General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- What are the causes of intraoperative ventricular tachycardia?
- What is the management of clinically stable ventricular tachycardia?
- Describe the ECG findings in right bundle branch block.
INTRODUCTION
This is the second of a two part tutorial. Tutorial one focussed on a basic introduction to the ECG as well as assessment and management of narrow complex arrhythmias and tachycardias.
This second tutorial will focus on the following:
- Broad complex arrhythmias
a. Ventricular ectopics
b. Ventricular tachycardia
c. Supraventricular tachycardia with aberrant conduction
d. Ventricular fibrillation
e. Torsade des pointes - Disturbances of conduction
a. Bundle branch block
b. Right bundle branch block
c. Left bundle branch block
d. First degree heart block
e. Second degree heart block
f. Third degree heart block/complete block
g. Bifasicular block
h. Trifasicular block
BROAD COMPLEX ARRHYTHMIAS
Ventricular ectopics
Ventricular ectopics are usually benign in the absence of structural heart disease. In young fit patients, normalising cardiovascular and respiratory variables or manipulating the depth of anaesthesia is often all that is needed to terminate ventricular ectopic beats. However, they can sometimes precede runs of ventricular tachycardia.
The ECG appearances are of occasional broad QRS complexes as shown in figure 13 below.
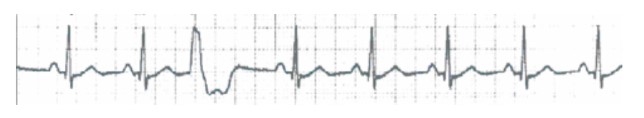
Figure 13. Ventricular ectopic beat
Ventricular ectopics are usually due to a metabolic imbalance, especially hypokalaemia or hypercarbia. Other documented causes include dental surgery, anal stretch, the use of halothane, and light depth of anaesthesia.
The management of ventricular ectopics focuses on correction of obvious causes. Small doses of IV (intravenous) beta-blocker such as metoprolol 3-5mg may be used once other factors have been normalised. If the patient has a bradycardia under 50 beats per minute (bpm), ectopics may in fact be ventricular escape beats. In this instance, a trial of an anticholinergic such as atropine or glycopyrronium to increase ventricular rate, can be used.
Ventricular tachycardia
Ventricular tachycardia (VT) is caused by high frequency electrical depolarisation from a central focus within the ventricular muscle causing a fast (>100bpm) widened QRS (>0.12s) of variable shape (fig 14). If atrioventricular dissociation is also present, p waves may be seen, along with capture/fusion beats, implying VT rather than SVT with aberrant conduction. This is a severe potentially life-threatening arrhythmia, which needs urgent diagnosis and management.
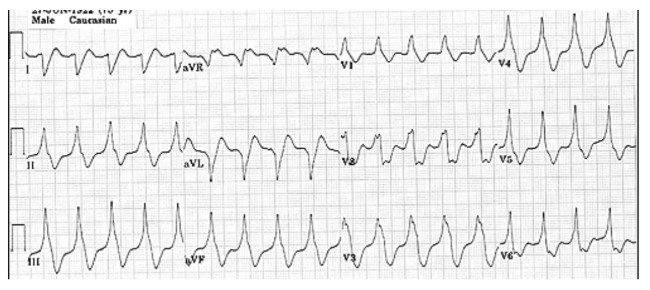
Figure 14. Ventricular tachycardia
The intra-operative causes of VT are varied. As always, electrolyte imbalance (particularly hypokalaemia and hypomagnesaemia) must be excluded. Other causes include myocardial ischaemia, hypoxia, hypotension, fluid overload, and the IV use of adrenaline and other chatecholamines.
The management of VT differs depending on the stability of the patient. A patient is unstable if there is a systolic blood pressure of <90 mmHg, a tachycardia >150 bpm, heart failure, syncope, or evidence of myocardial ischaemia. If the patient has these signs and has a pulse, synchronised DC (direct current) cardioversion at 150J (joules) rising to 360J biphasic is required. If no central pulses can be found, pulseless VT is a cardiac arrest rhythm and immediate cardiopulmonary resuscitation using the Advanced Life Support algorithm with DC cardioversion is a priority (see VF/pulseless VT management below).
If the patient is haemodynamically stable, correct any correctible cause such as an electrolyte imbalance immediately. If the patient takes a diuretic regularly, assume hypomagnesaemia is present and replace with 2g IV magnesium. This helps to stabilise myocardial cell membranes. Obtaining an ECG before and after correction will assist with the diagnosis retrospectively. There are multiple treatment options available. The Resuscitation Council (UK) recommends amiodarone 300mg IV over 1 hour via a central line (or large peripheral line in emergencies keeping in mind the risks of extravasation), followed by 900mg over 23 hours. If there is previously confirmed SVT with a bundle branch block, adenosine is recommended (see tutorial one).
Other treatment options are listed below:
- A lignocaine bolus of 100mg followed by a maintenance infusion of 4mg/min for 30minutes, 2mg/min for 2hours, then 1mg/min will chemically cardiovert back to sinus rhythm in 30-40% of cases. If lignocaine fails, consider:
- Sotolol 100mg IV over 5minutes
- Procainamide 100mg IV over 5minutes followed by a second or third bolus before beginning an infusion at 3mg/min.
- Electrical cardioversion as above.
The ongoing management of VT focuses on ensuring all metabolic abnormalities are corrected and reviewing usual medications to stop any possible precipitants of a prolonged QT interval. If VT persists, immediate cardiology referral is indicated for alternative anti-arrhythmic drugs or overdrive pacing.
Supraventricular tachycardia (SVT) with aberrant conduction
This occurs when a SVT exists with a broad complex ECG morphology due to aberrant conduction between atria and ventricles. It is usually caused by rate related ischaemia due to tachycardia.
SVT caused by an abnormal or accessory pathway e.g. Wolff-Parkinson-White syndrome, will be narrow complex if there is retrograde conduction (i.e. the normal pathway initiates the QRS). However, a broad complex will be seen if there is anterograde conduction in the accessory pathway.
Due to significant differences in management, it is very important to distinguish SVT with abberant conduction from VT. This can be difficult. Often, a detailed medical history will expose a history of heart disease (ischaemic, cardiomyopathy, congenital, or long QT syndrome) in VT. The absence of any previous structural heart disease makes SVT a more likely diagnosis. Other features of VT include a QRS morphology not consistent with a classic bundle branch block, and the existance of capture and fusion complexes which although rare (<7% prevelance) are highly specific.
All broad complex tachycardias should be treated as VT until proven otherwise (see above). Adenosine 0.2mg/Kg IV rapidly blocks AV node conduction therefore slows ventricular rate. The effects only last for 10-15s. Ensure recording of an ECG rhythm strip. In the case of an underlying SVT, the rhythm may revert to sinus.
Ventricular fibrillation
Ventricular fibrillation (VF) is characterised by fast, chaotic, irregular and disorganised broad complexes on the ECG (figure 15). There will be no palpable peripheral or central pulses due to the lack of cardiac output.
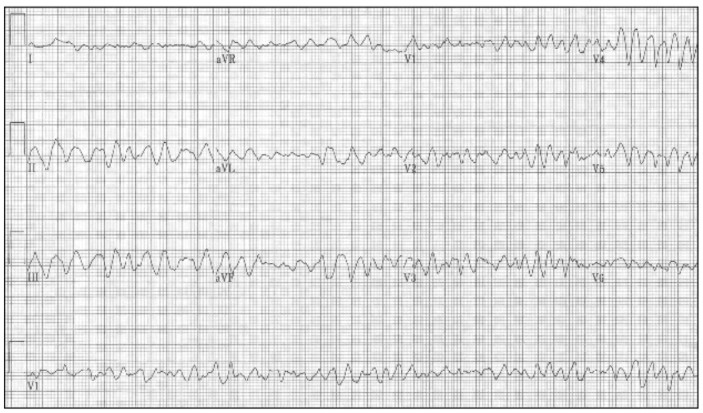
Figure 15. Ventricular fibrillation
Risk factors for VF include recent myocardial infarction, pre-existing ischaemic heart disease, hypokalaemia, excessive endogenous or exogenous chatecholamine levels, and myocardial irritation e.g. from a guidewire during the insertion of a central venous catheter.
Important exclusions
- Polymorphic VT – although in the absence of a pulse, management is still the same as VF (see below)
- AF with an atrio-ventricular accessory pathway
- ECG artefact secondary to surgical diathermy or patient movement
Treatment of VF
Various guidelines exist around the world to direct management of VF. However, the basic principles of effective chest compressions with early electrical defibrillation are consistent across the globe. The UK Resuscitation Council recommends immediately calling for help and non-synchronised DC cardioversion with an initial biphasic shock of 150-200J, followed by 150-360J for subsequent shocks. Each shock should be followed with 2 minutes of cardio-pulmonary resuscitation whilst correcting possible causes. Electrical defibrillation must occur as soon as possible due to the high success rate if the first shock is delivered within 3 minutes. The role of the precordial thump is being de-emphasised.
In either VF or pulseless VT occurring in the cardiac catheter lab or immediately post cardiac surgery, the use of up to three consecutive “stacked” shocks is recommended by the Resuscitation Council (UK).
If no defibrillator can be found immediately, chest compressions and ventilation with 100% oxygen at a 30:2 ratio must occur. The compressions should be at a speed of 100/min and intubation should occur during this time. Administer adrenaline 1mg 1:10,000 IV/IO every 3-5 minutes beginning after the third shock (during alternate cycles of cardio-pulmonary resuscitation).
Note: the management outlined for VF is the same for pulseless VT (see Resuscitation Council (UK) Advanced Life Support guidelines below). Regarding paediatric patients, the management principles remain the same. However, refer to Advanced Paediatric Life Support guidelines for appropriate ratios, drug doses and shock energies
Management algorithm for VF/pulseless VT (adapted from the ALS guidelines 2010, found at: http://www.resus.org.uk/pages/als.pdf) Please consult for full algorithm detail.
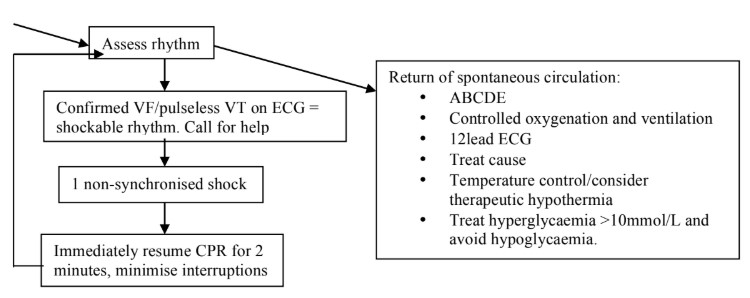
In resistant VF or pulseless VT, treat and normalise any reversible causes (below) and consider urgent cardiology advice. If VF/pulseless VT persists following 3 shocks, amiodarone 300mg IV though large peripheral/central access can be given. If in an intra-operative environment and resuscitation is successful, complete surgery as soon as possible (abandon electice surgical procedures). Then transfer the patient intubated and sedated, to the intensive care unit.

Table 1. Reversible causes of cardiac arrest
Torsade des pointes
Torsade des pointes (“twisting of the points”) is an atypical polymorphic form of VT characterised by beat-to- beat variation, prolonged QT interval, and a constantly changing/twisting QRS axis around the baseline (figure 16). A severe non-uniform delay in repolarisation, illustrated by QT prolongation, is the primary electrophysiological derangement. It begins with a distinguishing long-short initiating sequence.
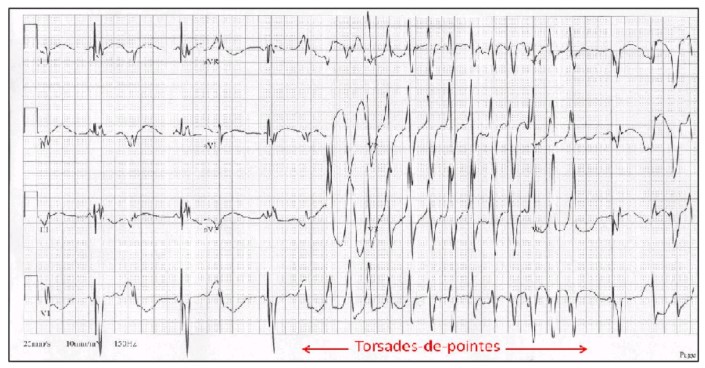
Figure 16. Torsades des pointes
It can be induced by the following causes:
- Metabolic: Hypokalaemia, hypomagnesaemia
- Drugs: Phenothiazine, procainamide, quinidine, dispyramide
- Cardiac: Bradycardia, acute ischemia, infarction.
Closely related to torsades is polymorphic ventricular tachycardia (PVT) with a normal QT interval. With PVT, there is no prolonged repolarisation, giving an ECG with a QT interval within normal limits. The management of torsade and PVT differs; consequently early diagnosis with 12-lead ECG is necessary.
The management of torsades is initially aimed at correcting the underlying cause (see above) and some clinicians advocate a bolus of 2g magnesium sulphate as emergency cellular membrane stabilising treatment. Further therapy preferentially includes overdrive atrial pacing if AV conduction is intact, before ventricular pacing. The management of PVT is the same as the management of VT with a pulse.
DISTURBANCES OF CONDUCTION
This final section includes information on the bundle branch blocks and conduction blocks.
Bundle branch block
Bundle branch block, either right or left, is associated with coronary disease and risks progression to complete heart block. This is especially true when considered with the possible anaesthetic drugs administered, electrolyte disturbances, intubation, hypothermia, surgical manipulation, and possible myocardial ischaemia. Other known causes of atrioventricular conduction block are immunological causes such as systemic lupus erythematous, and infections causes such as Lyme disease and diptheria.
Bundle branch block is illustrated by delayed depolarisation of the particular branch (left or right), resulting in delayed conduction and subsequently delayed contraction of that region of ventricular muscle.
Right bundle branch block
Often benign and found in 1% of all adult inpatients, blockage of the right bundle results in delayed right ventricular depolarisation because the usual right-to-left depolarisation pathway is damaged. The left ventricle depolarises normally, and then spreads to non-specialised conducting tissue to depolarise the right ventricle in a left-to-right manner.
The ECG shows wide QRS complexes (>120ms) with an “R-S-R pattern” or “M” shape in leads V1-V3. In V5, V6 and I, the opposite is present often with a “W” shape formed by a slurred S-wave (figure 17). In the right precordial leads, ST-segment depression and T-wave inversion may also be seen.
New onset right bundle branch block in patients with pre-existing heart disease does predict a higher rate of coronary artery disease, congestive cardiac failure and mortality. However, there are no specific anaesthetic concerns for these patients.
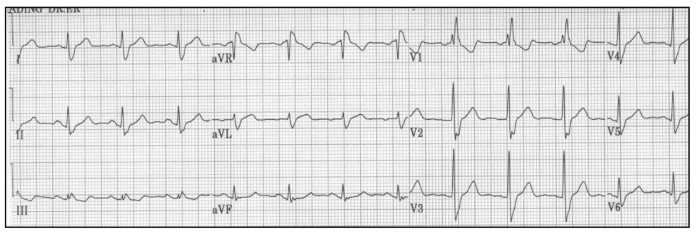
Figure 17. Right bundle branch block
Left bundle branch block
The left bundle branch consists of the large posterior fascicle and a smaller anterior fascicle (refer to figure 2, tutorial one). Left bundle branch block occurs when there is reversal of septal depolarisation (right-to-left instead of left-to-right) due to coronary obstruction, causing a change in the initial direction of the QRS complex on the ECG. The QRS complex is prolonged due to the depolarisation delay and the septal Q waves are lost and replaced with R waves.
Blockage of the anterior fascicle causes delayed activation of the anteriosuperior ventricular wall, with axis deviation of >60° to the left on the ECG. A posterior fasicular block is less common and results in delayed activation of the inferioposterior left ventricle. A right axis deviation >120° is found on the ECG.
Common ECG features to both types of left bundle branch block are QRS prolongation >0.12s, monophasic broad R waves in leads V5, V6, and I with poor R wave progression, and an absence of Q waves in V5 and V6. Depression of the S-T segments with T-wave inversion is also commonly found due to abnormal myocardial repolarisation.
Both hemiblocks (caused by anterior or posterior fascicle obstruction) are associated with coronary artery disease and patients need a thorough cardiovascular evaluation pre-operatively.
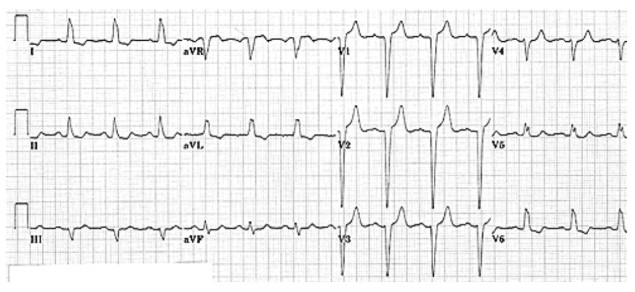
Figure 18. Left bundle branch block
Left bundle branch block makes further interpretation of the ECG except rate and rhythm very difficult. A left bundle branch block of new onset illustrates an evolving myocardial infarction until proven otherwise and urgent discussion with a cardiologist is essential, with management by cardiac catheterisation and angioplasty a possibility.
Heart blocks
First-degree conduction / ‘heart block’
First-degree block is caused by a conduction delay between the SA node and the ventricles, resulting in P-R prolongation to >0.2s (figure 19). It is common and if the patient is asymptomatic, does not need any specific investigation or treatment. It is usually not a problem itself during anaesthesia but can rarely progress to second-degree block, usually Mobitz type 1. If however, the patient is symptomatic, they will need pre-operative pacing.
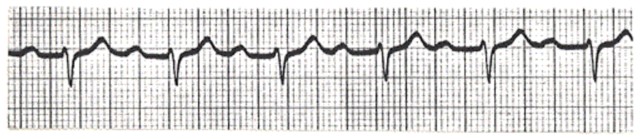
Figure 19. First degree heart block
Second-degree heart block
There are three types of second-degree block: Mobitz types I and II, and 2:1 atrioventricular block.
Mobitz type I (Wenkebach)
Type one second-degree block occurs commonly following myocardial infarction and is often self-limiting. The ECG changes are of progressive lengthening of the P-R interval, and then failure of conduction of an atrial beat. This is then followed by a normal conducted beat, before the cycle repeats (figure 20). Asymptomatic patients rarely need pre-operative treatment, although symptomatic patients will need pre-operative pacing due to the risk of developing 2:1 block resulting in haemodynamic instability. If emergency surgery is indicated, the insertion of a temporary pacing wire will suffice, before formal pacemaker insertion.
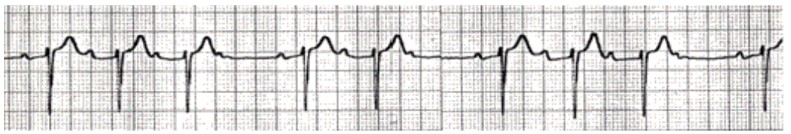
Figure 20. Mobitz type I (Wenkeback) second degree heart block
Second-degree block – 2:1 atrioventricular block
2:1 atrioventricular block is usually a Mobitz type I variant. It occurs when there is a high degree of atrioventricular blockade causing alternating conduction through the AV node of every electrical impulse from the SA node. Therefore in 2:1 block, two P waves will be found for every one QRS complex. Similarly, in 3:1 block, three P waves will be found for every one QRS complex. There is a risk of progression to third-degree block, as this type of block can represent Mobitz type II pathology. As such, senior cardiology advice is recommended.
Second-degree block – Mobitz type II
Type two second-degree block is more concerning due to the common progression to third-degree block. It occurs when there is intermittent conduction of the electrical impulse through the AV node or bundle of His, usually producing a prolonged QRS complex. Most beats are conducted normally but occasionally there is atrial systole without an associated ventricular systole. This type of block usually requires a pacemaker. If emergency surgery is indicated, the insertion of a temporary pacing wire will suffice, before formal pacemaker insertion.
In figure 21 below, the two red arrows highlight P-waves not followed by a QRS complex.
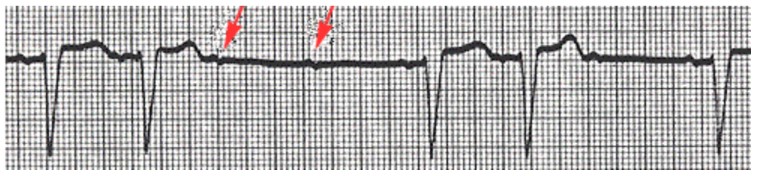
Figure 21. Mobitz Type II second degree heart block
Third-degree block / complete heart block
Third-degree block is characterised by complete dissociation between the atria and ventricles, where there is no impulse conduction at the AV node. This usually results in the ventricles reverting to an intrinsic rate of 45-55bpm with a normal QRS (figure 22 below) if the block is above the AV node. If the block is below the AV node, the ventricular rate is usually 30-40bpm with a global widening of the QRS complex (figure 23 below). Meanwhile the atria continue independently at a different, often higher rate.
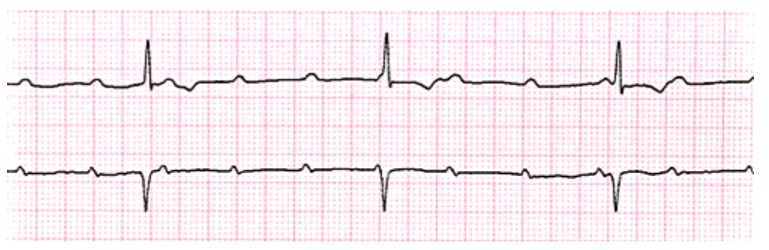
Figure 22. Third degree heart block
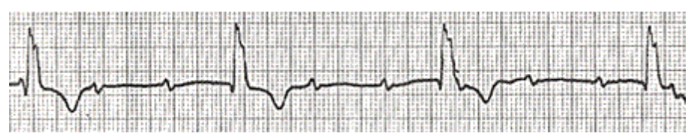
Figure 23. Third degree heart block
Occasionally this can occur due to severe vagal stimulation, and is terminated by stopping the stimulus and administering atropine IV. In very rare cases, third-degree block can be congenital. Third-degree block will need a pacemaker. If emergency surgery is indicated, the insertion of a temporary pacing wire will suffice, before formal pacemaker insertion.
The management of intraoperative heart block starts with ECG & rhythm strip assessment to diagnose the type of block. If the patient is haemodynamically unstable, give 100% oxygen and trial atropine, up to 20ug/kg. However, this is rarely effective. Other management options are:
- Start an isoprenaline infusion 1-10ug/min, to buy time if hypotension is severe.
- Dilute 0.2mg in 500ml of 5% glucose and titrate to effect (2-20ml/min)
- OR dilute 1mg in 50ml 5% glucose/dextrose-saline and titrate to effect (1.5-30ml/hr)
- Transcutaneous pacing if electrodes can be placed. Otherwise, oesophageal pacing is also effective. Pass the electrode into the oesophagus via the nasopharynx (similar to inserting a naso-gastric tube) and connect to the pulse generator. Reposition the electrode until ventricular capture.
- Transvenous pacing is more effective and reliable than the above. Senior help must be gained.
Bifasicular block
Bifasicular block is a combination of right bundle branch block with block of either the left anterior or posterior fascicle. Pathologically, it illustrates disease in two out of three myocardial conducting pathways (refer back to Figure 2, tutorial one).
The more common finding is right bundle branch block with left anterior hemiblock, and appears as an “R-S-R” pattern in V1 with left axis deviation, as shown in figure 24 below.
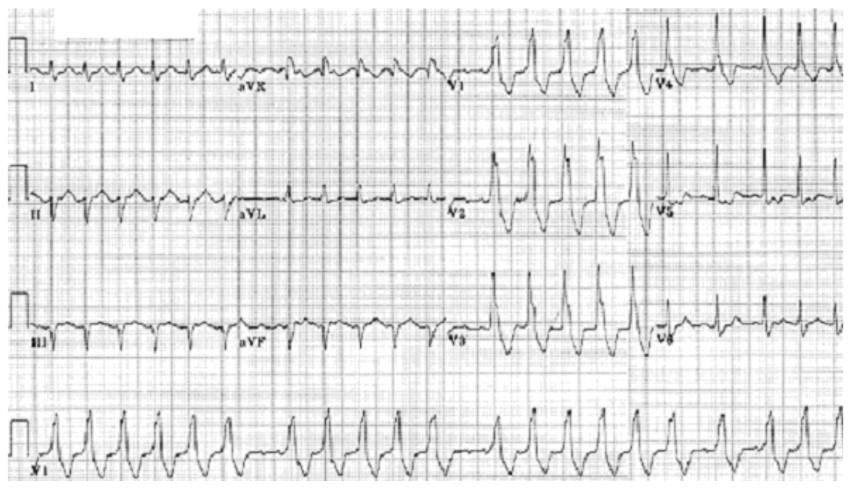
Figure 24. Bifasicular block
Right bundle branch block with left posterior hemiblock is less common due to the posterior fascicule being larger and more resistant in insult. However 60-70% of cases progress to a complete heart block and therefore its recognition is clinically important. The ECG features are a right bundle branch block, an abnormal degree of right axis deviation over +90o positive, a secondary R wave in V1, but with a normal QRS interval. However, other causes for right axis deviation should be considered and it can be a non-specific sign.
Both bifasicular blocks can occur in atherosclerotic coronary disease, congestive and hypertrophic cardiomyopathy, calcific aortic stenosis, and endocardial cushion defects.
Trifasicular block
Trifasicular block is the combination of a prolonged P-R interval (first-degree heart block) with a bifasicular block (figure 25 below). It is an illustration of disease in all three myocardial conducting pathways.
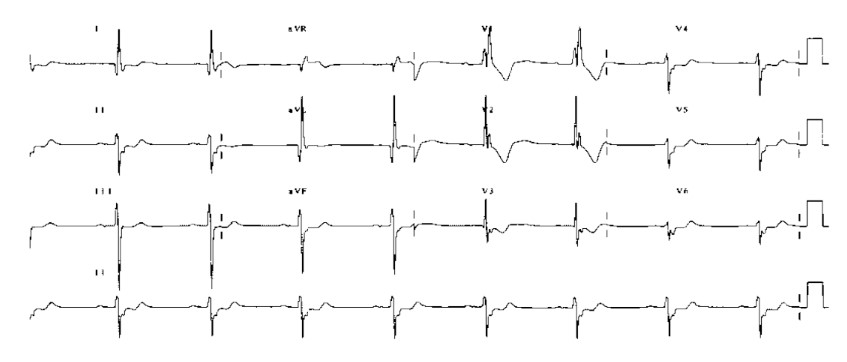
Figure 25. Trifasicular block
Unless the patient has suffered past episodes of syncope, it is rare during anaesthesia that any of the bundle branch or fasicular blocks will progress to complete heart block. Therefore, temporary pacing wires are not routinely inserted.
Both bifasicular and trifasicular blocks illustrate significant coronary and myocardial disease. Prolonged myocardial stresses pre and intra-operatively may result in myocardial ischaemia and careful cardiovascular control should aim to avoid this.
Summary
- Perioperative arrhythmias are relatively common.
- Diagnosis and appropriate management is essential to enable delivery of safe and controlled anaesthesia throughout the perioperative period.
- Your anaesthetic must be tailored to the individual patient and their co-morbidities.
- If clinically appropriate, surgery can be delayed whilst the patient is optimised and any arrhythmia managed.
- If in doubt, seek expert cardiology advice.
ANSWERS
- The causes of intraoperative ventricular tachycardia include electrolyte imbalance (particularly hypokalaemia and hypomagnesaemia), myocardial ischaemia, hypoxia, hypotension, fluid overload, and the IV use of adrenaline and other chatecholamines.
- The management of clinically stable ventricular tachycardia includes immediate treatment of any correctible cause such as electrolyte imbalance. Consider an amiodarone bolus followed by a maintenance infusion. If this fails, consider lignocaine, sotolol, procainamide, and electrical cardioversion.
- The ECG findings in right bundle branch block are of wide QRS complexes (>120ms) with an “R-S-R pattern” or “M” shape in leads V1-V3. In V5, V6 and I, the opposite is present often with a “W” shape formed by a slurred S-wave (figure 17). In the right precordial leads, ST-segment depression and T-wave inversion may also be seen.
REFERENCES AND FURTHER READING
- Valchanov K, Webb ST, Sturgess J. Anaesthetic and Perioperative Complications. Cambridge University Press, UK. 2011.
- Aitkenhead AR, Rowbotham DJ, Smith G. Textbook of Anaesthesia, 4th Ed. Churchill Livingstone,UK. 2001.
- Allman KG, Wilson IH. Oxford Handbook of Anaesthesia, 3rd Ed. Oxford University Press, UK. 2011.
- World Federation of Societies of Anaesthesiologists. Cardiovascular responses to anaesthesia. Update in Anaesthesia. Issue 10 (1999). Article 2: Pp4.
- Staikou C, Chondrogiannis K, Mani A. Perioperative management of hereditary arrhythmogenic syndromes. Br J Anaesth 108 (5): 730-44 (2012).
- Nathanson G, Gajraj NM. Perioperative management of atrial fibrillation. Anaesthesia, 53, 665-676 (1998).
- Vaughan Williams E. Classification of antiarrhythmic drugs. In: Sandoe E, Flensted-Jensen E, Olesen K, Eds. Cardiac Arrhythmias. Sodertaljie, Sweden. AB Astra, 1971: 449-72.
- Allman KG, McIndoe AK, Wilson IH. Emergencies in Anaesthesia. Oxford University Press, UK. 2005.
- Advanced Life Support algorithm. Resuscitation Council (UK). 2010. http://www.resus.org.uk/pages/als.pdf
- Tachycardia algorithm. Resuscitation Council (UK). 2010. http://www.resus.org.uk/pages/tachalgo.pdf
- Miller RD. Miller’s Anaesthesia, 7th Ed. Churchill Livingstone. Philapedphia, USA. 2010.
- Pollard BJ. Handbook of Clinical Anaesthesia, 3rd Ed. Hodder Arnold. London, UK. 2011.
- Swanton RH. Pocket Consultant Cardiology, 5th Ed. Blackwell. Oxford, UK. 2003.
- Tse HF, Lip GYH, Stewart Coats AJ. Oxford desk reference Cardiology. Oxford University Press. Oxford, UK. 2011.
- Longmore M, Wilkinson I, Rajagopalan S. Oxford handbook of clinical medicine, 6th Ed. Oxford University Press, UK. 2006.
- Da Costa D, Brady WJ, Edhouse J. ABC of clinical electrocardiography: Bradycardias and atrioventricular conduction block. BMJ 2002;324;535-538.
- Anaesthesia UK. www.frca.co.uk. 2012.
- Australian resuscitation council guidelines http://www.resus.org.au/policy/guidelines/section_11/managing_acute_dysrhythmias.htm
- EB Medicine. An evidence based approach to supraventricular tachydysrhythmias. 2013. http://www.ebmedicine.net/topics.php?paction=showTopicSeg&topic_id=133&seg_id=2668
- Varosy PD. ECG diagnosis of wide complex tachycardia: SVT vs. VT. Oct 2006. Accessed at http://www.ucsfcme.com/2006/MDM07A08/Paul%20Varosy_ECG%20Criteria%20for%20Distinguishing%20SVT%20from%20VT.pdf
IMAGE REFERENCES
- Yanowitz FG. ECG Learning Centre. University of Utah School of Medicine. Accessed at: http://ecg.utah.edu/
- Anaesthesia UK. www.frca.co.uk. 2012.
- Jenkins D. Gerred S. ECG library. Trifasicular heart block ECG only. 2009. Accessed at http://www.ecglibrary.com/ecghome.html



