Paediatric Anaesthesia
KEY POINTS
- Anxiety in children at induction of anaesthesia is common, and it is important to develop several strategies to overcome this problem.
- Children with behavioural disorders needing an anaesthetic can be managed using non-pharmacological and pharmacological methods to minimise distress.
- Children’s rights and consent should be considered when performing the induction of anaesthesia.
- Specific co-morbidities may require modifications to standard induction processes; e.g difficult IV access, the unfasted child and the unwell child.
INTRODUCTION
The conduct of paediatric anaesthesia presents many unique challenges. One of the most striking is the variability of behaviour and responses of children and their parents at induction. Behavioural problems, the need for restraint, difficult IV access and co-morbidities add complexity and can make the art of maintaining a calm and smooth induction incredibly difficult. This tutorial will discuss six of the common problems that arise at induction and how these may best be overcome.
CHALLENGE 1: THE ANXIOUS CHILD
Children who are distressed at induction are more likely to experience distress on emergence, and into the post operative period (1).
Known risk factors include
- Children with previous behavioural problems (e.g distress or anxiety) during a health care encounter or greater than five previous hospital admissions have an increased risk of high anxiety at induction(2).
- Anxious parents
- Behavioural disorders including autistic spectrum disorder (ASD), attention deficit disorder (ADD) and attention deficit hyperactivity disorder (ADHD).
- Age specific considerations
- 0 -12 months: Demonstrate general distress but it is not until several months later that they demonstrate fear responses(3). Children of this age also respond to separation with soothing and distraction. For this reason pharmacological anxiolytics are seldom used, and parental presence is usually at the discretion of the anaesthetist.
- 1-2 years: Increasing attachment and fear with increased mobility and strength. Premedication again seldom used in this group, but use of distraction techniques may be of benefit.
- 2 to 5 years: Increased likelihood of separation anxiety from parents. At this age increased physical strength and inability to rationalise behaviour may make them a particularly important age group to assess for anxiolytic strategies.
- 5 years to Adolescence: Increased sense of self and potential harm. School aged children may respond to explanation and reason. Distraction techniques are also particularly useful in this age. A pilot study looking specifically at adolescents found over 80% of this age group reported significant anxiety at the time of induction. Underlying baseline anxiety, depression, somatization (physical symptoms occurring secondary to psychological distress) and fearful temperament are predictors of this phenomenon(4).
Non-pharmacological anxiolysis:
A Cochrane review (5) of 28 trials divided these heterogeneous interventions into five main categories. Below is a summary of their findings:
- Child interventions (Passive): Different studies showed variable response to videos in terms of anxiety reduction. One study suggested a video of patient’s choice may decrease anxiety, but another using a fairytale video could not demonstrate such a difference.
- Child interventions (Interactive): When compared with parental presence only, clowns or clown doctors significantly lessened children’s anxiety in the operating/induction room in three trials with a total of 133 children. Video game usage also decreased anxiety compared to controls and patients who had received midazolam.
- Child interventions (Mask introduction): A single study on mask familiarity showed no reduction in anxiety but better co-operation
- Parental presence: Parental presence does not decrease a child’s anxiety, and is less effective than a premedication. It is, however, important to recognize that the majority of studies demonstrating this are from a similar patient group and may not reflect cultural, religious and ethnic variations.(6)
- Parental interventions: Children of parents having sham acupuncture may be less anxious.
It is important to use age appropriate distraction techniques. There is increasing research being done into use of modern technology including tablet devices and virtual reality. It is important to consider all the available resources, and not disregard simple games or the expert storyteller as a distraction method.
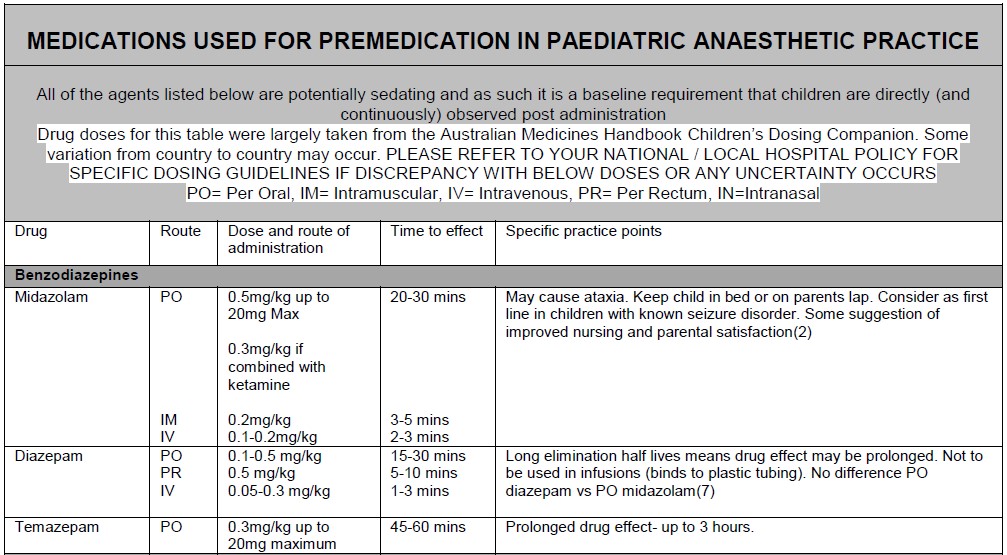
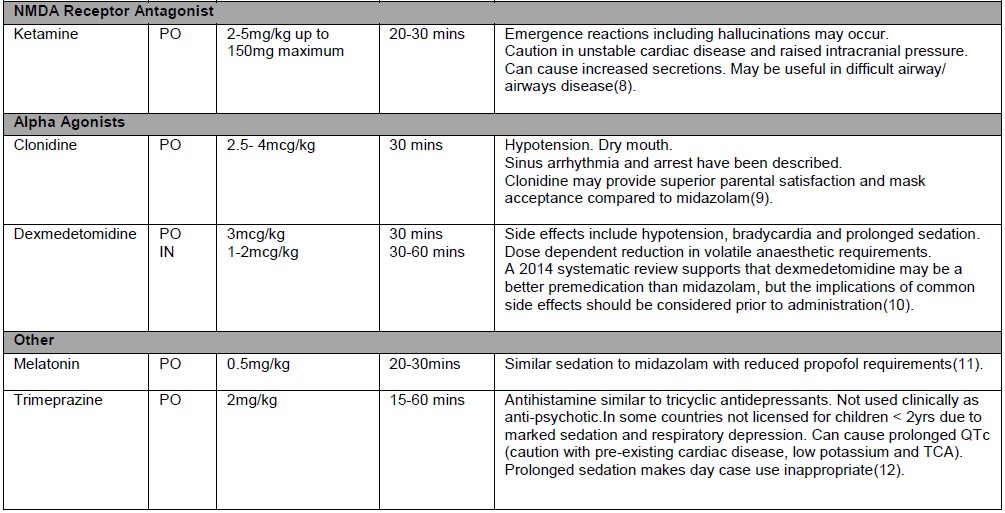
Pharmacological anxiolysis:
The practice of premedication is heterogeneous across institutions and can be dependent on institutional culture and patient populations. Nonetheless, whatever the practice in your institution, there are some key factors for safe and successful premedication:
- Give the premedication enough time to work
- Ensure adequate monitoring facilities are available
- Select appropriate agents based on previous experiences, severity of anxiety or behavioural disturbance and comorbidities.
- Consider combination therapies using lower dose ranges of each agent: e.g.
- Oral ketamine (3 mg/kg) plus midazolam (0.3 mg/kg)
- Oral ketamine 3mg/kg and clonidine 3mcg/kg
- Consider possible contraindications (e.g. unfasted, difficult airway, critically unwell, end organ dysfunction such as liver or renal failure)
There are a number of excellent freely available articles that cover the topic of perioperative anxiety in more detail. (13, 14)
CHALLENGE 2: THE CHILD WITH A BEHAVIOURAL DISORDER
Wide ranges of behavioural disorders exist. These include ASD, ADD and ADHD (see above). Some children may not have a specific diagnosis, but parental reporting of difficulties with behaviour should also be considered when planning in the perioperative period. Also consider the implication of developmental delay on how the child will respond when stressed (see age specific considerations above).
Parents or carers form a vital part of a smooth perioperative pathway. Discussion with the parents regarding behaviour and compliance is important to assess likelihood of peri-operative co-operation. This includes the usual rituals and routines of the child, and how these can be disrupted as little as possible. Asking the parents about the signs of the child becoming upset, any specific triggers and what strategies the family use to try and ease this will assist with recognition of increasing distress.
Children with ASD are more likely to have associated co-morbidities(15) including seizure disorders, sleep apnoea and developmental delay. A study looking at the experience of children with ASD in the peri-operative period found that children with ASD were more likely to receive no premedication or a non-standard premedication (anything other than oral midazolam)(16). It may be that children with this condition are not receiving a premedication when they may benefit from it. Children with ASD need a tailored anxiolytic strategy, best formulated by assessing their preoperative behaviour and talking to their parents. Another literature review strongly suggests premedication, as part of a ‘flexible’ preoperative process, e.g measure height and weight at home, talk to the parents before the day of surgery, minimise waiting time and provide access to a quiet room(15). This is important because difficulties with social interactions form a prominent feature of the disorder, and placing the child in a crowded, noisy room may lead to increased distress, and lessen cooperation.
Children with ADHD can be less cooperative at induction of anesthesia and have a greater incidence of post operative maladaptive behaviors.(17)
It is also important to consider the implications of therapy on anaesthetic practice. Children with ASD, ADD and ADHD can be medicated with a wide range of psychoactive drugs and stimulants. There have been case reports of increased drug requirements for sedation and anaesthesia, and various haemodynamic effects e.g. refractory hypotension with risperidone (18). There is currently no clear evidence based guidelines on perioperative management of stimulant medication and the decision is usually made by the prescribing doctor and the parent or guardian(19).
CHALLENGE 3: THE RESISTANT CHILD – RESTRAINT AND CONSENT
Children are often reluctant to have a stranger hold a mask on their face (mask acceptance) or insert an intravenous cannula. The question then becomes what to do when a patient refuses these interventions. How much restraint is ok? Who can enforce it?
In a USA based survey of paediatric anaesthetists, 44% of respondents used restraint in the majority of children <1 year of age, whereas only 2% did so in children >11 years(20). In a similar UK based survey, the majority of respondents rarely or never allow a child to be restrained by staff members, the size of this majority growing with the increasing age of the child from 214 (74%) under 1 year to 261 (88%) over 6 years. Notably 110 (25%) had cancelled a case within the past 5 years and 196 respondents (45%) had cancelled at least one case during their entire career because of child refusal (21). Local policy and laws will dictate the exact requirements for consent. For example, in the UK and Australia the laws surrounding consent in children is guided by the concept of ‘Gillick competence’ where in order to withhold consent a child must have sufficient understanding and intelligence to enable him or her to fully understand what is proposed(22). This means if a child is capable and refuses treatment but is held down against their will, we would be enforcing treatment without consent. In this sense every effort should be made to follow children’s wishes. The 2003 working group of the Confederation of European Specialists in Paediatrics (CESP) stated that children may effectively refuse treatment or procedures that are not necessary to save their lives or prevent serious harm(23).
There are a limited number of specific guidelines on restraint in paediatrics. Local policies should be consulted to ensure local protocols are adhered to. Homer and Bass (21) reference the UK Royal College of Nursing guidelines on procedural restraint, outlining the difference between restraint and holding still. These guidelines distinguish these two techniques through the amount of force applied and consent. The American Society of Pediatricians has a statement on the use of physical restraint in the acute care setting(24). In this context it outlines restraint as physical or mechanical methods to restrict movement. This statement is not specific to anaesthetics but an example of this in anaesthetic practice would be wrapping a patient in a blanket to keep arms and legs still. Therapeutic holding as per this same statement is the physical restraint of a patient by at least two people to assist the patient who has lost control of behavior to regain control of strong emotions. Consistently, across the available resources, there is emphasis on the importance of explaining this type of intervention to the child and their parents, and ensuring the child is not injured in any way during this process. The ‘competent’ child also has the right to refuse a treatment or intervention.
CHALLENGE 4: THE UNFASTED CHILD OR POTENTIAL FULL STOMACH
Some clinical circumstances require anaesthesia to commence prior to adequate fasting. Children are also more prone to gastric stasis with minimal trauma, opioid analgesia, and pain(25). The place of paediatric rapid sequence induction remains controversial. At times, even when a rapid sequence induction is indicated, performance is impossible e.g. difficult IV access or non-compliance with pre-oxygenation(25). In this setting attempts should be made either to site an IV prior to induction or minimize disturbing the patient during an inhalational induction (e.g. movements or cannulation when ‘light’). It is important to have suction available and voice concerns about potential aspiration to other staff. The anaesthetist should also ensure airway assistant is comfortable performing cricoid pressure.
For general information about rapid sequence induction please refer to ATOTW 331.
CHALLENGE 5:THE CHILD WITH DIFFICULT IV ACCESS
Careful assessment of venous options prior to any attempts will allow selection of the best site. Application of a topical local anaesthetic agent can be used to reduce pain associated with cannulation without negatively impacting on success rates(26). Parental application (e.g at home prior to presenting to hospital) of these agents has also been shown to be effective(27). If an IV induction is planned it is reasonable to use topical local anaesthetic agents when able.
Use of specialized lights for vein identification or transillumination in infants can be helpful. Ultrasound can be of use if available, the anaesthetist has the expertise, and the patient is cooperative. The ultrasound can be used either real time or to identify and mark the location of veins that are not visible/ palpable. Be sure to maintain the arm in the same position during scanning and the cannulation attempt.
If an IV induction is considered essential or desirable but an IV cannot be placed IM ketamine can be used as a general anaesthetic agent. The IO (intraosseous) route has been successfully used for rapid induction of anaesthesia with minimal complication(28).
It is also well known that cannula placement is easier in the anaesthetized patient (immobile and vasodilated) and an inhalational induction may be preferred in patients with a history of difficult access. All of the strategies outlined above for vein localization can be used following a gas induction. Whilst not routine, in a patient with difficult access some anaesthetists will perform airway instrumentation without IV access. This allows establishment of a more secure airway (LMA/ ETT). The key to this approach is ensuring the patient is sufficiently deep (with slower heart rate, no response to stimulation such as jaw thrust, non-divergent eyes and mid-sized pupils) prior to airway instrumentation to avoid coughing or airway stimulation resulting in laryngospasm or regurgitation. A dose of IM suxamethonium 4mg/kg can facilitate paralysis if required although it takes longer than the IV route to achieve adequate muscle relaxation and muscle fasciculations may not be visible.
CHALLENGE 6: THE UNSTABLE OR UNWELL CHILD
The increased work and sympathetic surge associated with being stressed or upset prior to induction can cause clinical decompensation in unwell or unstable children with limited physiological reserve. This can occur before induction agents have been given. Alternatively the critically ill patient may make little attempt to ‘fight’ induction – a marker of how unwell they are. Cautious premedication in a monitored and supervised environment may be appropriate and may decrease the induction doses required. Preparation is again vitally important in these patients. If possible, attach monitoring prior to induction and ensure resuscitation drugs are immediately available. When administering any agents, start ‘low and slow’, using small doses slowly titrated to effect. Propofol is used less commonly in these circumstances with many practitioners favoring agents such as ketamine, midazolam, fentanyl and remifentanil.
For a more comprehensive summary of intubation of sick children please refer to ATOTW 169.
SUMMARY
Anyone who has spent time in paediatrics will have been witness to inductions of anaesthesia that have gone very well, or been very difficult. Outlined above are some of the challenges faced in the day-to-day conduct of paediatric anaesthesia. Flexibility in planning and implementation of an induction strategy will allow a tailored perioperative pathway for each patient. Assessment of children for factors that may complicate induction including pre-operative anxiety, possible behavioural problems, need for restraint, fasting status, critical illness and difficult IV access may allow selective use of appropriate pharmacological and/or non-pharmacological strategies. This will hopefully improve the child and their parents’ experience of what can be an incredibly stressful and difficult event.
REFERENCES AND FURTHER READING
- Holm-Knudsen RJ, Carlin JB, McKenzie IM. Distress at induction of anaesthesia in children. A survey of incidence, associated factors and recovery characteristics. Paediatric Anaesthesia 1998;8(5):383-92.
- Davidson AJ, Shrivastava PP, Jamsen K, Huang GH, Czarnecki C, Gibson MA, et al. Risk factors for anxiety at induction of anesthesia in children: a prospective cohort study. Pediatric Anesthesia 2006;16(9):919-27.
- Rothbart MK, Ahadi SA. Temperament and the development of personality. Journal of Abnormal Psychology 1994;103(1):55-66.
- Fortier MA, Martin SR, Chorney JM, Mayes LC, Kain ZN. Preoperative anxiety in adolescents undergoing surgery: a pilot study. Pediatric Anesthesia 2011;21(9):969-73.
- Manyande A, Cyna AM, Yip P, Chooi C, Middleton P. Non-pharmacological interventions for assisting the induction of anaesthesia in children. Cochrane Database of Systematic Reviews 2015(7).
- Rosenbaum A, Kain ZN, Larsson P, Lonnqvist PA, Wolf AR. The place of premedication in pediatric practice. Pediatric Anesthesia 2009;19(9):817-28.
- Vetter TR. A comparison of midazolam, diazepam, and placebo as oral anesthetic premedicants in younger children. Journal of Clinical Anesthesia 1993;5(1):58-61.
- Jamora C, Iravani M. Unique clinical situations in pediatric patients where ketamine may be the anesthetic agent of choice. American Journal of Therapeutics 2010;17(5):511-5.
- Almenrader N, Passariello M, Coccetti B, Haiberger R, Pietropaoli P. Premedication in children: a comparison of oral midazolam and oral clonidine. Pediatric Anesthesia 2007;17(12):1143-9.
- Sun Y, Lu Y, Huang Y, Jiang H. Is dexmedetomidine superior to midazolam as a premedication in children? A meta-analysis of randomized controlled trials. Pediatric Anesthesia 2014;24(8):863-74.
- Gitto E, Marseglia L, D’Angelo G, Manti S, Crisafi C, Montalto AS, et al. Melatonin versus midazolam premedication in children undergoing surgery: A pilot study. Journal of Paediatrics and Child Health 2016;52(3):291-5.
- Bramwell R, Manford M. Premedication of children with trimeprazine tartrate. British Journal of Anaesthesia 1981;53(8):821-6.
- O’Sullivan M, Wong GK. Preinduction techniques to relieve anxiety in children undergoing general anaesthesia. Continuing Education in Anaesthesia Critical Care & Pain 2013;13(6):196-9.
- Tan L, Meakin GH. Anaesthesia for the uncooperative child. Continuing Education in Anaesthesia Critical Care & Pain 2010;10(2):48-52.
- Taghizadeh N, Davidson A, Williams K, Story D. Autism spectrum disorder (ASD) and its perioperative management. Pediatric Anesthesia 2015;25(11):1076-84.
- Arnold B, Elliott A, Laohamroonvorapongse D, Hanna J, Norvell D, Koh J. Autistic children and anesthesia: is their perioperative experience different? Pediatric Anesthesia 2015;25(11):1103-10.
- Tait AR, Voepel-Lewis T, Burke C, Doherty T. Anesthesia induction, emergence, and postoperative behaviors in children with attention-deficit/hyperactivity disorders. Pediatric Anesthesia 2010;20(4):323-9.
- Wong G, Lee R. Refractory hypotension in an autistic child on risperidone: Association of Anaesthetists of Great Britain and Ireland; 2017 [Case Report]. Available from: http://www.anaesthesiacases.org/case-reports/2017-0008.
- Cartabuke RS, Tobias JD, Rice J, Tumin D. Hemodynamic profile and behavioral characteristics during induction of anesthesia in pediatric patients with attention deficit hyperactivity disorder. Pediatric Anesthesia 2017;27(4):417-24.
- Lewis I, Burke C, Voepel-Lewis T, Tait AR. Children who refuse anesthesia or sedation: a survey of anesthesiologists. Pediatric Anesthesia 2007;17(12):1134-42.
- Homer JR, Bass S. Physically restraining children for induction of general anesthesia: survey of consultant pediatric anesthetists. Pediatric Anesthesia 2010;20(7):638-46.
- Bird S. Consent to medical treatment: the mature minor. Australian Family Physician 2011;40:159-60.
- De Lourdes Levy M, Larcher V, Kurz R. Informed consent/assent in children. Statement of the Ethics Working Group of the Confederation of European Specialists in Paediatrics (CESP). European Journal of Pediatrics 2003; 162(9):629-33.
- Committee on Pediatric Emergency Medicine, American Academy of Pediatrics. The use of physical restraint interventions for children and adolescents in the acute care setting. Pediatrics 1997;99(3):497-8.
- Newton R, Hack H. Place of rapid sequence induction in paediatric anaesthesia. BJA Education 2016;16(4):120-3.
- Arendts G, Stevens M, Fry M. Topical anaesthesia and intravenous cannulation success in paediatric patients: a randomized double-blind trial. British Journal of Anaesthesia 2008;100(4):521-4.
- Koh JL, Fanurik D, Stoner PD, Schmitz ML, VonLanthen M. Efficacy of parental application of eutectic mixture of local anesthetics for intravenous insertion. Pediatrics 1999;103(6):e79.
- Neuhaus D, Weiss M, Engelhardt T, Henze G, Giest J, Strauss J, et al. Semi-elective intraosseous infusion after failed intravenous access in pediatric anesthesia. Pediatric Anesthesia 2010;20(2):168-71.



