General Topics
QUESTIONS
Before continuing, try answering the following questions. Answers will be covered throughout the tutorial and listed at the end.
- Draw a diagram of the myocardial conduction pathways.
- List five causes of a sinus tachycardia.
- Give an example of a class III anti-arrhythmic drug.
INTRODUCTION
The aim of this two-part tutorial is to present the main cardiac arrhythmias along with their accompanying complications and peri-operative management. This tutorial will inform, examine and supply a basic introduction to this topic. It will not cover the acute coronary syndromes or non-shockable cardiac arrest rhythms. It is also worth noting that it may be helpful to seek advice from senior anaesthetic colleagues, as well as specialist cardiology advice where appropriate.
Tutorial one will cover supraventricular arrhythmias. Tutorial two will cover ventricular arrhythmias.
Tutorial one will include:
- Overview of initial rhythm assessment
- A normal ECG and the myocardial conduction pathways
- Narrow complex arrhythmias
a. Sinus arrhythmia
b. Sinus bradycardia
c. Atrial ectopics - Narrow complex tachycardias
a. Sinus tachycardia
b. Supraventricular and junctional tachycardia
c. Atrial flutter
d. Atrial fibrillation
e. Sick sinus syndrome
f. Wolff Parkinson White syndrome - Anti-arrhythmic drugs
RISK AND IMPLICATIONS OF DYSRHYTHMIAS WITH ANAESTHESIA
Life-threatening cardiovascular events under anaesthesia are uncommon. However, adverse cardiovascular events occurring during and after the emergence of anaesthesia occur more frequently. Although the incidence is higher during cardiac surgery, intra-operative dysrrhythmias affect up to 29% of patients undergoing non-cardiac surgery.
Relatively minor fluctuations in cardiovascular and haemodynamic parameters due to arrhythmias can have significant long-term implications, especially on the growing population of elderly co-morbid patients undergoing elective and emergency surgery. For example, post-operative atrial fibrillation is associated with a 2.3 fold increase in stroke risk, as well as a higher incidence of myocardial infarction, congestive cardiac failure, ventricular dysrrhythmias and renal failure.
Vital organs such as the brain, heart and kidneys, must be perfused adequately during general anaesthesia and surgery. An alteration of physiology or the administration of pharmaceuticals is usually the cause for autonomic changes resulting in intra-operative blood pressure and heart rate changes.
The majority of anaesthetic agents have direct myocardial depressant effects, resulting in reduced contractility and reduced sympathetic stimulation of the peripheral vasculature. The net effect is a fall in cardiac output with a fall in perfusing pressure of vital organs secondary to vascular vasodilation. This is particularly dangerous in the hypovolaemic patient at induction of anaesthesia. Agents such as ketamine and ether however, counter their direct depressant effect by also increasing sympathetic activity. This allows better maintenance of blood pressure and cardiac output. Conversely, tachycardia can have detrimental effects in patients susceptible to ischaemia due to reduced myocardial filling time.
Other factors such as electrolyte imbalances, co-morbidity, hormone mediators or direct physical stimulation either by the surgeon or anaesthetist can precipitate dysrrhythmias.
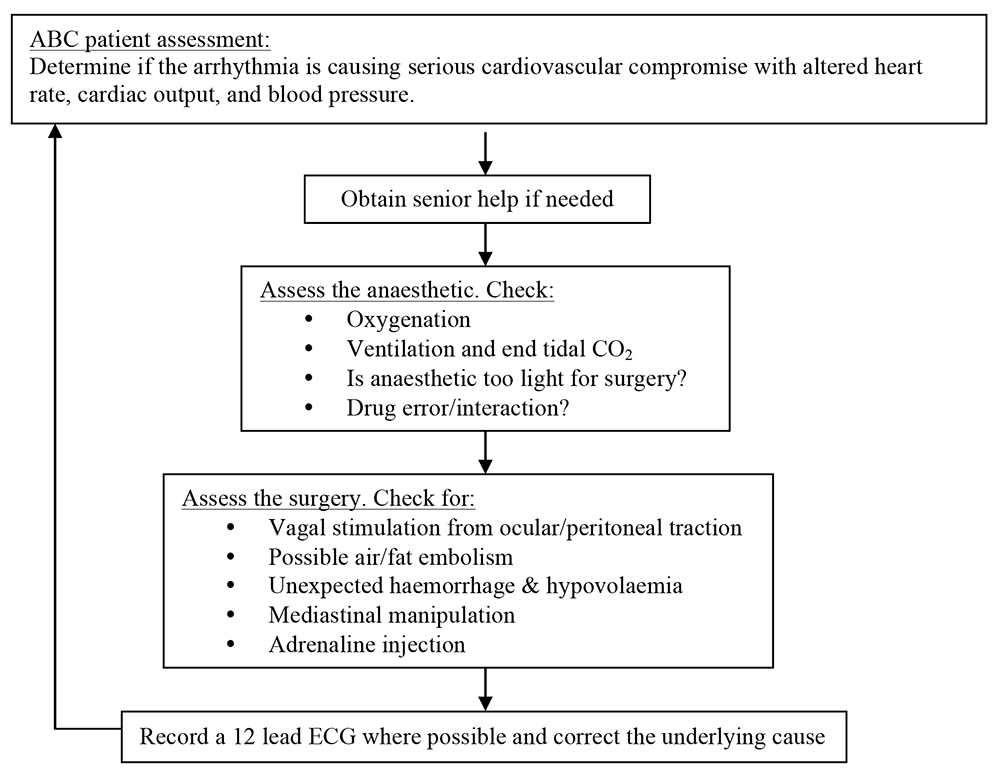
OVERVIEW OF INITIAL RHYTHM ASSESSMENT
The initial assessment of any arrhythmia is the same, with:
A NORMAL ECG AND THE MYOCARDIAL CONDUCTION PATHWAYS
Before discussing arrhythmias and their implications for the anaesthetist, it is worth revisiting a normal ECG and the myocardial conduction pathways. This will provide a point of reference to help recognise an arrhythmia and understand the origin of pathology of that arrhythmia.
The normal ECG consists of P, QRS and T wave deflections, which correspond to electrically-induced mechanical events during the cardiac cycle. When the Sino-Atrial (SA) node fires, electrical impulses are passed through intermodal tracts to the Atrio-Ventricular (AV) node. During this passage, atrial contraction occurs, illustrated by the P wave. Atrial repolarisation then follows and occurs at the same time as ventricular depolarisation, However, the atrial repolarisation wave is usually obscured by the larger QRS complex denoting ventricular depolarisation. The interval between atrial contraction and ventricular contraction is the PR interval, usually about 0.16s. However, this value varies according to heart rate.
Ventricular depolarisation occurs by passage of electrical impulses down specialised conducting fibres in the bundle of His to the ventricular apices. The impulses are then carried in the Purkinje fibres to ascend the ventricular walls, producing an ‘inferior to superior’ pattern of contraction. This allows efficient emptying of the ventricular contents into the pulmonary and systemic circulations. The amplitude of the waveform is dependent on multiple factors including myocardial mass, the cardiac axis, the anatomical orientation of the heart, and the distance between the ECG electrodes and the ventricles due to soft tissue mass. The normal QRS duration is <0.12s.
The ST segment and T wave illustrate ventricular repolarisation.
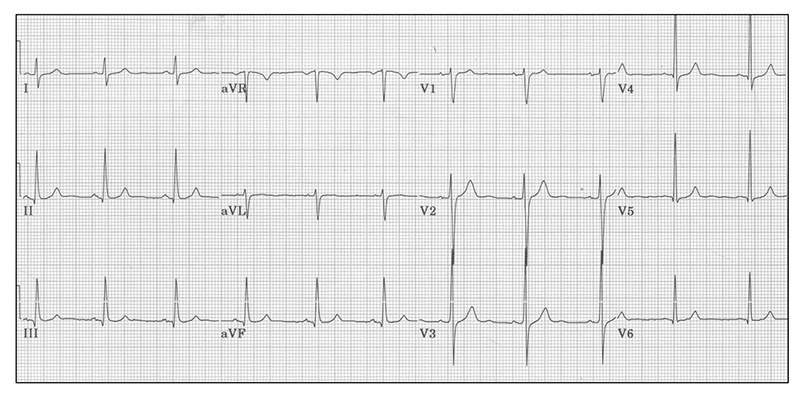
Figure 1. A normal 12-lead ECG, with P-QRS-T wave complexes occurring in regularity at a rate of 72bpm.
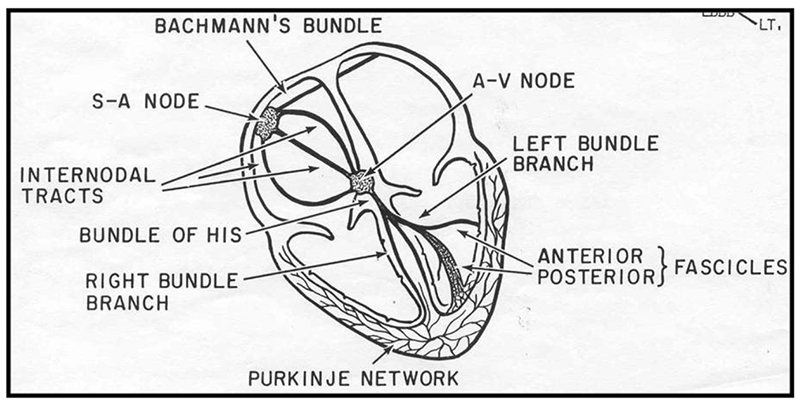
Figure 2. Myocardial conduction pathways
NARROW COMPLEX ARRHYTHMIAS
Sinus arrhythmia
A sinus arrhythmia (Fig 3) is of normal rate (>60bpm and <100bpm) with normal complexes and P-R intervals, but irregularly spaced R-R intervals. Sinus arrhythmia alters with respiration and can be a normal finding, especially in young patients.3 A sinus arrhythmia is usually of no concern peri-operatively.
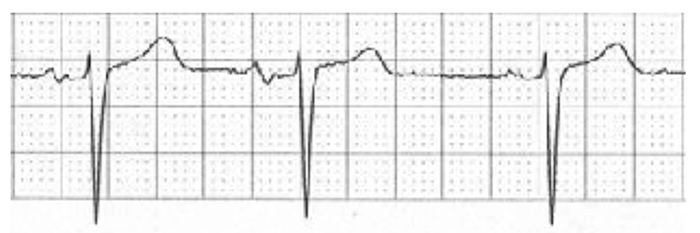
Figure 3. Sinus arrhythmia
Sinus bradycardia
Sinus bradycardia (Fig 4) is a sinus rhythm with normal P-QRS-T complexes and morphology, but a rate <60bpm. Bradycardia can often be normal finding in athletic patients or patients with high vagal tone, and it is seldom necessary to correct in fit patients until the rate is <40 beats per minute (bpm).
Other causes of sinus bradycardia include surgical factors such as vagal stimulation by anal or genito-cervical dilatation. Medical causes can be cardiac in origin (myocardial infarction, sick sinus syndrome), or non-cardiac in origin, such as hypothermia, raised intracranial pressure, or hypothyroidism. Finally, pharmaceutical causes can be either direct e.g beta-blockers or digoxin, or indirect effects from specific drug side effects of halothane or anticholinesterases such as neostigmine.
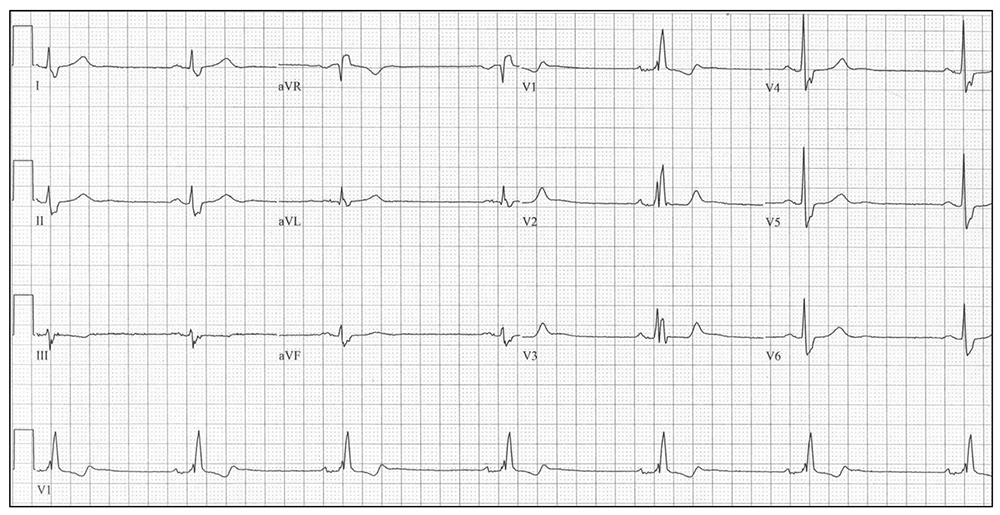
Figure 4. Sinus bradycardia (with right bundle branch block-discussed later).
Management must always be to correct any reversible causes first. Then consider atropine (up to 20μg/kg) or glycopyrronium (10μg/kg) by the intravenous route. If the bradycardia is resistant to the above and the patient is known to take beta-blockers, consider adrenaline or isoprenaline by infusion (0.5-10μg/min). If the bradycardia is entirely drug resistant, a pacemaker is required. If emergency surgery is indicated, the insertion of a temporary pacing wire will suffice, before formal pacemaker insertion.
Atrial ectopics
Atrial ectopics are usually common and benign. In figure 5 below, the red arrow indicates a premature atrial complex which is then conducted and followed by an atrial pause. However, before attributing these to a benign cause, other causes should be excluded. These include a side effect of the anaesthetic drugs used, light depth of anaesthesia, sepsis, shock, and ischaemia.
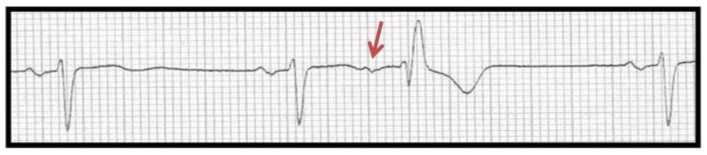
Figure 5. Atrial ectopic beat
The management of atrial ectopics is exclusion of correctable and serious causes, with modification of clinical management if necessary. Specific treatment is often unnecessary unless consistent runs of atrial tachycardias occur.
NARROW COMPLEX TACHYCARDIAS
Narrow complex tachycardias are defined as a regular rhythm with a QRS complex <0.12s with a rate >100bpm.
The differential diagnosis for a narrow complex tachycardia are listed below:
- Sinus tachycardia
- Supraventricular tachycardias (SVT) such as atrial flutter, atrial tachycardia, multifocal atrial tachycardia and junctional tachycardias
- Paroxysmal supraventricular tachycardia (PSVT)
- Atrial fibrillation (AF)
- Sick sinus syndrome
- Wolff-Parkinson-White syndrome
Some/all of the above may cause severe cardiovascular compromise with circulatory disturbance. The ECG will show a narrow complex tachycardia. However, if bundle branch block or anterograde conduction down an accessory pathway exists, a broad complex (>0.12s) will be seen on the ECG.
Sinus tachycardia
Sinus tachycardia (Fig 6) is a sinus rhythm with normal P-QRS-T complexes and morphology, but with a rate >100bpm.
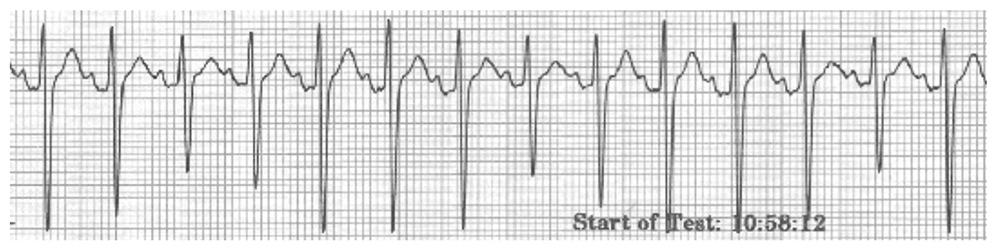
Figure 6. Sinus tachycardia
Causes can be multi-factorial. Operative causes are pain, surgical stimulation and light depth of anaesthesia. Pharmacological factors include administration of catecholamines, atropine, or ketamine. Medical factors such as sepsis, hypovolaemia, heart failure, anaemia, and thyrotoxicosis should also be considered.
The management process should consider and correct the precipitating cause.
Supraventricular and junctional tachycardia
A supraventricular tachycardia (SVT) is any tachycardia originating from above the ventricles. The nature of a SVT depends on the origin of the electrical impulse. Impulses from the sino-atrial node can be sinus tachycardias, sino-atrial re-entrant tachycardias and atrial flutter. However, impulses from the atrial myocardium itself can be ectopic unifocal tachycardias, multifocal atrial tachycardias, and atrial flutter or fibrillation. Atrioventricular (junctional) electrical sources can be defined as AV re-entrant tachycardias, atrioventricular reciprocating tachycardia, and junctional ectopic tachycardias.
ECG features of an atrial tachycardia are of abnormally shaped P waves, often outnumbering QRS complexes. In contrast, the features of a multifocal atrial tachycardia are of 3 or more P wave morphologies with irregular QRS complexes, as shown in figure 7 below.
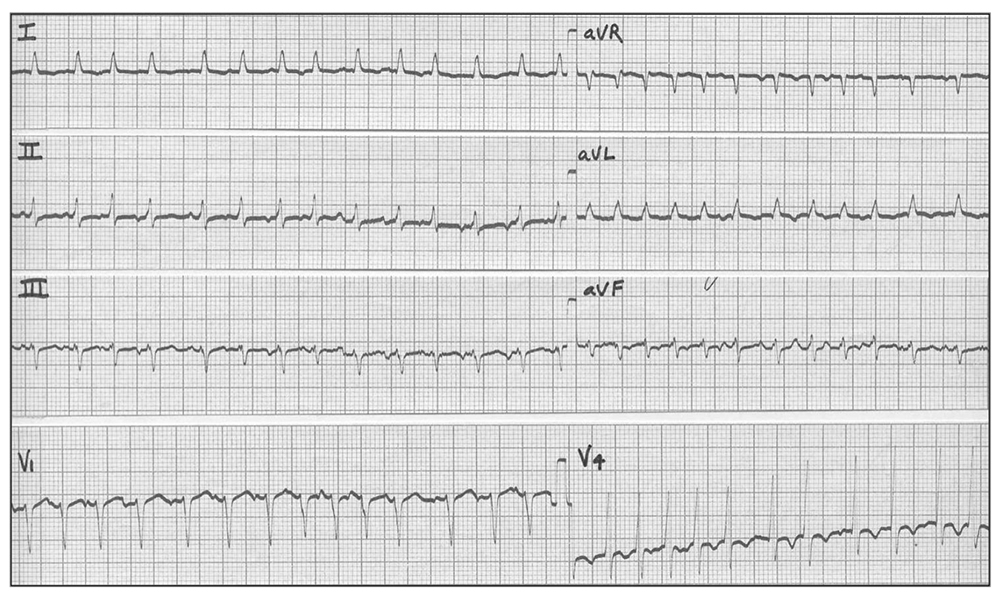
Figure 7. Multifocal atrial tachycardia
Junctional tachycardias however often have a rate of 150-200bpm with P waves either buried or closely following the QRS complex, as shown in figure 8 below.
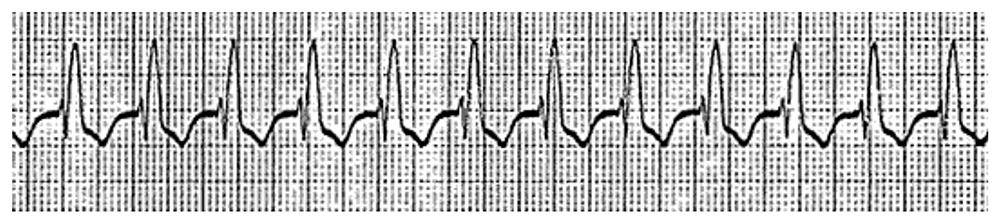
Figure 8. Junctional tachycardia
Management for the common SVTs has been grouped according to their similar management strategies. For the more uncommon arrhythmias, discussion with a senior anaesthetist with or without cardiology input is paramount. These SVTs must be differentiated from the potentially more perilous ventricular tachycardias originating from ventricular myocardium (discussed later).
Management of Junctional/AV node/SVTs
If the patient is anaesthetised and demonstrates haemodynamic instability, the immediate management is synchronised DC cardioversion with 200J, increased to two shocks at 360J if refractory. In the non-anaesthetised patient, sedation with a benzodiazepine and synchronised DC cardioversion is needed.
If the patient is haemodynamically stable, the initial management should include carotid sinus massage. This activates the baroreceptors in the carotid sinus within the internal carotid artery, resulting in increased vagal activity and transient AV block. This aids differentiation between SVT, atrial flutter and AF. The valsalva manoeuvre can also be used. If this fails, the following pharmacological therapies should be considered:
- Adenosine 6mg IV bolus, given as a fast push and followed with a 20ml 0.9% saline flush. This rapidly blocks AV node conduction therefore slows the ventricular rate, cardioverting a junctional rhythm to sinus or terminating a re-entry SVT. If a second and third dose is needed, 12mg should be used with an interval period of at least 1 minute between doses. The effects only last for 10-15s. Ensure recording of an ECG rhythm strip during administration. Ensure the patient is aware of the common side effects of dizziness, chest tightness, dsypnoea, headache and flushing. Adenosine is contraindicated in patients taking dipyridamole due to the potentiation side effects and risk of severe myocardial depression. The use of adenosine in asthmatics is a relative contrainidcation due to the potential side effect of bronchospasm, and must only be used with extreme caution.
- Beta-blockers e.g. esmolol 50-200ug/Kg/min IV, or metoprolol 3-5mg IV over 10 minutes every 6 hours.
- Verapamil 5-10mg IV over 2 minutes, with a second dose of 5mg after 10 minutes if needed. Useful with SVT who relapse post-adenosine. Do not use with beta-blockers.
- Amiodarone 300mg IV over 1hour via a central line should be considered when the above interventions have failed.
- Digoxin should be avoided due to its facilitation of AV accessory conduction in Wolff-Parkinson-White syndrome and can worsen tachycardia. AF with an anatomical accessory pathway with rapid conduction can degenerate into ventricular fibrillation.
Atrial flutter
Atrial flutter is due to electrical impulse re-entry into the atria, often giving an atrial rate of 300bpm, with a ventricular rate of approximately 150bpm. This is illustrated in figure 9 below and is known as atrial flutter with 2:1 block, with every second flutter wave passing through the AV node. Also note the ‘flutter/sawtooth waves’ where this arrhythmia derives its name. A 3:1 block may also be encountered but is less common.
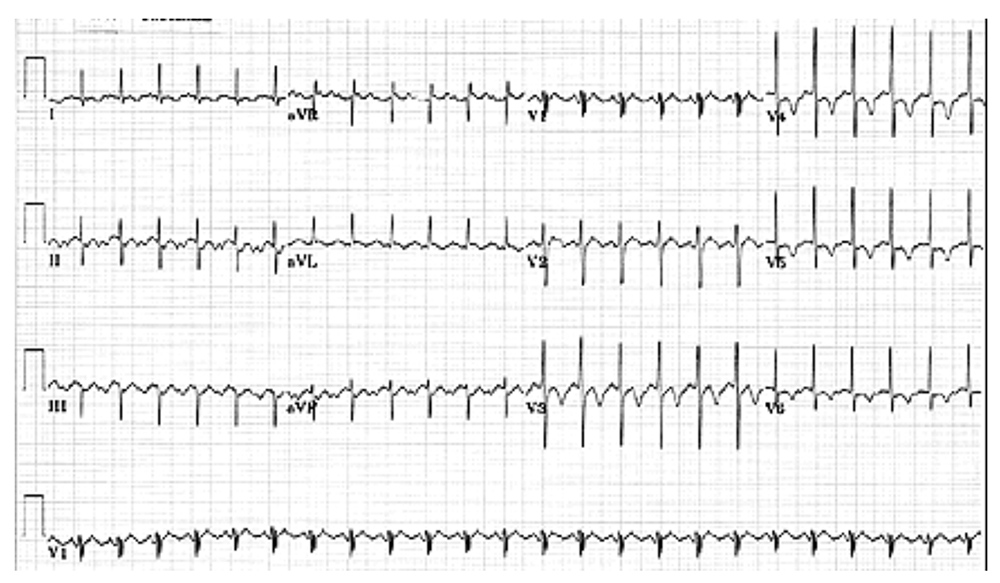
Figure 9. Atrial flutter
In atrial flutter and atrial tachycardia, the atria contract at >150bpm due to an ectopic focus, causing Pwaves to be superimposed on T waves from preceding beats. Both atrial flutter and atrial tachycardia can occur with any kind of AV block (e.g. 2:1, 3:1 etc) and carry the same thromboembolic risk as atrial fibrillation.
Management of atrial flutter/atrial tachycardia
If the onset is <48hours in patients with haemodynamic instability who are anaesthetised or appropriately sedated, synchronised DC cardioversion will convert the rhythm back to sinus in nearly all cases.
If the patient is not anaesthetised and sedation is not appropriate, carotid sinus massage, followed by the valsalva manoeuvre are initially indicated. If both fail to revert to a sinus rhythm, adenosine 0.2mg/Kg IV may be administered to slow AV conduction to reveal the underlying rhythm and any AV block. Further management is the same as acute AF (below).
Atrial fibrillation
Atrial fibrillation (AF) is independent and uncoordinated atrial electrical activity, with a ventricular rate dependant on the often-intermittent AV node transmission, usually at a rate of 120-180bpm.
Atrial contraction contributes 30-40% of normal ventricular filling. Therefore, new onset AF, especially fast AF (>90bpm) causes a marked reduction in ventricular filling, cardiac output, and haemodynamic status. Ischaemia then often follows due to reduced diastolic time and hypotension.
The clinical features of AF are of an irregularly irregular pulse, with no ‘a’ waves or ‘x’ descent seen in the jugular venous pulse. This is due to the lack of atrial relaxation. On auscultation, there is first heart sound variance and a discrepancy between apical pulse and radial pulse.
The ECG changes (figure 10 below) are of absent P waves with irregularly irregular QRS complexes, often with a wondering baseline, unless the AF is chronic in nature. Bundle branch block maybe seen in some complexes, but this can vary.
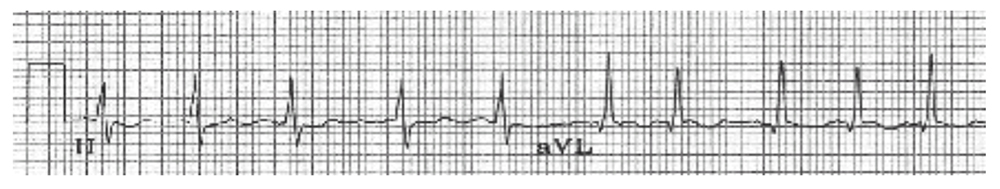
Figure 10. Atrial fibrillation
There are multiple causes of AF, some more likely than others depending on the patient. The most common cause is myocardial ischaemia. Other causes are sepsis (especially pneumonia), electrolyte abnormality (especially hypokalaemia or hypomagnesaemia), hypertension, heart failure, thyroxtoxicosis, alcohol, mitral valve disease, pericarditis/mediastinitis, and thoracic surgery.
The management of AF differs depending on whether or not it began within 48 hours. This is due to the risk of atrial clot formation in AF sustained for longer than a 48 hour period.
AF management if onset <48 hours
- Correct any correctable causes e.g. electrolyte disturbance. In AF secondary to sepsis or surgery, sinus rhythm will often be restored once the underlying cause is controlled.
- Amiodarone 300mg IV over 1 hour via a central line (or large peripheral line in emergencies but beware risks of extravasation). Follow with 900mg over 23 hours. Used for rate control and cardioversion. Can be combined with beta-blockers or digoxin and is useful in new AF associated with critical illness, and well tolerated with LV impairment. Beware multiple side effects including pulmonary fibrosis.
- Beta-blockers (esmolol, sotolol, or metoprolol 5mg IV) slow ventricular rate and are useful before or in theatre whilst waiting for other drugs to take effect. Beta-blockers will slow any tachycardia whilst also chemically cardioverting back to sinus rhythm. However, they have a marked hypotensive effect therefore should only be used in patients with an adequate blood pressure. Caution must be used in patients with an impaired myocardium, thyrotoxicosis and asthma. Avoid use with calcium-channel blockers.
- Digoxin 500ug IV over 20 minutes, repeated in 4-8 hours. Max 1-1.5mg. Digoxin has a narrow therapeutic index and needs dose reduction in patients with acute kidney injury. Ensure normal serum potassium levels, otherwise risk of side effects increased as it competes with potassium for the Na+/K+ ATPase pump. Digoxin is used for rate control only and will not cardiovert the arrhythmia back to sinus rhythm.
- Flecainide 2mg/Kg (max 150mg) IV over 30minutes with cardiac monitoring is the best drug for cardioversion back to sinus. However, it cannot be used if there is structural/ischaemic heart disease.
- If the patient is haemodynamically unstable, synchronised DC cardioversion should be considered at 200J, increased to 360J if necessary for a further two shocks. Synchronisation ensures the shock is not delivered on a T wave, which avoids the risk of precipitating ventricular fibrillation. It is successful in <20% of cases.
AF management if onset > 48 hours
Patients suffering AF for > 48 hours have a high risk of atrial blood clots forming and embolising systemically, causing a cerebrovascular event (CVE). Due to this raised thromboembolic risk, patients should be anticoagulated for at least 3 weeks before ventricular rate control to <100bpm. Oral anticoagulation brings the risk of CVE down to 2%/year for patients aged 75.
- Digoxin for rate control. Load with 0.5mg/12hours for 24 hours, followed by maintenance doses of 0.125-0.25mg daily. Dosages may need reducing in the elderly.
- Beta-blockers (as above)
- Amiodarone (as above)
- Verapamil 5-10mg IV is useful in patients unable to tolerate beta-blockers e.g. asthmatics. Avoid if evidence of ischaemia, impaired LV function, or in combination with beta-blockers.
- Anticoagulate with warfarin unless contraindicated.
Uncontrolled chronic AF management
These patients need pre-operative rate control to keep ventricular rate <100bpm. This heart rate allows time for ventricular filling and myocardial perfusion.
- Digoxin loading 1-2 days preoperatively unless already digitalised. This can be achieved with oral preparations but may need to be IV if urgent surgery is indicated. Check serum digoxin levels (0.8-2.0μg/L)
- Additional beta-blocker (e.g. metoprolol, atenolol) or verapamil if blood pressure and good LV function exist.
- If poor LV function, amiodarone IV.
- If haemodynamic compromise is present and anticoagulation is not appropriate (such as in the immediate postoperative period after major surgery), synchronised DC cardioversion under sedation/anaesthesia must be administered as soon as possible.
Sick sinus syndrome
Sick sinus syndrome (SSS) can be familial, congenital, rheumatic, ischaemic, and/or hypertensive in aetiology. The sinus node dysfunction usually progresses gradually, and presentation is normally due to non-specific symptoms secondary to vital organ hypoperfusion. These include dizzy spells, syncope, fatigue, memory impairment, paresis, and pulmonary oedema.
Severe bradycardia resistant to atropine is a hallmark of SSS and cases of SSS have been uncovered with general anaesthesia (with both IV and volatile agents used at induction) and epidural anaesthesia, often with associated cardiovascular collapse.
The ECG appearances are of sinus bradycardia, alternating sinus bradycardia with tachycardia (tachy-brady syndrome), sino-atrial block, atrial flutter or AF. A permanent pacemaker, either atrial or dual chamber is required if symptomatic or suffering tachy-brady syndrome.
Wolff-Parkinson-White syndrome
Wolff-Parkinson-White (WPW) syndrome is an example of a re-entry arrhythmia where an accessory conduction pathway exists between the atria and ventricles. All re-entry arrhythmias are due to an extra conduction pathway that branches and then rejoins the main conduction pathway. If the impulse arrives when the main pathway is no longer refractory, the impulse passes around the circuit repeatedly activating it.
WPW syndrome occurs in 1-3/1000 of the general population, although fewer than 25% of those suffer episodes of sustained tachycardia. The pathology is of ventricular pre-excitation in sinus rhythm with recurrent SVTs, due to the accessory pathway. A shortened PR interval with a premature but slurred R wave, the “delta” wave, is seen on the ECG. Two main types are recognised: Type A is a left ventricular pathway giving a positive delta wave in lead V1, and type B is a right ventricular pathway giving a negative delta wave in V1. In addition, repolarisation abnormalities resembling ischemia may also be present and false positive exercise tests are common.
The ECG in figure 11 below highlights the shortened PR interval and delta waves (V1-V5 leads are the best illustrations). Pseudo Q-waves in II, III, and aVF are negative delta waves.
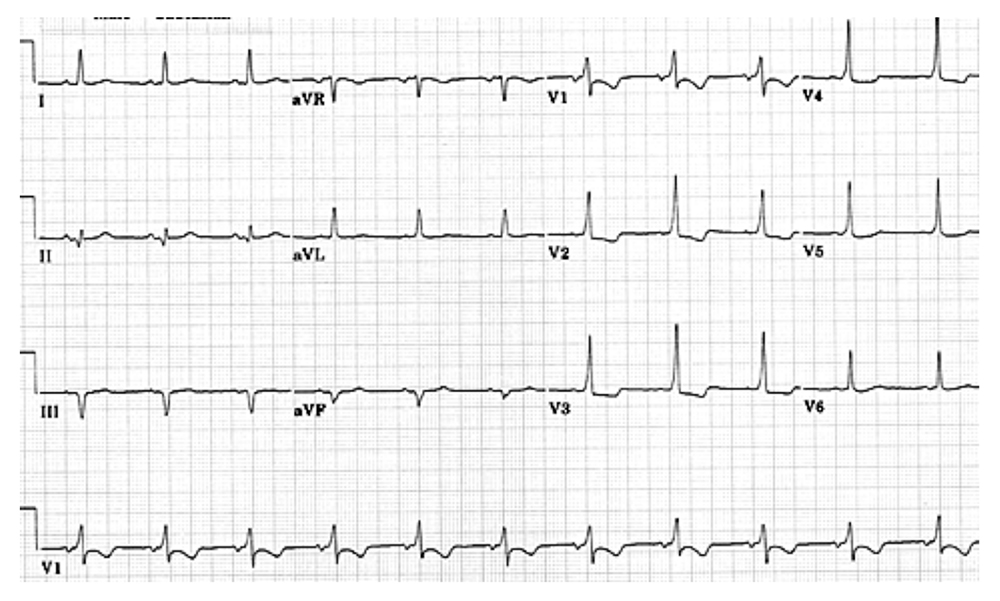
Figure 11. Wolff-Parkinson-White Syndrome
The ECG in WPW syndrome is therefore a blend of normally and abnormally activated myocardium. The ECG may mimic right bundle branch block, right ventricular hypertrophy or true posterior infarction in type A WPW. The ECG in type B may imitate left bundle branch block.
The management of WPW induced tachycardias consists of vagal manoeuvres and pharmaceutical agents such as flecainide 50-150mg IV, propanolol 1-10mg IV, disopyramide 50-150mg IV, or ajmaline 50-100mg IV. If cardiovascular instability occurs, DC cardioversion is indicated. Chronic control and prophylaxis usually requires combination therapy e.g. beta-blockade and amiodarone, disopyramide and beta-blockade. Ablation therapy is often definitive. Digoxin and verapamil can accelerate conduction down specific accessory pathways and are therefore best avoided.
ANTI-ARRHYTHMIC DRUGS
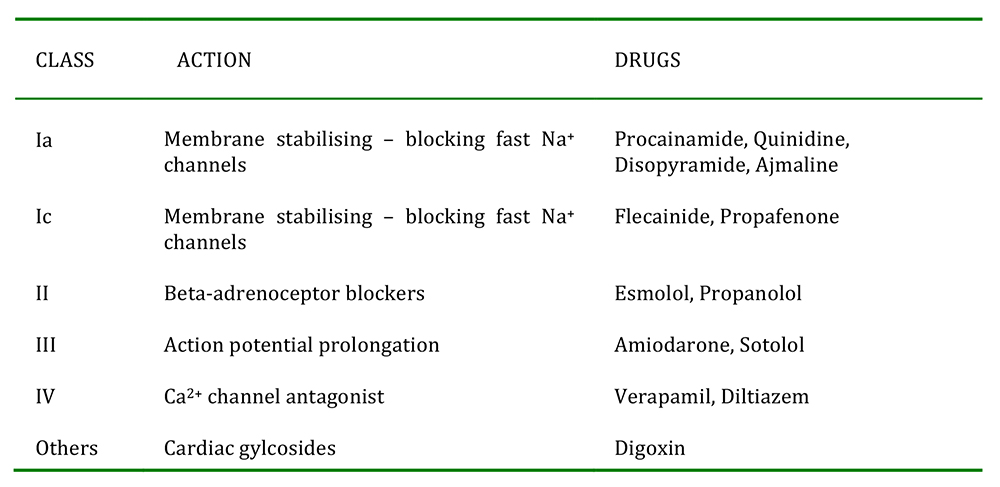
Table 1. Anti-arrhythmic
SUMMARY
- Dysrhythmias can occur in the perioperative period and may have harmful or life threatening effects on a patient.
- Assessing the ECG will usually allow the dysrhythmia to be diagnosed, and an appropriate treatment protocol commenced.
- Antiarrhythmic drugs have potentially severe side effects. Side effects should be considered when selecting medications suitable for a specific patient.
ANSWERS
- The myocardial conduction pathways are illustrated in figure 2.
- The causes of a sinus tachycardia can be multi-factorial. Operative causes are pain, surgical stimulation or light depth of anaesthesia. Pharmacological factors include administration of catecholamines, atropine, or ketamine. Medical causes are sepsis, hypovolaemia, heart failure, anaemia, and thyrotoxicosis.
- Examples of Class III anti-arrhythmic drugs include amiodarone and sotolol.
REFERENCES and FURTHER READING
- Valchanov K, Webb ST, Sturgess J. Anaesthetic and Perioperative Complications. Cambridge University Press, UK. 2011.
- Aitkenhead AR, Rowbotham DJ, Smith G. Textbook of Anaesthesia, 4th Ed. Churchill Livingstone, UK. 2001.
- Allman KG, Wilson IH. Oxford Handbook of Anaesthesia, 3rd Ed. Oxford University Press, UK. 2011.
- World Federation of Societies of Anaesthesiologists. Cardiovascular responses to anaesthesia. Update in Anaesthesia. Issue 10 (1999). Article 2: Pp4.
- Staikou C, Chondrogiannis K, Mani A. Perioperative management of hereditary arrhythmogenic syndromes. Br J Anaesth 108 (5): 730-44 (2012).
- Nathanson G, Gajraj NM. Perioperative management of atrial fibrillation. Anaesthesia, 53, 665-676 (1998).
- Vaughan Williams E. Classification of antiarrhythmic drugs. In: Sandoe E, Flensted-Jensen E, Olesen K, Eds. Cardiac Arrhythmias. Sodertaljie, Sweden. AB Astra, 1971: 449-72.
- Allman KG, McIndoe AK, Wilson IH. Emergencies in Anaesthesia. Oxford University Press, UK. 2005.
- Advanced Life Support algorithm. Resuscitation Council (UK). 2010. http://www.resus.org.uk/pages/als.pdf
- Tachycardia algorithm. Resuscitation Council (UK). 2010. http://www.resus.org.uk/pages/tachalgo.pdf 11) Miller RD. Miller’s Anaesthesia, 7th Ed. Churchill Livingstone. Philapedphia, USA. 2010.
- Pollard BJ. Handbook of Clinical Anaesthesia, 3rd Ed. Hodder Arnold. London, UK. 2011.
- Swanton RH. Pocket Consultant Cardiology, 5th Ed. Blackwell. Oxford, UK. 2003.
- Tse HF, Lip GYH, Stewart Coats AJ. Oxford desk reference Cardiology. Oxford University Press. Oxford, UK. 2011.
- Longmore M, Wilkinson I, Rajagopalan S. Oxford handbook of clinical medicine, 6th Ed. Oxford University Press, UK. 2006.
- Da Costa D, Brady WJ, Edhouse J. ABC of clinical electrocardiography: Bradycardias and atrioventricular conduction block. BMJ 2002;324;535-538.
- Anaesthesia UK. www.frca.co.uk 2012.
- Australian resuscitation council guidelines http://www.resus.org.au/policy/guidelines/section_11/managing_acute_dysrhythmias.htm
- EB Medicine. An evidence based approach to supraventricular tachydysrhythmias. 2013. http://www.ebmedicine.net/topics.php?paction=showTopicSeg&topic_id=133&seg_id=2668
Image references
- Yanowitz FG. ECG Learning Centre. University of Utah School of Medicine. Accessed at:
http://ecg.utah.edu/ - Anaesthesia UK. www.frca.co.uk. 2012.
- Jenkins D. Gerred S. ECG library. Trifasicular heart block ECG only. 2009. Accessed at
http://www.ecglibrary.com/ecghome.html



