Regional Anaesthesia
KEY POINTS
- The aetiology of peripheral nerve injury (PNI) secondary to nerve blocks can be from mechanical, direct toxicity, ischaemic, or inflammatory causes.
- A variety of methods have been proposed to reduce the risk of block-related nerve injury, including needle choice, use of peripheral nerve stimulation, monitoring injection pressure, ultrasound guidance, or a combination of these strategies.
- Ultrasound guidance is unique because it provides real-time trajectory of the needle path and can detect intraneural needle placement, potentially minimising the risk of nerve injury.
- Effective management of PNI includes excellent communication with the patient and other health care professionals and, if necessary, early referral to a specialist with expertise in peripheral neuropathies.
INTRODUCTION
Peripheral nerve blockade (PNB) is associated with a number of potential complications, including inadvertent vascular puncture and associated haematoma, inadvertent damage to other structures (eg, pleura), local anaesthetic systemic toxicity, myotoxicity, infection, misplacement or migration of catheters, wrong-site block, and nerve injury. Although nerve injuries associated with PNB are often temporary, the potential long-term consequences of nerve injury following PNB include sensory and motor deficits, neuropathic pain, and permanent disability, with an associated decrease in patient quality of life. The estimated incidence of serious long-term PNB-related nerve injury is 2 to 4/10 000 blocks.1 This incidence is difficult to quantify because of the lack of comprehensive epidemiological data, potential underreporting, and difficulties in attributing causation. The incidence may also vary according to the type of block performed.2 This article aims to explore the pathophysiology of nerve injury from PNB, suggest preventative strategies therein, and provide a commonsense approach to their management.
ANATOMY
Peripheral nerves consist of axons bundled together by 3 separate connective tissue layers (see Figures 1a, b). Most axons are myelinated, with a Schwann cell that covers the axon in myelin and gaps known as the nodes of Ranvier spaced out at intervals to enhance nerve impulse conduction. The axon and Schwann cell structure responsible for myelination is termed the nerve fibre and is the functional unit of the peripheral nerve complex. This is covered along the length of the fibre by the endoneurium, the innermost connective tissue layer that contributes to the specialised environment that protects the axons.
Nerve fibres are then grouped together into fascicles surrounded by the perineurium, which acts as the principle protective layer of the axon. Fascicles are bundled into peripheral nerves, surrounded by an outer layer called the epineurium.
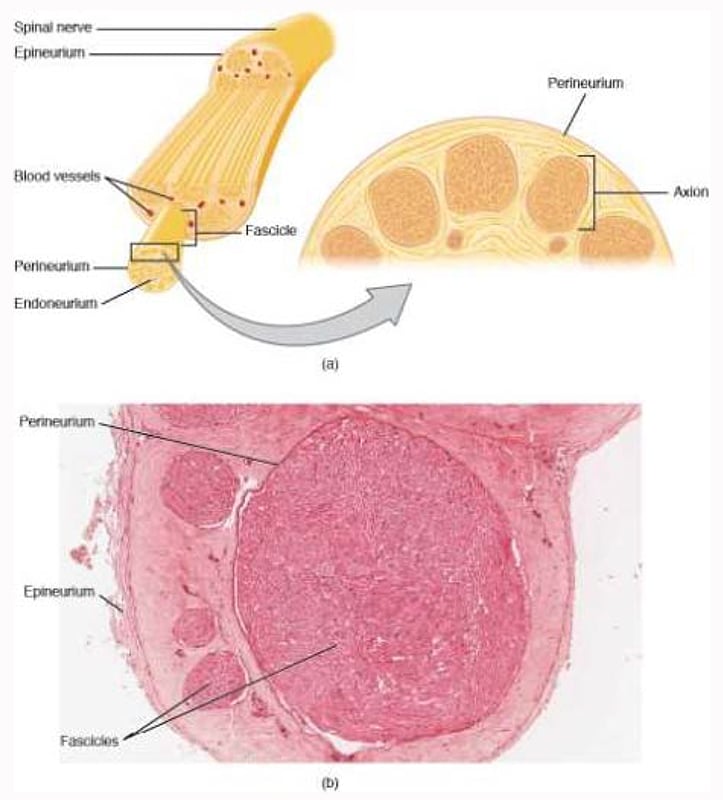
Figure 1. (a, b) Nerve structure. The structure of the nerve is organized by the layers of connective tissue on the outside, around each fascicle, and surrounding the individual nerve fibres (tissue source: simian). LM x 40. (Micrograph provided by the Regents of University of Michigan Medical School 2012.) Source: Anatomy and Physiology OpenStax textbook, distributed under Creative Commons Attribution 4.0 International License. No changes to picture or accompanying text were made.
Whilst the perineurium is a tough squamous epithelial sheath with collagen fibres and fibroblasts acting as an impermeable barrier to potential toxins (including local anaesthetics), the epineurium is a permeable layer consisting of collagen fibres and blood vessels, which gives nerves their characteristic macroscopic and sonographic appearance. Peripheral nerves receive their blood supply via a capillary network within the endoneurium, known as the vasa nervorum, and via arterioles, veins, and capillaries in the epineurium.
Moving distally, a nerve’s physical properties and resilience to mechanical injury change. Nerve roots have reduced tensile strength and elasticity compared with peripheral nerves. Spinal nerve roots are at increased risk of mechanical and chemical injury because of an absence of the protective layer of both the perineurium and intraneural connective tissue. More distally, the course of the peripheral nerve complex undulates, providing slight excess length. This, combined with a relatively loose attachment to surrounding structures and the use of connective tissue acting as a filler between specialised structures (eg, nerves or muscles), affords the distal nerve some toleration of stretch, movement, and elasticity.3
CLASSIFICATION OF NERVE INJURY
Nerve injury can be classified according to the degree of dysfunction using either Seddon’s or Sunderland’s classification (see the Table). Seddon’s classification is more commonly used, with 3 levels of injury described. Most nerve injuries are mixed, with different fascicles showing different injury characteristics.
PATHOPHYSIOLOGY OF NERVE INJURY
The pathophysiology of nerve injury through PNB is not always clear, although mechanical injury (either through direct trauma of the needle or pressure effects), toxicity of injectate, inflammatory injury, and ischaemic injury can be responsible.4 There may

Table. Nerve Injury Classification
also be non–PNB-related factors including surgical manipulation or direct mechanical injury, tourniquet ischaemia, preexisting neuropathies, patient positioning for surgical requirements (including of insensate limbs), or postoperative casts. Finally, unintended blockade of other nerves can occur, for example, sympathetic, recurrent laryngeal, and phrenic nerve blockade in association with interscalene blocks (see the Phrenic Nerve Information box).
Mechanical Injury
Mechanical injury may occur via direct needle trauma or peripheral nerve catheter placement. Disrupting the perineurium can lead to direct axonal injury or fascicular content leakage and loss of the protective environment, which can subsequently expose axons to higher concentrations of local anaesthetic (with associated toxicity). There can also be some localised cellular and inflammatory changes. Another deleterious effect related to the lack of perineurial compliance is increased pressure on the injection, which leads to disruption of blood supply and ischaemia.
Mechanical injury risk also differs according to the site of injection due to differing neural to connective tissue ratios. Having relatively more connective tissue decreases the possibility of a needle entering a fascicle itself. Even if a needle penetrates a nerve, the fascicles are more likely to be pushed out of the way, decreasing the risk of intraneural injection, whilst having more neural tissue and less connective tissue increases the risk of fascicular injury (see Figure 2). By way of example, the truncal brachial plexus has a higher neural tissue ratio and a higher rate of postoperative neuropathy following interscalene PNB than might be expected.5,6 Generally, we can state that the ratio of nonneural connective tissue to neural tissue increases from proximal to distal, with a concomitant decrease in the risk of peripheral nerve injury (PNI).
Toxicity
All local anaesthetics used in clinically relevant concentrations can be potentially neurotoxic. A number of pathophysiological processes have been postulated, from vasoconstriction to mitochondrial disruption, although the effect is dose dependent. The site of local anaesthetic administration is the most important factor for toxicity, with intrafascicular injection leading to more injury than intra- or extraneural injections. The addition of adjuncts may also play a role in neurotoxicity (eg, adrenaline may disrupt neural blood flow).
Inflammatory Injury
Inflammatory injury is being increasingly recognised as playing a role in some PNB-related neuropathy; for example, nonspecific inflammatory responses have been suggested as the mechanism of chronic diaphragmatic paralysis following
BOX 1. PHRENIC NERVE INFORMATION
Phrenic nerve blockade and hemidiaphragm paralysis are relatively common complications of interscalene brachial plexus blocks (ISBs). During ISBs, there can be a spread of local anaesthetic under the prevertebral fascia anterior to the anterior scalene muscle or cranial spread of local anaesthetic to the ventral root of C4. The risks of phrenic nerve involvement increase as volume increases (with 100% blockade reported with 30- to 40-mL volumes). Dose reduction lowers this risk, as does using ultrasound, and injecting lower (having identified C7). Finally, performing alternative brachial plexus blocks (such as supraclavicular or axillary) negates the risk of phrenic nerve blockade.
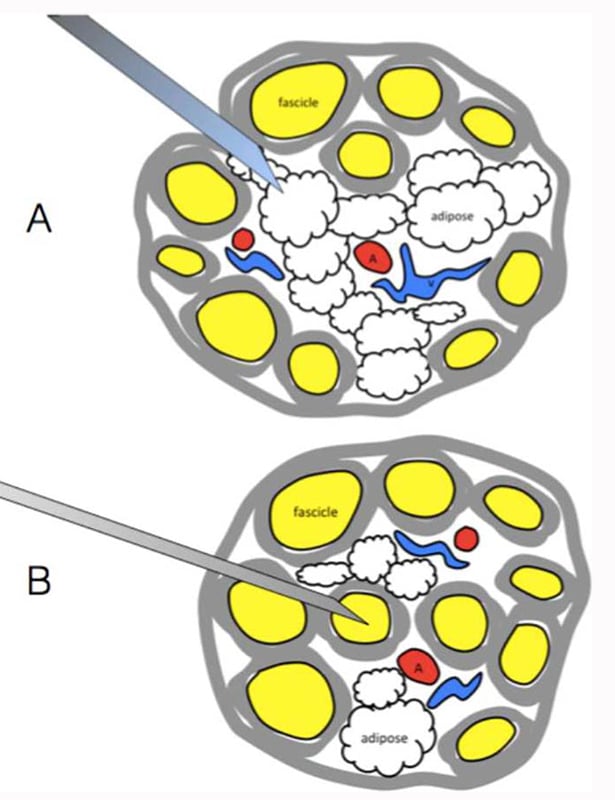
Figure 2. Relative differences between neural and connective tissue. Note in (A) the relative abundance of connective tissue, so that the needle is more likely to miss the neural tissue and push the neural tissue aside. However, in (B), there is a higher neural to connective tissue ratio, so fascicular penetration is more likely, with associated PNI. Source: Courtesy of M. Barrington.
interscalene block, resulting in adhesions, fascial thickening, vascular changes, and scar tissue compressing the nerve.7 Intraneural injection of ultrasound gel has also been implicated in inflammatory changes.8
Ischaemic Injury
Local or diffuse ischaemia occurs either from direct occlusion by acute haemorrhage or via direct vascular injury, both of which can disrupt neural vascular supply. About 50% of a peripheral nerve’s vascular supply comes via the network of capillaries known as the vasa nevorum. This means that disruption should localise (and minimise) damage, with the remaining collateral blood supply aiding perfusion, as no single vessel dominates perfusion. As mentioned previously, local anaesthetics and their adjuncts may affect vascular supply, as can trauma, high injection pressure, tourniquets, or extra- or intraneural haematomas.
PNI RISK MINIMISATION
A number of strategies can be employed to reduce the potential risk to your patient. These can range from basic to more advanced techniques and can be used in combination (eg, using peripheral nerve stimulation or injection pressure monitors with ultrasound).
Needle Choice
A short bevelled needle is less likely to penetrate the perineurium compared with a long bevelled one, probably because the fascicles slide away from the tip, resulting in reduced likelihood of damage. However, should fascicular damage occur, the lesions secondary to short bevelled needles are more severe.9 Increased needle diameter is also associated with increased severity of nerve injury,10 as is increased force of advancement.
Paraesthesia
The evidence for linking paraesthesia and subsequent PNI is unclear.11 As such, not eliciting paraesthesia on injection does not reliably exclude PNI. However, severe paraesthesia or pain on needle advancement or injection may imply intraneural placement. So as to preclude any potential further injury, the clinician should stop and either reposition the needle and/or stop injecting. Factors such as sedation, comorbidities (eg, peripheral neuropathy), the patient’s ability to communicate or perceive pain, and sensory blockade from local anaesthetic already present can affect evaluation of paraesthesia.
Peripheral Nerve Stimulation
Studies have shown that the minimal thresholds of current needed to elicit a motor response are different depending on whether the needle is extra- or intraneurally placed. A clinical study showed that there was no motor response at ≤0.2 mA unless the needle was intraneural.12 This supports animal evidence showing that similar current thresholds suggest intraneural tip placement. Peripheral nerve stimulation is thought to be a specific indication of intraneural placement (so that a motor response elicited at ≤0.2 mA can be obtained only from intraneural placement) but not sensitive (so that high current intensity needed may still be required to elicit a motor response even if it is intraneural). This is likely to be because the motor response is related to needle axon distance. As a rule of thumb, eliciting no motor response at ≤0.5 mA prior to injection of local anaesthetic is likely to be safe.
Injection Pressure Monitoring
Intrafascicular needle tip placement can result in high opening injection pressures and is associated with nerve injury. In contrast, extrafascicular needle placement is associated with lower pressures. Needle tip placement against a fascial plane or the epineural border of a nerve may also result in high injection pressures.13 Most research in this area is with animal models, but a study in human cadavers showed high pressures (>30 psi) followed injection directly into brachial plexus roots (with 1 of 30 cases leading to the spread of injectate into the epidural space).14 Research suggests a good sensitivity but low specificity for pressure monitoring, but currently accurate assessment of manual injection pressures is limited. Novel technologies are being developed and tested in small-scale studies, but evidence is limited so far. However, avoidance of high injection pressures (postulated at ≥15 psi) is likely to improve safety and be clinically justifiable.
Ultrasound
Ultrasound guidance has become the preeminent method of performing PNBs because needle visualisation is believed to offer a greater margin of safety. However, the incidence of PNI has not changed as ultrasound use has increased. This may be due to operator- and patient-dependent differences in obtaining good-quality images, the fact that ultrasound resolution at present cannot differentiate between intra- and extrafascicular needle tip placement, and that by the time a fascicle expands with injectate, it may be too late to prevent PNI. There are, however, a number of techniques available to the ultrasound operator to minimise risk.
Avoiding Intraneural Injection
Aiming for the connective tissue that surrounds the nerve, and so being extraneural, should provide safe and effective regional anaesthesia, which in practical terms means aiming at the side (not directly at) of the nerve. Although the author of one study suggested that intraneural injection may not necessarily result in neural injury,15 the study was limited by a small sample size, selection bias, and loss of patient follow-up. A second study documented a lack of difference in electrophysiological injury between intraneural and extraneural injection. However, their findings suffered from generalizability related to the recruitment of only healthy subjects and the expertise of the proceduralists halting injection if paraesthesia or pain was detected, maintaining an injection pressure less than 15 psi. We recommend aiming the needle extraneurally, outside of the outer border (epineurium) of the nerve, but still under the connective tissue, allowing for a good spread of injectate (see Figure 3). On ultrasound, a low- pressure injection into the area between the epineurium and connective tissue should result in hydrodissection (ie, expansion of this area via hydrostatic pressure with the dark signature of the injectate). Note that whilst intraneural injection can be detected peri-injection as nerve expansion, this does not always happen, especially with small volumes.
Planning a Trajectory to Avoid Damage to Structures Including Nerves
Some structures do not appear on ultrasound as clearly as others, thus increasing the risk of accidental damage, so caution must be taken when choosing a potential needle trajectory. For example, when performing a popliteal sciatic block, the common peroneal nerve may not be as visible as the tibial nerve and may also have an oblique course. As such, care must be taken when choosing a trajectory to avoid damage to the common peroneal nerve when attempting to block the tibial nerve (see Figure 4).
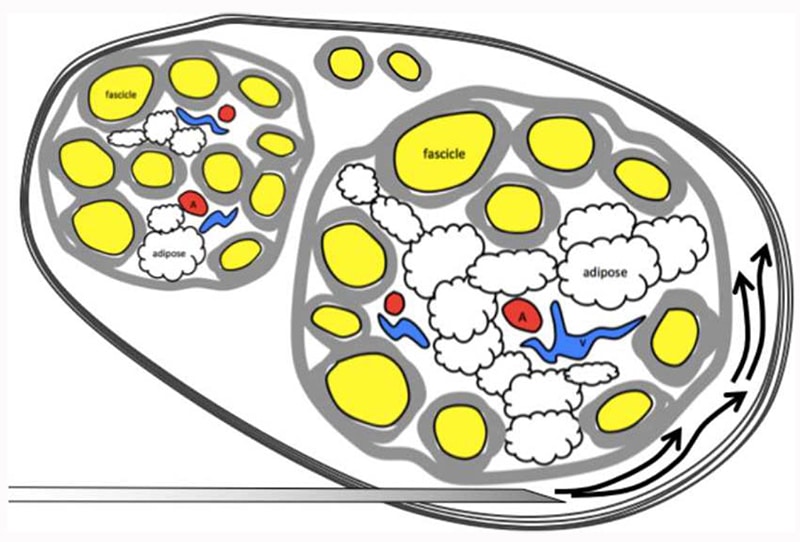
Figure 3. Aiming extraneurally with the needle tip for a popliteal sciatic nerve block, with good spread of local anaesthetic between the epineurium and connective tissue. On ultrasound, a low-pressure injection into the area between the epineurium and connective tissue should result in hydrodissection. Source: Courtesy of M. Barrington.
Using hydrodissection to open a needle trajectory route may displace structures so that they are out of the way, providing a simpler route to the intended target (see Figure 5). In addition, reducing the number of passes required to achieve spread also reduces the likelihood of injury; often going on one side of the nerve is sufficient to achieve the required effect, for example, if you are attempting to achieve analgesia postoperatively rather than perioperative surgical anaesthesia (and so trying to surround the nerve in local anaesthetic) or if a nerve is small (eg, the musculocutaneous nerve), so that the injectate diffuses quickly to the whole nerve. Often by injecting on one side of the nerve, the local anaesthetic spreads successfully to surround the nerve, although you may need to scan up and down to see this.
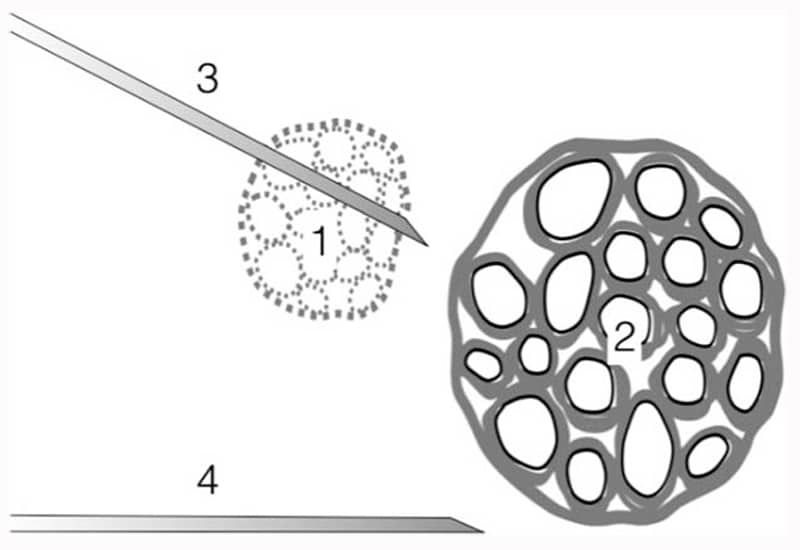
Figure 4. Planning a needle trajectory. Care must be taken to look for nerves that may not be easy to see under ultrasound. For example, when performing a popiteal sciatic block, the common peroneal nerve (1) may not be as visible as the tibial nerve (2) and may also have an oblique course. As such, care must be taken when choosing a trajectory to avoid damage to the common peroneal nerve when attempting to block the tibial nerve (prudent to choose path 4 over 3). Source: Courtesy of M. Barrington.
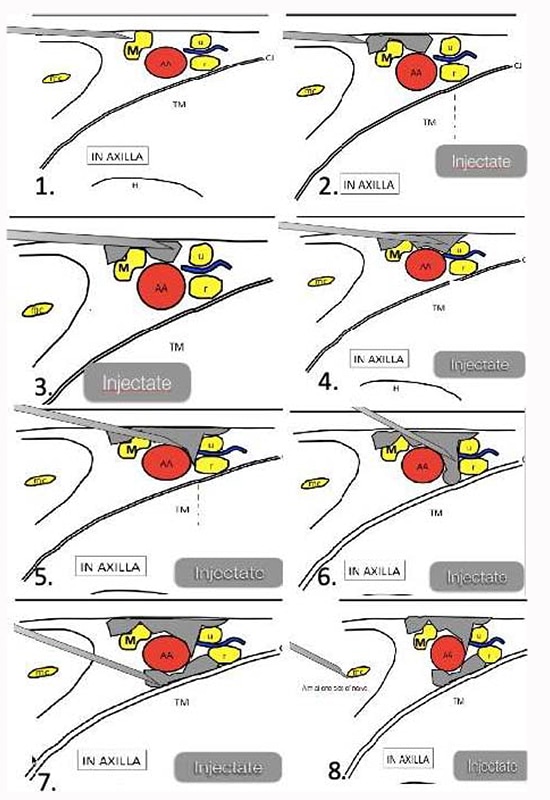
Figure 5. An in-plane axillary brachial plexus block us illustrated, providing an example of how the injectate can hydrodissect the tissue close to a nerve and create space for the needle to advance without causing trauma to the nerve. M, median nerve; u, ulnar nerve; r, radial nerve; mc, musculocutaneous nerve; AA, axillary artery; CJ, conjoined tendon; TM, teres major muscle. Source: Courtesy of M. Barrington. (1) Halt needle advancement at this point and begin hydrodissection. (2) Hydrodissection can create space for needle advancement and improve the visibility of the needle and nerve. Maintain constant pressure on the transducer so that the vein remains collapsed. (3) Median nerve displaced and room provided for needle advancement. (4-6) Inject and advance. (7) Alternate approach to radial nerve. (8) Blocking the musculocutaneous nerve.
Management of Peripheral Nerve Injury
Even if the steps outlined above are enacted, PNIs can still occur, although fortunately, this is a very infrequent or rare event. Any acute motor deficit requires rapid assessment and management, including appropriate imaging (see Figure 6). It is important to rule out or diagnose reversible pathology, including compartment syndrome, surgical nerve transection, or external compression (eg, from tight casts, haematoma), requiring timely management. Even where PNI is suspected, establishing a diagnosis can be difficult secondary to lingering anaesthesia (both general and local), functional or pain-related limitations to examination, external dressings/casts and so forth, or a misunderstanding of the duration of block (from both patients and fellow health care professionals). Once diagnosed, good communication with the patient is key, with clear annotation of events leading to PNI and what management steps and communication with the patient has occurred. In particular, having a thoroughly documented neurological examination is important, as this can act as a baseline against which any future progress can be compared. Comprehensive documentation is important both from an interdisciplinary communication perspective and for any medicolegal consequences that may follow.
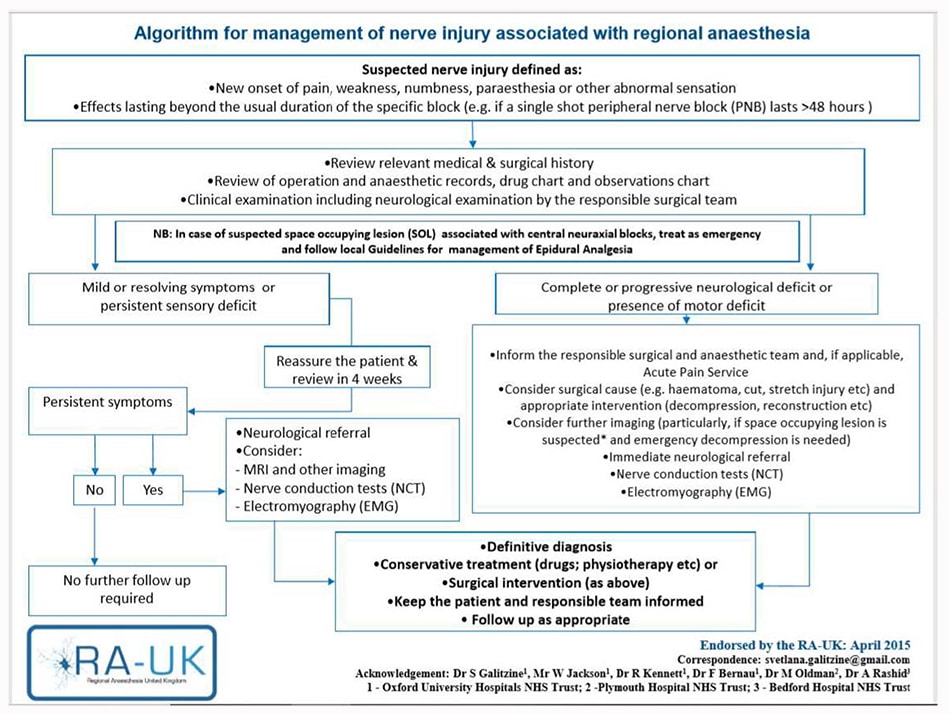
Figure 6. RA-UK algorithm for management of nerve injury associated with regional anaesthesia. Reprinted with kind permission from Dr. Svetlana Galitzine.
Initial communication should include reassuring the patient and noting that most PNIs will self-resolve; about 95% of postoperative sensory changes will resolve within 4 to 6 weeks (with the vast majority healing within the first week) and 99% in the first year. However, motor deficits and evolving, severe, or complete neuropathies are a sign of a more serious PNI, and referral to a neurologist is urgently warranted. Investigations recommended may include electromyography or nerve conduction studies. In all cases, the patients’ GP should be contacted, with follow-up to ensure that the PNI is resolving. It is also good practice to have a safety net, that is, ask the patient either to contact the anaesthetic team or to re-present to a health care professional if symptoms do not resolve or worsen.
REFERENCES
- Neal JM, Barrington MJ, Brull R, et al. The second ASRA practice advisory on neurologic complications associated with regional anesthesia and pain medicine: executive summary 2015. Reg Anesth Pain Med. 2015;40:401-30.
- Brull R, McCartney CJL, Chan VWS, et al. Neurological complications after regional anaesthesia: contemporary estimates of risk. Anesth Analg. 2007;104:965-974.
- Barrington MJ, Brull R, Reina M, et al. Complications and prevention of neurological injury with peripheral nerve blocks. NYSORA. https://www.nysora.com/foundations-of-regional-anesthesia/complications/complications-prevention-neurolog ic-injury-peripheral-nerve-blocks/. Accessed March 30, 2019.
- Brull R, Hadzic A, Reina MA, et al. Pathophysiology and etiology of nerve injury following peripheral nerve blockade. Reg Anesth Pain Med. 2015;40:479-490.
- Moayeri N, Bigeleisin PE, Groen GJ. Quantitative architecture of the brachial plexus and surrounding compartments, and their possible significance for plexus blocks. Anesthesiology. 2008;109:299-304.
- Moayeri N, Groen GJ. Differences in quantitative architecture of sciatic nerve may explain differences in potential vulnerability to nerve injury, onset time, and minimum effective anesthetic volume. Anesthiology. 2009;111:1128-1135.
- Kaufman MR, Elkwood AI, Rose MI, et al. Surgical treatment of permanent diaphragm paralysis after interscalene nerve block for shoulder surgery. Anesthesiology. 2013;119:484-487.
- Pintaric TS, Cvetko E, Strbenc M, et al. Intraneural and perineural inflammatory changes in piglets after injection of ultrasound gel, endotoxin, 0.9% NaCl, or needle insertion without injection. Anesth Analg. 2014;118:869-873.
- Selander D, Dhuner KG, Lundberg G. Peripheral nerve injury due to injection needles used for regional anaesthesia: an experimental study of the acute effects of needle point trauma. Acta Anaesthesiol Scand. 1977;21:182-188.
- Steinfeldt T, Nimphius W, Werner T, et al. Nerve injury by needle nerve perforation in regional anaesthesia, does size matter? B J Anaesth. 2010;104:245-253.
- Fanelli G, Casati A, Garancini P, et al. Nerve stimulator and multiple injection technique for upper and lower limb blockade: failure rate, patient acceptance and neurologic complications. Study Group on Regional Anaesthesia. Anesth Analg. 1999;88:847-852.
- Biegeleisen PE, Moayeri N, Groen GJ. Extraneural versus intraneural stimulation thresholds during ultrasound guided interscalene block. Anesthesiology. 2009;110:1235-1243.
- Gadsen J, Latmore M, Levine DM, et al. High opening pressure is associated with needle-nerve and needle-fascia contact during femoral nerve block. Reg Anesth Pain Med. 2016;41:50-55.
- Orebaugh SL, Mukalel JJ, Krediet AC, et al. Brachial plexus root injection in a human cadaver model: injectate distribution and effects on the neuraxis. Reg Anesth Pain Med. 2012;37:525-529.
- Biegeleisen PE. Nerve puncture and apparent intraneural during ultrasound-guided axillary block does not invariably result in neurologic injury. Anesthesiology. 2006;105:779-783.



