteria commonly transfer genetic material from one individual to another as part of their natural biology. It is this process that accounts for the rapid development of resistant colonies. Once a colony of resistant Staphylococcus is established on an individual human it is almost impossible to eradicate. In the normal course of events, individuals are not aware of their status of having MRSA on their skin, but when surgery is contemplated it becomes important to identify these individuals for their own safety and also because of the risk of contamination of the hospital itself and the risk of transmission to other patients. Surgical site infections due to MRSA represent a difficult treatment challenge, and are potentially of high morbidity and mortality.
Although eradication is almost impossible to achieve it is possible to suppress the MRSA bacteria for a short time. This greatly reduces the risk to individual and hospital. Patients can be screened for the presence of MRSA by swabbing the skin, especially the axilla, and the nostrils. In the presence of MRSA colonisation, suppression therapy is indicated. Daily washing of the body and the hair with 4 % Chlorhexidine solution, dusting the axillae and feet with 4% chlorhexidine powder, and instilling antibacterial cream into the nostrils, 2% mupirocin is generally used, is effective in lowering the bacterial load of resistant organisms. This regime should be undertaken for at least 3 days prior to surgery. Since 2010 all UK NHS elective surgical admissions are screened for MRSA. In 2011 screening of emergency admissions is planned to start.
ANTIBIOTIC PROPHYLAXIS FOR RESPIRATORY AND URINARY TRACT INFECTIONS
The Cochrane review of antibiotic prophylaxis in long bone fractures has shown a reduced incidence of both chest and urinary tract infections. Other meta analyses have shown that antibiotic prophylaxis with cephalosporins is effective in reducing both wound, respiratory and urinary tract infections in abdominal, gynaecological and general orthopaedic surgery. However it conveys no advantage in cardiac surgery, in closed heart surgery giving antibiotics was associated with more infections with resistant organisms.
ANTIBIOTIC PROPHYLAXIS FOR INFECTIVE ENDOCARDITIS
The UK based National Institute for Clinical Excellence (NICE) has issued guidelines on the use of antibiotics for prevention of infective endocarditis in vulnerable patients. The patients at risk are children or adults with an underlying cardiac defect, such as a replacement valve, congenital abnormality or cardiomyopathy. These also apply to any patient who has previously had endocarditis with or without a cardiac defect.
Procedures which increase the risk of endocarditis have been defined as
- Any Dental Procedures
- Any gynaecological procedure including childbirth
- Any procedure on the urethra or bladder
- Any procedure on the upper airway or gastrointestinal tract.
Likely infecting flora can be predicted from the normal flora table.
In the current guidelines antibiotic prophylaxis is NOT recommended in any of the vulnerable groups for any of these procedures. There is no evidence to support the use of antibiotics in these patients, and use in the past has contributed to increase resistance rates in bacterial populations. In the case of dentistry, it has been shown that normal cleaning of the teeth produces a bacteraemia of higher organism load than any routine dental treatment. These guidelines are also recommended by the American Heart Association.
ANTIBIOTIC IMPREGNATED CENTRAL VENOUS CATHETERS
Infection of central venous catheters has potentially grave consequences. The colonisation of central venous catheters most usually occurs at the tip of the catheter and is most commonly with organisms from the normal skin flora. Organisms are most like to travel down the outer surface of the catheter. There are guidelines for the management of central venous catheters which minimise colonisation of the line which emphasis standard infection control measures including hand washing and the use of 2% chlorhexidine solution for skin preparation. Tunnelling of indwelling central venous catheters has been shown to greatly reduce the rate of colonisation. Catheters have been produced that are impregnated with antiseptic (chlorhexidine) antibiotic (rifampicin) and bacteriostatic (Silver compounds) on either the external or internal surface. Evidence suggests that catheters impregnated with antiseptic and antibiotic on their external surface will reduce the incidence of catheter colonisation. In current US guidelines it is recommended that these catheters should be used in high risk cases, as part of an infection control strategy which ensures that all standard precautions are taken when managing the line. This is not part of the NICE guidelines in the UK. Particular caution is recommended with these lines, the dose of antibiotic/antiseptic that the patient is exposed to is quite small, but because it is introduced into a central vein the risk of immediate anaphylactic shock in allergic patients is very high.
ANSWERS TO QUESTIONS
- FFFTF
- FTFFT
- FFFTF
WEBLINK
REFERENCES and FURTHER READING
- Woodfield JC et al. A meta-analysis of randomised controlled trials assessing the prophylactic use of Cefriaxone Asssesing wound, chest and urinary tract infection. World Journal of Surgery 2009 Dec 33(12)2538-50



