Basic Sciences
Self assessment
- How do you assess a patient’s nutritional status?
- How would you calculate the energy requirements for a 33 year old man (height 1.80m and weight 75kg) admitted post-laparotomy for a ruptured appendix and sepsis (temperature 39°C)?
- How would you calculate the energy requirements for a 33 year old man (height 1.80m and weight 75kg) admitted post-laparotomy for a ruptured appendix and sepsis (temperature 39°C)?
- What are the advantages of enteral nutrition over parenteral nutrition?
- How can you reduce the risk of aspiration pneumonia in a patient who is enterally fed?
- Is glucose control important? If so, what should the target be?
Introduction
Malnutrition (sometimes called undernutrition) is the state in which a deficiency either of total energy or of protein (or other nutrients) leads to a reduction in body cell mass and organ dysfunction. It can be the result of any combination of inadequate intake, reduced absorption or increased requirements.
Malnutrition is common in hospital patients throughout the world and tends to be an under recognised and under treated problem. It has been estimated that up to 60% of hospital patients in the UK are either malnourished or are at risk of becoming malnourished and figures for developing countries are likely to be even higher. Malnutrition has been shown to be strongly linked to increased length of stay in hospital, increased incidence of complications and increased mortality.
The aim of nutritional support is to prevent and treat both macro- and micronutrient deficiencies. The patients likely to benefit most are those who are already malnourished and who would otherwise undergo a long period of starvation. There is now evidence that nutritional support can reduce length of stay and the incidence of complications in patents in intensive care.
Assessment of nutritional status
Nutritional status in critically ill patients can be difficult to assess. Anthropometric measurements (eg. skin fold thickness and mid-arm circumference) are commonly used to assess populations but are not particularly useful in individuals. Biochemical tests also have their limitations: albumin levels fall rapidly as part of the acute phase response and haemoglobin is affected by haemorrhage, haemolysis, transfusion and bone marrow suppression. Transferrin, prealbumin and lymphocyte counts can be useful however they are dependent on the patient being well hydrated.
Body Mass Index (mass [kg] / height [m] 2) is a frequently used tool (with a BMI < 18.5 classed as underweight) and has been shown to be an independent predictor of mortality in seriously ill patients. Nevertheless it does not reflect the acute changes in nutritional status important in critical illness and is used most for the assessment of long term health risks of obesity.
Probably the most useful measure of nutritional status is a targeted history and examination. One such method which is widely accepted is known as the Subjective Global Assessment.
Subjective Global Assessment
This is a structured approach to taking a history that includes the following categories:
- Weight change – both chronic (over 6 months) and acute (over 2 weeks )
- Changes in food intake
- Gastrointestinal symptoms – nausea, vomiting, diarrhoea and anorexia
- Functional impairment
and is combined with a physical examination looking for evidence of the following:
- Loss of subcutaneous fat – looking especially at the chest and triceps
- Muscle wasting – looking especially at temporal region, deltoids & gluteals
- Oedema
- Ascites
As the name implies it is a subjective tool, however it has been shown to be reproducible and also to correlate with mortality in a variety of conditions. Assessment of nutritional status should be used to identify patients at risk of refeeding syndrome.
Nutritional requirements
To actually measure energy use requires sophisticated equipment so requirements are more often estimated using formulae. One such formula is the Harris Benedict Equation which estimates basal metabolic rate (BMR) in kcal/day.
Harris Benedict Equation:
- For _: BMR = 13.75 x weight (kg) + 5 x height (cm) – 6.78 x age (years) + 66
- For _: BMR = 9.56 x weight (kg) + 1.85 x height (cms) – 4.68 x age (years) + 655
This will usually give a result of around 25 kcal/kg/day. The equation estimates BMR in afebrile healthy individuals and will therefore need to be modified according to the situation to calculate resting energy expenditure (REE).
- Fever increase by 10% for each 1°C above 37 (up to max of 40°C)
- Sepsis increase by 9% regardless of temperature
- Surgery increase by 6% if patient has had surgery or trauma
- Burns increase by 100% if any size over 30% (or use Toronto formula)
A careful balance of macronutrients (protein, lipid and carbohydrate) provides the energy requirements whilst micronutrients (vitamins and minerals) are required in very small amounts to maintain health but not to provide energy.
- Protein around 1.5 – g/kg/day (range 1.2 to 2.0 g/kg/day for ICU patients) Use 2g/kg/day if severely catabolic eg. severe sepsis/burns/trauma Provides 5.3 kcal/g
- Lipid – Provides 9.3 kcal/g Calories from lipid should be limited to 40% of total calories
- Carbohydrate – Provides 3.75 kcal/g in vivo Give the remaining the energy requirements as carbohydrate
The proportion of a feed made up by protein is sometimes expressed as a calorie: nitrogen ratio. 6.25g of protein contains 1g of nitrogen. The ratio is then calories (kcal) ÷ nitrogen (g). Recommended calorie: nitrogen ratios are around 100:1 which will be achieved using the above figures. The optimal ratio for lipid: carbohydrate is not known.
Micronutrients Vitamins are organic compounds that usually act as cofactors for enzymes involved in metabolic pathways. Trace elements are ions that act as cofactors for enzymes or as structurally integral parts of enzymes and are often involved in electron transfer. These will be discussed further with parenteral nutrition.
Route of nutrition
Nutritional support can be given through one of three routes:
- Oral
- Enteral – via a tube directly into gastrointestinal tract
- Parenteral – intravenous (via either peripheral or central vein)
If at all possible oral or enteral are the preferred routes for nutritional support. They are far cheaper, more physiological, reduce the risk of peptic ulceration, minimise mucosal atrophy (food in the gut lumen is a potent stimulus for mucosal cell growth) and may reduce translocation of bacteria from the intestinal lumen. Situations previously thought to preclude enteral nutrition, including major gastrointestinal surgery or acute pancreatitis, have now been shown to be best treated with enteral nutrition.
1. Oral
If the patient can eat then they should be encouraged to do so, ideally by giving them food they enjoy and helping them if necessary. It is important to know how much the patient is eating to see whether they are receiving adequate nutrition. If not they will need supplementation either orally or enterally.
2. Enteral
The various methods of enteral feeding are discussed below:
i. Nasogastric – this is the most common method of feeding in Intensive Care. Potential problems include malposition, difficulty swallowing or coughing, discomfort, sinusitis and nasal tissue erosion.
Insertion can be difficult in intubated patients as the tube can catch on the piriform sinuses or the arytenoid cartilages. This can be minimised by either ipsilateral lateral neck compression or by turning the head (if it is safe to do so) 90° to the side towards the nostril being used for insertion. The insertion of a nasal tube is contra-indicated in a patient with a base of skull fracture due to the risk of intracranial penetration.
ii. Oral tubes – not suitable for awake patients however should be considered in intubated patients to reduce sinusitis (a risk factor for ventilator-associated pneumonia).
iii. Enterostomy – gastrostomy or jejunostomy and can be placed at the time of surgery or as a separate procedure. Once inserted they are well tolerated, however there are risks associated with insertion, displacement and infection (including peritonitis). Evidence from non-ICU patients suggests that there are significant benefits for those who require nutritional support for over 4 weeks.
iv. Post-pyloric feeding – nasojejunal or jejunostomy. A nasojejunal tube should be over 120cm long to ensure correct placement. Feeding directly into small bowel avoids the problem of gastroparesis. Small bowel ileus is much less common than gastric ileus and tends to be less prolonged; the small bowel recovers normal function 4-8 hours post laparotomy. Post-pyloric feeding is recommended for patients at high risk of aspiration, those undergoing major intra-abdominal surgery and patients who are intolerant of gastric feeding.
2. Parenteral
Parenteral nutrition is most often given via a central vein as the solutions are usually hypertonic. Preparations for peripheral use are available however they have to be isotonic which means that very large volumes would have to be given to provide adequate nutritional support. This severely limits their usefulness.
Enteral Nutrition
There is much discussion in the literature but these are some simple guidelines:
- Confirm tube position Tube position must be confirmed both clinically and radiographically if possible. Otherwise it is easy to administer feed directly into the lungs.
- Secure tube well There should always be a high index of suspicion that the tube may have become dislodged.
- Sit patient up The patient should be sat up to an angle of at least 30o to minimise the risk of reflux and subsequent aspiration of gastric contents. This can still occur around a cuffed tracheal tube.
- Start feeding early Early feeding (within 24 hours of admission or surgery) has been shown to reduce septic complications, length of hospital stay and readmission rates in patients after both upper and lower gastrointestinal surgery. Enteral feeding does not appear to increase, and may in fact decrease, the incidence of anastomotic breakdown.
- Aspirate regularly (eg. 4 hourly) to ensure that gastric residual volume is less than 200mls. Volumes above this greatly increase the risk of reflux and subsequent pulmonary aspiration and feeding rates should be reduced accordingly. This procedure can be via the feeding tube itself if the distal lumen is in the stomach and if it is wide enough. Otherwise (eg. if a fine bore tube or if jejunal feeding is being used) a separate gastric tube should be used. Once feeding is established this can stop.
- Avoid bolus feeds Large volumes of feed in the stomach will increase the risk of aspiration of gastric contents so bolus feeding should be avoided if possible.
- Use pro-kinetics If the patient is not tolerating enteral feed (ie large volumes of feed are being aspirated from the gastric tube) then pro-kinetic agents should be given such as metoclopramide 10mg iv tds +/- erythromycin usually 250mg iv tds ( recent research suggests this can be reduced to erythromycin 70mg iv tds.)
Which enteral feed to use?
There are many commercially prepared feeds available and some hospitals prepare their own either using commercially prepared dried feed or by following a recipe with normal foodstuffs as ingredients and then blending them to a consistency that will pass through a feeding tube.
Hospital-prepared feeds Recipes vary according to country and available ingredients but can include hard-boiled eggs, milk powder, soya, maize oil, rice, squashes, flour, sugar and fruit. These hospital-prepared feeds are much cheaper than commercially prepared feeds but can block tubes and some recipes have been shown to give unpredictable levels of both macro- and micronutrients. In addition, they may contain contaminated ingredients and are not sterile. As a result, they must not be used for postpyloric feeding or in patients with achlorhydria. These feeds should only be used where commercial feeds are either not available or not affordable.
Polymeric preparations These contain intact proteins, fats and carbohydrates, which require digestion prior to absorption, in addition to electrolytes, trace elements, vitamins and fibre. Fibre is broken down by colonic bacteria to produce a variety of compounds including butyric acid, an energy substrate for colonic enterocytes. These feeds tend to be lactose-free as lactose intolerance is common in unwell patients. The different preparations vary in their osmolality, calorie: nitrogen ration and carbohydrate: lipid ratios and can provide between 0.5 and 2 kcal/ml although most are around 1 kcal/ml. Commonly used ingredients include the protein casein (from milk), soy protein, maize and soya oils and the carbohydrate maltodextrin. The vast majority of patients can be given standard polymeric feeds.
Elemental preparations These preparations contain the macronutrients in a readily absorbable form (i.e. proteins as peptides or amino acids, lipids as medium chain triglycerides and carbohydrates as mono- and disaccharides). They are expensive and only really indicated for patients with severe malabsorption or pancreatic insufficiency.
Disease-specific formulae Are usually polymeric and include feeds designed for:
- Liver disease – low sodium and altered amino acid content (to reduce encephalopathy)
- Renal disease – low phosphate and potassium, 2kcal/ml (to reduce fluid intake)
- Respiratory disease – high fat content reduces CO2 production.
Again they are expensive and there is not enough evidence to justify widespread use.
Specific additives There is a lot of interest in the effects of various additives to nutritional support including omega-3-fatty acids and glutamine. Glutamine is thought to promote anabolism and may be an important intestinal growth factor however the benefits are still unclear. In general these feeds tend to be more expensive and there is no consensus as to their role in critically ill patients.
Parenteral Nutrition
The only absolute indication for parenteral nutrition (PN) is gastrointestinal failure. All efforts to improve tolerance of enteral feeding such as the use of pro-kinetic agents and/or a post-pyloric feeding tube, should be tried before starting PN. PN can be used to supplement enteral nutrition (for example in short gut syndrome) or can be used as the sole source of nutrition: total parenteral nutrition. How long a patient needs to be without nutrition before they should be given PN is unclear and will depend on their pre-existing nutritional status and the disease process. There is evidence that PN is better than no nutritional support after 14 days and many units will start PN in patients expected not to tolerate adequate enteral nutrition for 7 days. Patients receiving less than 25% of their predicted needs are at increased risk of sepsis and those who are not tolerating enteral nutrition despite all attempts to improve this should be considered for parenteral supplementation.
PN can be given as separate components but is more commonly given as a sterile emulsion of water, protein, lipid, carbohydrate, electrolytes, vitamins and trace elements according to the recommendations discussed earlier regarding nutritional requirements.
Protein – given as amino acids and needs to include essential amino acids – those which cannot be synthesized from other amino acids by transamination (histidine, leucine, isoleucine, lysine, threonine, methionine, phenylalanine, tryptophan and valine). It should also ideally include most of the non-essential amino acids. Glutamine tends not to be included because of problems with stability and solubility but there is much interest regarding glutamine supplementation.
Lipid – commonly given as Intralipid, an emulsion made from soya with chylomicron sized particles. Provides a source of essential fatty acids – linolenic acid (an omega-3 fatty acid) and linoleic acid (an omega-6 fatty acid) – and is a vehicle for giving fatsoluble vitamins.
Lipid preparations are expensive and it is possible to give parenteral nutrition with low levels of lipid; giving 6% of total energy requirement as lipid is enough to avoid essential fatty acid deficiency. This would involve giving the patient in the example earlier 500 ml of 20% Intralipid (providing 1000kcal) once a week.
If no parenteral lipid is given, vegetable oil should be massaged into the patient’s limbs once a day; lipid is absorbed through the skin and may prevent or delay essential fatty acid deficiency although requirements in critical illness may be too high for this to be sufficient. Watch for signs of deficiency: dry, scaly skin with or without hair loss and abnormal liver function tests. Most vegetable oils can be used (safflower, corn, soya, groundnut or sunflower) but not palm oil as it contains virtually no linolenic acid. Fat soluble vitamins will need to be given separately.
Carbohydrate – given as glucose.
Electrolytes & Micronutrients – some may be included in the emulsion. If not, they need to be given separately. Guidelines for requirements for electrolytes, vitamins and trace elements are given in the appendix although patients may have abnormal losses depending on the pathology involved. Patients with sepsis have been shown to have large vitamin A losses in their urine, burns patients lose selenium, zinc and copper via their exudates and trauma patients lose selenium and zinc through their drains.
Complications of nutritional support
There are a number of complications it is important to be aware of and to look for in patients receiving nutritional support.
1. Refeeding syndrome
Patients who are severely malnourished or have undergone a significant period of starvation are at risk of refeeding syndrome during the first few days of nutritional support regardless of route. The clinical features include weakness, respiratory failure, cardiac failure, arrhythmias, seizures and death. Starvation causes a loss of intracellular electrolytes, secondary to leakage and reduced trans-membrane pumping, and intracellular stores can become severely depleted even though serum levels may be normal. When carbohydrate is available again there is an insulin-dependent influx of electrolytes into the cells which can result in rapid and severe drops in serum levels of phosphate, magnesium, potassium and calcium.
At risk patients should be identified and feeding must be introduced slowly, starting with only 25-50% of energy requirements and gradually increasing after four days. The above electrolytes should be generously supplemented at the same time as starting feeding and should be closely monitored. Thiamine and other B vitamins should also be given intravenously before starting feeding and then daily for at least three days.
2. Overfeeding
Deliberate overfeeding has been tried in an attempt to reverse catabolism but this does not work and is associated with a poor outcome. It can cause uraemia, hyperglycaemia, hyperlipidaemia, fatty liver (hepatic steatosis), hypercapnia (especially with excess carbohydrates) and fluid overload. It is probable that at least some of the risks of parenteral nutrition are actually related to overfeeding and some people now recommend deliberate underfeeding (aiming to meet roughly 85% of requirements).
Propofol (either 1% or 2%) comes in 10% Intralipid and where it is being used for sedation this must be included in the calculations for nutritional support if over feeding is to be avoided.
3. Hyperglycaemia
Hyperglycaemia can be related to overfeeding but is often not; critically ill patients become insulin resistant as part of the stress response. Strict control of blood glucose has been shown to have a profound effect on mortality in a study primarily of post surgical intensive care patients, reducing ICU mortality by almost half (from 8.0% to 4.6%) as well as reducing in-hospital mortality, length of stay, ventilator days, incidence of septicaemia and requirements for both dialysis and blood transfusion. The patients who benefited most were those who stayed on ICU for more than 5 days.
Medical patients on ICU for more than 3 days have also been shown to derive some benefit from tight glycaemic control however the effects were not so marked and there was an apparent increase in mortality in patients on ICU for less than 3 days. It has been suggested that prevention of hyperglycaemia should be the target of treatment in the first three days (glucose < 8.3 mmol/l or 150mg/dl) and then if the patient is still critically ill to then aim for tighter control (4.4 – 6.1 mmol/l or 80 – 110 mg/dl).
4. Specific complications of enteral nutrition
The complications that relate to the various routes of enteral feeding have already been discussed. The commonest risk with enteral feeding is aspiration of feed causing pneumonia. The implementation of a combination of measures similar to those listed earlier as well as a nurse education programme and good oral hygiene has been shown to decrease the risk of ventilator-associated pneumonia. Diarrhoea can also be a problem but is not an indication to stop feeds. Other causes of diarrhoea need to be excluded but if enteral feeds are the cause then a feed with more fibre can be tried.
5. Specific complications of parenteral nutrition
The insertion and presence of a central venous catheter has inherent risks including pneumothorax, haemothorax, arterial or nerve damage, infection, central vein thrombosis and erosion through the wall of the vein or right atrium. It is thought that some of the increased risk of sepsis seen in patients receiving parenteral nutrition has actually been the result of overfeeding and uncontrolled hyperglycaemia rather than due to parenteral nutrition itself. Nevertheless, infection is a serious risk and the bags must be sterile and discarded within 24 hours of starting. Sterile precautions must be used when changing bags and the lumen of the central venous line must not be used to take blood or give drugs or fluids (this may also result in precipitation of the emulsion).
Parenteral nutrition also predisposes to hepatobiliary disease including fatty liver, cholestasis and acalculous cholecystitis and great care must be taken to avoid electrolyte imbalances and micronutrient deficiencies particularly in those requiring nutritional support for prolonged periods.
Summary
Malnutrition is associated with a poor outcome in critical illness. Enteral nutrition is the mainstay of nutritional support and should be started early in all patients in whom it is safe to do so. It is not without risk but these risks can be significantly reduced with simple measures. Parenteral nutrition has a definite role but only in selected patients. In all patients receiving nutritional support it is vital to achieve glucose control with insulin therapy and important not to overfeed.
Appendix
1. Example of a recipe for enteral feeding
Preetika A, Padmini G, Shobha U. Nutrient dense mixes for enteral feeding in India. Nutrition and Food Science 2004; 34: 277-81. http://www.emeraldinsight.com/Insight/viewContentItem.do?contentType=Article&hdAc tion=lnkpdf&contentId=866255
In this recipe from India the seeds and grains were germinated and malted prior to being dried and ground to decrease viscosity and improve availability of B vitamins, carbohydrates, amino acids and minerals. All the ingredients were available in the local market including the whey protein (British Biologicals). Finger millet and whey protein have been shown to complement each other in terms of amino acid content.
Further details regarding this and other recipes can be found in the above article. Although the nutritional content and microbiological contamination of the mixture was assessed, the feed was not actually given to patients.
If you would like more information on suitable enteral recipes for ICU patients then please contact the author for details.
2. Daily Electrolyte requirements:
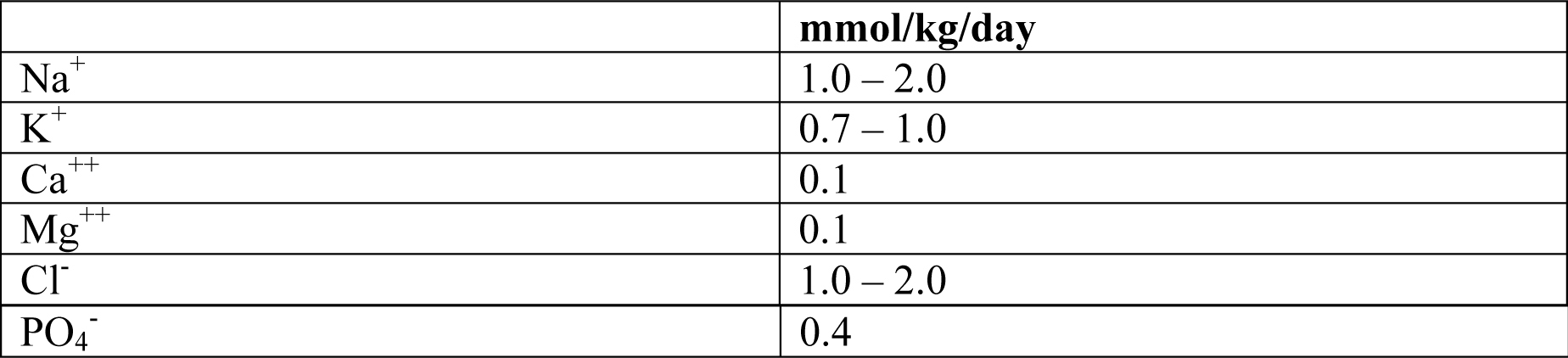
3. Daily requirements for Trace Elements and Vitamins for patients receiving TPN:

Self assessment answers
- How do you assess a patient’s nutritional status?
- Body mass index is frequently used but fails to assess severe acute changes seen in critical illness. The subjective global assessment is reproducible and applicable to this population of patients.
- How would you calculate the energy requirements for a 33 year old man (height 1.80 m and weight 75kg) admitted post-laparotomy for a ruptured appendix and sepsis (temperature 39°C)?
- First estimate BMR then modify this value according to his clinical situation to calculate resting energy expenditure (REE).
- The modifications for temperature and sepsis etc are additive and the caloric requirement works out to approximately 2460 kcal/day as follows:
- BMR = 13.75 x 75kgs + 5 x 180cms – 6.78 x 33years + 66 = 1773.5 kcal/day
- REE = 1773.5 x 1.2 (as add 20% for temperature) x 1.09 (for sepsis) x 1.06 (surgery ) = 2458.9 kcal/day (ie. roughly 2460 kcal/day)
- How do you calculate how much protein / carbohydrate / fat he should receive to make up his energy requirement?
- Provide 40% of his requirement with lipid [2460 x 0.4 = 984 kcal = 106 g lipid]
- Provide 1.5 g/kg/day protein [1.5 x 75 = 112.5 g protein = 112.5 x 5.3 = 596 kcal]
- Make up remaining caloric requirement with carbohydrate
- 2460 – 984 – 596 = 880 kcal = 234 g carbohydrate
- His feed should provide 106 g lipid, 112 g protein and 234 g carbohydrate per day.
- What are the advantages of enteral nutrition over parenteral nutrition?
- Advantages of enteral feeding include cost, reduced risk gastric ulceration, less bacterial translocation and reduced risk of nosocomial infection. Enteral feeding also avoids the risks of parenteral feeding (line insertion, infection.)
- How can you reduce the risk of aspiration pneumonia in a patient who is enterally fed?
- Confirm tube position, secure it well to prevent migration, feed with the patient semirecumbent, feed early, aspirate the NG tube regularly to avoid large residual gastric volumes and avoid bolus feeds for the same reason. Prokinetics have also been shown to reduce residual gastric volumes.
- Is glucose control important? If so, what should the target be?
- Control of glucose has been shown to reduce mortality and ICU morbidity. The benefits seem to be greater for surgical rather than medical patients but the results have been difficult to reproduce so effectively elsewhere. However most ICU’s now regulate glucose more tightly but there is variation in target values. Treatment generally starts if glucose climbs above 8.3 and aims to reduce glucose to a varying degree (the original research suggested a glucose target of 4.4-6.1 mmol/l)
Suggested further reading
- Subjective Global Assessment – Form: www.nephrology.rei.edu/SGA_Original.doc & Instructions: www.nephrology.rei.edu/sga_instr.pdf
- Toronto formula for energy requirements in burns patients-http://www.medbc.com/annals/review/vol_18/num_1/text/vol18n1p16.asp
- Stroud M, Duncan H, Nightingale J. Guidelines for enteral feeding in adult hospital patients. Gut 2003; 52: vii 1. http://gut.bmj.com/cgi/content/full/52/suppl_7/vii1
- Heyland DK. Nutritional support in the critically ill patient – a critical review of the evidence. Critical Care Clinics 1998; 14: 424-40. http://gi.vghtc.gov.tw/GIpersonal/Ko/Johns%20Hopkins%20Hospital%20Residency/Osle rNet%20-%20the%20Osler%20Intranet/oslernet.med.som.jhmi.edu/jhu/ curriculum_FELL/micu/nu trition/heyland.pdf
- Van den Berghe G, Wouters P, Weekers F, Verwaest C et al. Intensive insulin therapy in critically ill patients. New England Journal of Medicine 2001; 345: 1359-67.



