Paediatric Anaesthesia
Key Points
- Postoperative nausea and vomiting (PONV) is distressing and increases postoperative morbidity.
- The Vomiting Centre co-ordinates the physiological reflex of vomiting, with central and peripheral inputs.
- PONV prevention requires identification of children at increased risk, utilising anaesthetic techniques which minimise PONV incidence, and implementing antiemetic prophylaxis adjusted for their level of risk.
- 5-HT3 antagonists and/or dexamethasone are the mainstay of prophylaxis. Evidence also exists for alternative pharmacological and non-pharmacological POV prophylaxis.
- Guidelines are likely to evolve as more evidence is gathered regarding children in particular, and as novel anti-emetic agents emerge.
Introduction
Postoperative nausea and vomiting (PONV) and postoperative vomiting (POV) are nausea (the subjective feeling of sickness) and/or vomiting (the objective response of ejecting matter from the stomach) following anaesthesia or surgery. They are distressing symptoms that may adversely affect patient and carer satisfaction as well as increase the length of stay and readmission rates.1 Prolonged POV may lead to other complications such as dehydration and electrolyte imbalance, wound breakdown or bleeding or aspiration pneumonitis,1 making its preventionand management crucial to perioperative care. Given the difficulty that younger or preverbal children have in indicating the sensation of nausea,postoperative vomiting is considered a more appropriate measurable endpoint, and children are reported to have twice the incidence of POV (13% vs. 42%), compared to adults.2
This tutorial will discuss paediatric POV and cover the physiological mechanism of nausea and vomiting (due to paucity of studies in children, this is based on adult studies), the assessment and mitigation of risk factors, and the pharmacological and non-pharmacological options for the control of nausea and vomiting.
Physiological Pathway of Vomiting
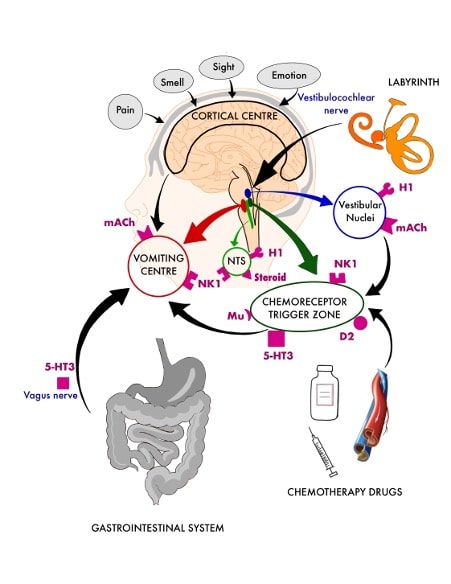
Figure 1: Pathways involved in nausea and vomiting
- D2 = Dopamine 2 receptor;
- 5-HT3 = 5-Hydroxytryptamine 3 receptor;
- NK-1 = Neurokinin type-1 receptor;
- H1 = Histamine 1 receptor;
- mACh = Muscarinic acetylcholine receptor;
- Mu = m–opioid receptor
The vomiting centre, located in the medulla, controls the act of vomiting. It receives signals from muscarinic acetylcholine (mACh) and neurokinintype 1 (NK1) receptors and integrates signals from the following sources via neural pathways1,3 (Figure 1).
- Chemoreceptor trigger zone (CTZ): The CTZ is situated in the area postrema of the medulla, which is outside the blood-brain barrier; therefore, it is quicker to respond to emetogenic stimuli in blood than the vomiting centre. The CTZ also relays information from various other sources, such as the vestibular apparatus.
Receptors: Dopamine 2 (D2), 5-hydroxytryptamine 3 receptor (5HT3), NK1 and l-opioid receptors (Mu)
- Gastrointestinal (GI) tract: In response to infection, the enterochromaffin cells present in the GI tract release serotonin (5HT); themechanoreceptors and chemoreceptors then relay the information to the vomiting centre via 5HT3 receptor activation of vagal afferents in the GI tract.
Receptor: 5HT3 receptors
- The vestibular apparatus, located close to the vomiting centre, sends information regarding the position and head movement either directly to the vomiting centre or via the CTZ.
Receptors: Histamine 1 (H1) and mACh
- Cortical higher centres: This area mediates aspects of pain, objectionable psychological factors, and unpleasant smells and
- The nucleus tractus solitarius lies in the medulla close to the It acts as a relay centre, gathering all visceral afferents (via cranial nerves IX and X) and is responsible for organisation of the final common pathway of vomiting.
Receptors: H1, NK1 and corticosteroid
POV Risk Factors
To devise appropriate antiemetic prophylaxis and treatment, it is essential to estimate the child’s risk of developing POV/PONV. The risk factors in the paediatric population can be categorised into pre-, intra- and postoperative4, summarised in Table 1.
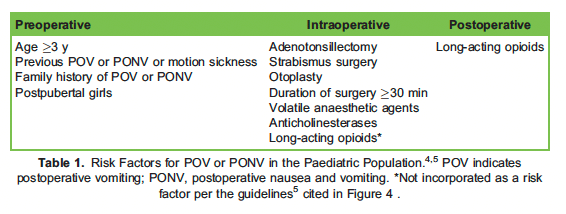
Table 1: Risk factors for POV/PONV in the paediatric population 4,5. POV indicates postoperative vomiting; PONV, postoperative nausea and vomiting. *Not incorporated as a risk factor per the guidelines5 cited in Figure 4 .
Preoperative factors
- Age: The risk of POV increases in individuals aged 2:3 years and continues to rise with increasing 5
- Sex: Girls of postpubertal age are at a higher risk compared with their male counterparts; this is hypothesised to be related to the increase in sex hormone production.5
- History of POV, PONV or motion sickness: Previous and family history of POV or PONV are considered to be independent risk factors, and prior motion sickness is a specific risk factor in adults and children. 5
Intraoperative factors
- Type of surgery: Children undergoing adenotonsillectomy, strabismus surgery or otoplasty are at a higher risk of POV or PONV. 5
- Volatile anaesthetic agents: the use of total intravenous anaesthesia and thus avoiding volatiles for maintenance has been shown to reduce the risk significantly.4,5 There is still no clear consensus on whether nitrous oxide is a risk factor.4,5
- Duration of surgery 2:30 minutes: Longer operations require a longer duration of With volatile agents, this is an established risk factor for vomiting in both adults and children.4
- Use of anticholinesterases: UK guidelines advise avoiding the use of neostigmine in children who are at high risk of vomiting.5
- Long-acting opioids: intraoperative administration of longer acting opioids can increase POV compared with short-acting opiates. 6
Postoperative factors
– Use of long-acting opioids
- The use of long-acting opioids in the postoperative ward has been shown to increase the incidence of vomiting, hence opiate sparing-techniques are recommended, especially for high-risk patients.4

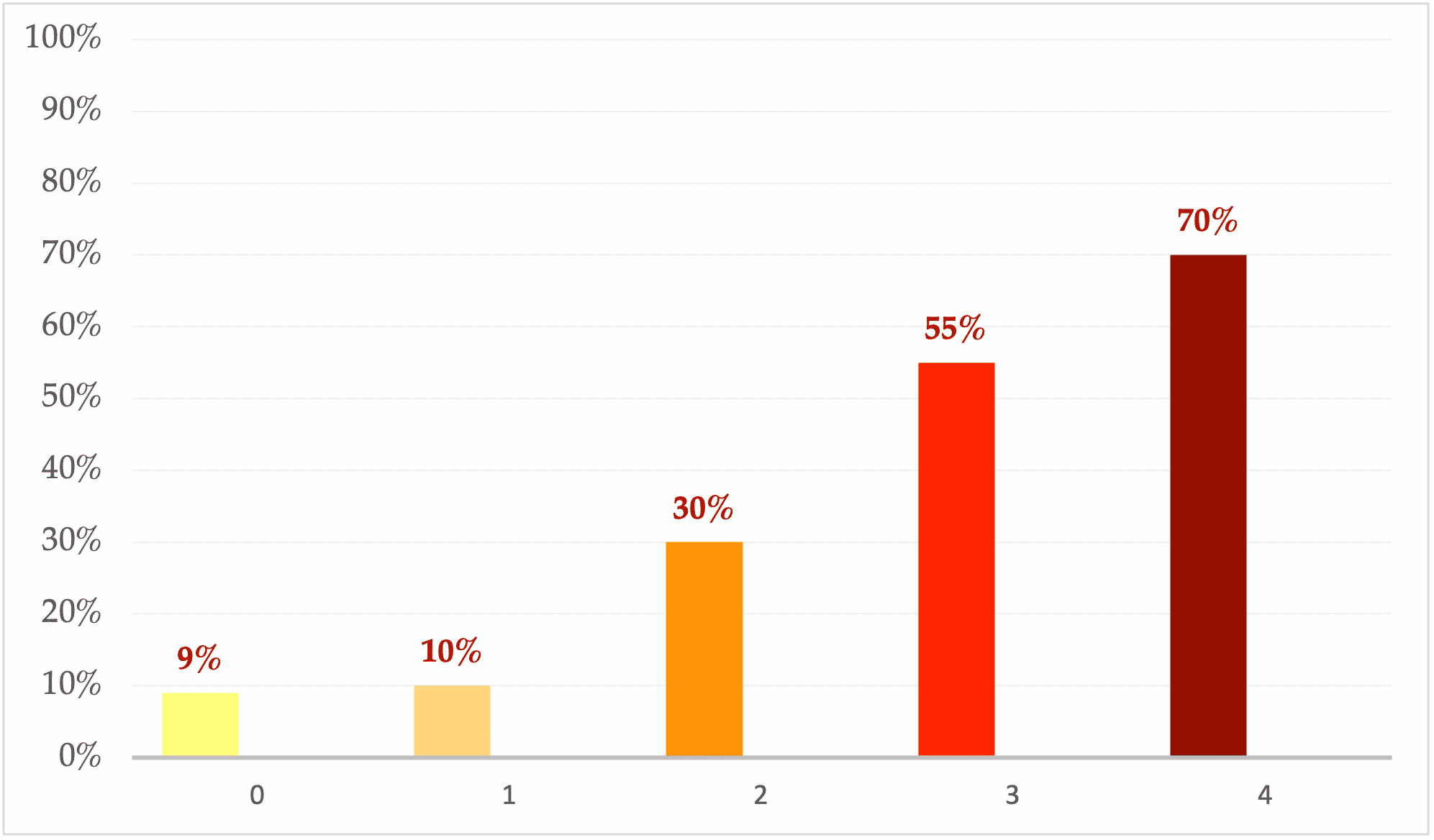
Figure 2: Eberhart’s simplified predicted risk score for POV in children (POVOC-score) 7
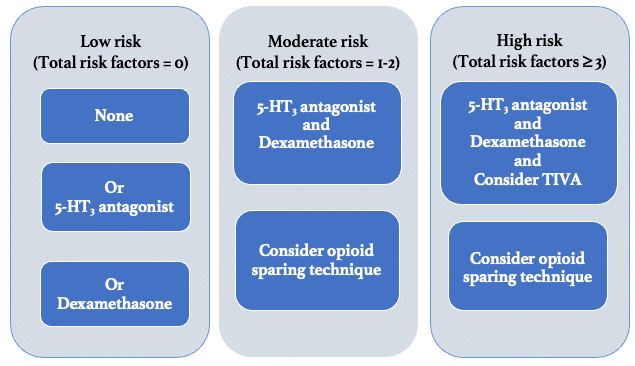
Figure 3. Example Paediatric POV prophylaxis regimen4. For risk factors, see Table 1.
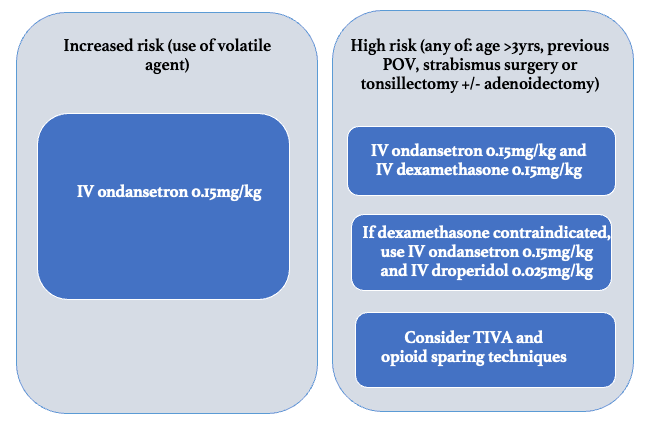
Figure 4. Example Paediatric POV prophylaxis regimen 5
Risk stratification
The simplified risk score for POV in children (POVOC score) of Eberhart et al7 demonstrates how accumulating risk factors is associated with an increase in the incidence of POV. This risk score incorporates duration of surgery 2:30 minutes, age 2:3 years, strabismus surgery and previous or family history of POV (Table 2). When 0, 1, 2, 3 or 4 of the depicted independent predictors are present, the corresponding risk for PONV is quoted as approximately 9%, 10%, 30%, 55% or 70%, respectively (Figure 2). Alternative methods of risk stratification have been incorporated in national POV prophylaxis guidelines4,5 and are summarised (along with prophylaxis recommendations, which will bediscussed later) in Figures 3 and 4.
Risk Reduction
Choice of anaesthetic technique
The use of total intravenous anaesthesia (TIVA) has been shown to reduce paediatric POV, however it is associated with an increased risk of bradycardia in particular during strabismus surgery secondary to the oculocardiac reflex 4.
Opioid sparing techniques
Evidence supports the use of opioid sparing techniques in preventing paediatric POV 5,6. This can be achieved using nonopioid systemic analgesics (such as paracetamol and/or lidocaine) and regional anaesthetic procedures (e.g. caudal, transversus abdominis plane or rectus sheath blocks which are safe and efficacious). 4
Fluid administration
It has been suggested that prolonged fasting is associated with nausea and vomiting. A recent review recommends early and liberal resumption of oral fluid intake postoperatively as it is associated with reduced POV 8.
Premedication
The concurrent use of α2 agonists such as intranasal dexmedetomidine or oral clonidine are associated with reduced rates of PONV in children compared to other premedication or placebo 9,10, but further studies would be beneficial.
Pharmacological Agents for Prophylaxis/treatment
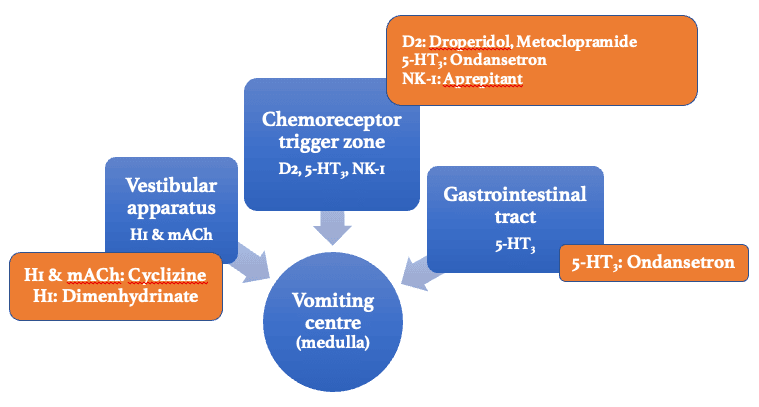
Figure 5: Summary of antiemetic sites of action
- D2 = Dopamine 2 receptor;
- 5-HT3 = 5-Hydroxytryptamine 3 receptor;
- NK-1 = Neurokinin type-1 receptor;
- H1 = Histamine 1 receptor;
- mACh = Muscarinic acetylcholine receptor.
The following are antiemetic medications as they relate to their receptors (Figure 5).
Primary 5-HT3 Antagonist: Ondansetron
This class of medications has a large body of evidence supporting its use in preventing paediatric POV,4 and the use of ondansetron iswidespread. Ondansetron has been demonstrated to be effective at reducing POV in children in a dose-related manner, with the oral route (if tolerated) having similar efficacy to the intravenous route, and the timing of the dose (from preinduction to postoperative) having no bearing on effect.5 It has been shown to be superior to both droperidol and metoclopramide11 and is therefore recommended as first line for prevention of POV in the United Kingdom and United States.4,5 Other 5-HT3 agonists such as tropisetron, granisetron, dolasetron, ramosetron andpalonosetron have varying levels of evidence and are not all licensed for use in children.
Ondansetron is a carbazole derivative antagonising 5-HT3 receptors (hypothesised to have both central and peripheral effects1).
- Dose: 50–150 μg/kg up to 4 mg
- Side effects: headache, constipation, cardiac arrhythmias; it is contraindicated in long QT syndrome12
Dopamine Antagonists: Metoclopramide, Prochlorperazine, Droperidol
Due to a lack of evidence to support the use of metoclopramide in preventing paediatric POV, and an increased rate of extrapyramidal sideeffects in children compared with adults,13 it is not recommended for this purpose in the United Kingdom.5 However, in the United States,metoclopramide is considered an option for treatment of POV if other prophylactic agents have failed.4 Prochlorperazine is not recommended to prevent POV in children as it has not been studied in this context and has a wide side effect profile.5 The use of droperidol has been limited in practice due to potential extrapyramidal side effects or arrhythmias; however, UK guidelines recommend a low dose as second-linemedication for prevention or treatment of POV, as it has been shown to be effective and safe unless contraindicated (eg, in patients with long QT syndrome).5
Metoclopramide
Metoclopramide is a benzamide derivative that acts as a dopamine antagonist and also acts directly on the GI tract.
- Dose: 100-150 μg/kg (maximum 10 mg)
- Side effects: extrapyramidal effects (eg, oculogyric crisis or tardive dyskinesia); it can cause neuroleptic malignant syndrome, as well as hypotension or arrhythmias12
Droperidol
Droperidol is a butyrophenone that antagonises D2 receptors at the CTZ and peripheral a-receptors.1,3
- Dose: 10-25 μg/kg up to 25 mg; owing to the potential for side effects, the lower dose of 10 μg/kg tends to be used in clinical practice
- Side effects: sedation, apprehension, hypotension, dose-related extrapyramidal side effects and neuroleptic malignant syndrome12
Anticholinergics: Scopolamine
Anticholinergics are selective antagonists at muscarinic receptors and thus have a wide side effect profile including sedation, dry mouth, urinary retention, blurred vision, restlessness and hallucinations. Transdermal scopolamine may have a role in safely ameliorating PONV in adolescents14; however, a meta-analysis recommended against its use for younger paediatric patients.15
Antihistamines: Cyclizine, Dimenhydrinate
Studies on cyclizine for prevention of paediatric POV are lacking; however, what evidence there is indicates that it is ineffective and therefore not recommended in the United Kingdom.5 UK guidelines also state that, although there is literature substantiating the use of dimenhydrinate toreduce paediatric POV, this protective effect may not extend to tonsillectomies.5
Cyclizine
Cyclizine is a H1 antagonist with combined anticholinergic properties. It is used as an antiemetic in motion sickness, radiotherapy and opioid-induced emesis in adults.
- Dose: 0.5-1 mg/kg up to 50 mg
- Side effects: tachycardia with intravenous injection, pain on injection, extrapyramidal symptoms12
Dimenhydrinate
Dimenhydrinate has antiemetic properties attributed to competitive H1 antagonism in the vestibular system
- Dose: 0.5 mg/kg up to 25 mg
- Side effects: antimuscarinic symptoms such as sedation or dry mouth5
NK1 Antagonists: Aprepitant, Fosaprepitant
These newer agents are emerging as potentially efficacious. In ongoing pharmaceutical trials, aprepitant appears to be safe and to have similar efficacy to ondansetron in preventing POV in adults.16 A clinical trial is also underway for paediatric patients.17 Fosaprepitant is the intravenous prodrug of aprepitant.18
Aprepitant acts as a central NK1 receptor antagonist to prevent the binding of substance P to these receptors and the resulting emetic effects.18
- Dose: 3 mg/kg up to 125 mg4
- Side effects: fatigue, headache, reduced appetite, constipation, dyspepsia, hiccups, flushing12
Corticosteroids: Dexamethasone
Multiple studies have shown dexamethasone to reduce paediatric POV compared with placebo, with a low side effect profile.4,5 Suggested mechanisms of action are reduction of either prostaglandins or 5-HT release.19
- Dose: 0.15 mg/kg up to 6.6 mg
- Side effects: administered awake, it can cause perineal warmth. Other adverse effects include hyperglycaemia, mood disturbance and thromboembolism.12 Dexamethasone may precipitate tumour lysis syndrome in patients with large, high- grade tumours (eg, lymphoma or leukaemia).5
Combination Therapies
Used in combination, these agents can have synergistic effects. Evidence shows that ondansetron and dexamethasone used together are more effective at reducing paediatric POV than either agent alone and that ondansetron and droperidol in combination is more effective than ondansetron alone.5
Non-Pharmacological Management
Acustimulation (including acupressure and acupuncture)
Several randomised controlled trials and meta-analyses in children suggest that acupoint stimulation of various modalities at different areas (but in particular the Pericardium 6 [PC6] acupuncture point) is as efficacious at reducing POV as antiemetic drug prophylaxis5. The PC6 point lies between the tendons of the palmaris longus and flexor carpi radialis muscles, 4 cm proximal to the wrist crease.
Other non-pharmacological
The use of ginger, chewing gum, and carbohydrate loading needs more valid studies to claim importance.
Examples of POV Prevention and Treatment Strategies
Paediatric POV risk stratification and prophylaxis recommendations from a recent USA consensus statement4 are summarised in figure 3. For rescue therapy of POV, it is recommended that an alternative agent from a different class is added, e.g. droperidol, dimenhydrinate or metoclopramide, or to consider acustimulation.
UK guidelines 5 categorise POV risk slightly differently but suggest similar prophylaxis regimens with doses (see figure 4). For rescue therapy, ondansetron is recommended if not already given, otherwise an antiemetic from a different class (e.g. dexamethasone or droperidol) is suggested.
Summary
Postoperative nausea and vomiting in children is unpleasant for both children and their carers, and can lead to serious complications, therefore it is important to predict who is at risk and manage them accordingly. This tutorial has explored the physiology of vomiting, highlighted risk factors predisposing to paediatric POV and looked at the evidence base for pharmacological and non-pharmacological prophylactic anti-emetic therapies. There are various guidelines for how to stratify risk and which specific agents to use, examples of which are presented in this tutorial. The common themes for preventing POV in children at higher risk include a combination of a 5-HT3 antagonist such as ondansetron with dexamethasone due to their synergistic effect, along with consideration of TIVA, and/or opiate sparing techniques. There is a lack of research into paediatric POV specifically, and identifying nausea in younger children can be challenging, adding further complexity to the issue. As more studies are conducted, and newer therapies (such as 5-HT3 antagonists not yet licensed for children, or NK1 antagonists) continue to be investigated, the optimal management strategies will also evolve.
References
- Aitkenhead AR, Moppett IK, Thompson JP. Smith and Aitkenhead’s Textbook of Anaesthesia. Sixth Edition. Churchill Livingstone Elsevier. 2013.
- Rose JB, Watcha MF. Postoperative nausea and vomiting in paediatric patients. Br J Anaesth. 1999 Jul;83(1):104-17. doi: 10.1093/bja/83.1.104. PMID: 10616338. Available at https://www.bjanaesthesia.org/article/S0007-0912(17)38436-2/pdf. [Accessed on 09/09/2021]
- Pierre S & Whelan R. Nausea and vomiting after surgery,Continuing Education in Anaesthesia Critical Care & Pain, 2013 Feb 13(1):28–32, https://doi.org/10.1093/bjaceaccp/mks046 [Accessed on 1/05/2022]
- Gan TJ, Belani KG, Bergese S, Chung F, Diemunsch P, Habib AS, Jin Z, Kovac AL, Meyer TA, Urman RD, Apfel CC, Ayad S, Beagley L, Candiotti K, Englesakis M, Hedrick TL, Kranke P, Lee S, Lipman D, Minkowitz HS, Morton J, Philip BK. Fourth Consensus Guidelines for the Management of Postoperative Nausea and Vomiting. Anesth Analg. 2020 Aug;131(2):411-448. doi: 10.1213/ANE.0000000000004833. Erratum in: Anesth Analg. 2020 Nov;131(5):e241. PMID: 32467512. Available at https://journals.lww.com/anesthesia-analgesia/Fulltext/2020/08000/Fourth_Consensus_Guidelines_for_the_Management_of.16.aspx [Accessed on 10/06/2021]
- Baines MS, Holtby H, Carr A, The Association of Paediatric Anaesthetists of Great Britain & Ireland. Guidelines on the prevention of post-operative vomiting in children. 2016. Available at https://www.apagbi.org.uk/sites/default/files/inline-files/2016%20APA%20POV%20Guideline-2.pdf. [ Accessed on 18/04/2021].
- Mukherjee K, Esuvaranathan V, Streets C, Johnson A, Carr AS. Adenotonsillectomy in children: a comparison of morphine and fentanyl for peri-operative analgesia. 2001 Dec;56(12):1193-7. doi: 10.1046/j.1365-2044.2001.02084-4.x. Erratum in: Anaesthesia 2002 Feb;57(2):208. PMID: 11736779. Available at https://onlinelibrary.wiley.com/openurl?spage=1193&genre=article&issn=0003-2409&date=2001&sid=nlm%3Apubmed&issue=12&volume=56. [Accessed on 10/06/2021]
- Eberhart LHJ, Geldner G, Kranke P, Morin AM, Schäuffelen A, Treiber H, Wulf H. The development and validation of a risk score to predict the probability of postoperative vomiting in pediatric patients. Anesth Analg. 2004 Dec;99(6):1630-1637. doi: 10.1213/01.ANE.0000135639.57715.6C. PMID: 15562045. Available at https://journals.lww.com/anesthesia-analgesia/Fulltext/2004/12000/The_Development_and_Validation_of_a_Risk_Score_to.11.aspx [Accessed on 30/08/2021]
- Frykholm, Peter; Disma, Nicola; Andersson, Hanna; Beck, Christiane; Bouvet, Lionel; Cercueil, Eloise; Elliott, Elizabeth; Hofmann, Jan; Isserman, Rebecca; Klaucane, Anna; Kuhn, Fabian; de Queiroz Siqueira, Mathilde; Rosen, David; Rudolph, Diana; Schmidt, Alexander R.; Schmitz, Achim; Stocki, Daniel; Sümpelmann, Robert; Stricker, Paul A.; Thomas, Mark; Veyckemans, Francis; Afshari, ArashPre-operative fasting in children, European Journal of Anaesthesiology: 2022 Jan. 39(1): 4-25 doi: 10.1097/EJA.0000000000001599 [Accessed on 1/05/2022]
- Jun JH, Kim KN, Kim JY, Song SM. The effects of intranasal dexmedetomidine premedication in children: a systematic review and meta-analysis. Can J Anaesth. 2017 Sep;64(9):947-961. English. doi: 10.1007/s12630-017-0917-x. Epub 2017 Jun 21. PMID: 28639236. Available at https://link.springer.com/article/10.1007%2Fs12630-017-0917-x [Accessed on 30/08/2021]
- Alizadeh R, Mireskandari SM, Azarshahin M, et al. Oral clonidine premedication reduces nausea and vomiting in children after appendectomy. Iran J Pediatr. 2012;22:399–403. [Accessed on 30/08/2021]
- Domino KB, Anderson EA, Polissar NL, Posner KL. Comparative efficacy and safety of ondansetron, droperidol, and metoclopramide for preventing postoperative nausea and vomiting: a meta-analysis. Anesth Analg. 1999 Jun;88(6):1370-9. doi: 10.1097/00000539-199906000-00032. PMID: 10357347.Available at https://journals.lww.com/anesthesia-analgesia/Fulltext/1999/06000/Comparative_Efficacy_and_Safety_of_Ondansetron,.32.aspx. [Accessed on 10/06/2021]
- Paediatric Formulary Committee.BNF for Children (online) London: BMJ Group, Pharmaceutical Press, and RCPCH Publications <http://www.medicinescomplete.com> [Accessed on 30/03/2022]
- European Medicines Agency. European Medicines Agency recommends changes to the use of metoclopramide. 2013. https://www.ema.europa.eu/en/news/european-medicines-agency-recommends-changes-use-metoclopramide. [Accessed 09/09/2021]
- Pergolizzi JV, Raffa R, Taylor R. Prophylaxis of postoperative nausea and vomiting in adolescent patients: a review with emphasis on combination of fixed-dose ondansetron and transdermal scopolamine. J Drug Deliv. 2011;2011:426813. doi: 10.1155/2011/426813. Epub 2011 Jul 2. PMID: 21773046; PMCID: PMC3134884. [Accessed on 01/05/2022]
- Kassel L, Nelson M, Shine J, Jones LR, Kassel C. Scopolamine Use in the Perioperative Patient: A Systematic Review. AORN J. 2018 Sep;108(3):287-295. doi: 10.1002/aorn.12336. PMID: 30156728. [Accessed on 01/05/2022]
- Salman FT, DiCristina C, Chain A, Afzal AS. Pharmacokinetics and pharmacodynamics of aprepitant for the prevention of postoperative nausea and vomiting in pediatric subjects. J Pediatr Surg. 2019 Jul;54(7):1384-1390. doi: 10.1016/j.jpedsurg.2018.09.006. Epub 2018 Sep 21. PMID: 30381138. Available at https://www.jpedsurg.org/article/S0022-3468(18)30576-1/fulltext. [Accessed on 10/06/2021]
- NIH U.S. National Library of Medicine Clinicaltrials.gov. https://clinicaltrials.gov/ct2/show/study/NCT00819039 [Accessed on 01/05/2022]
- Langford P, Chrisp P. Fosaprepitant and aprepitant: an update of the evidence for their place in the prevention of chemotherapy-induced nausea and vomiting.Core Evid. 2010;5:77-90. Published 2010 Oct 21. doi:10.2147/ce.s6012 [Accessed on 01/05/2022]
- Kakodkar PS. Routine use of dexamethasone for postoperative nausea and vomiting: the case for. Anaesthesia. 2013 Sep;68(9):889-91. doi: 10.1111/anae.12308. Epub 2013 Jul 15. PMID: 23848292. [Accessed on 30/08/2021]



