General Topics
KEY POINTS
- Dexmedetomidine is a highly selective a2-adrenoceptor agonist with multiple applications in anaesthesia and intensive care medicine.
- Dexmedetomidine exerts its effects via inhibition of noradrenaline release in noradrenergic neurons. Side effects are dose dependent and include bradycardia and hypotension.
- Due to its sympatholytic, analgesic and sedative effects, dexmedetomidine is an attractive option for premedication and procedural sedation in both adult and paediatric populations.
- Dexmedetomidine can be used as an analgesic adjunct in cases in which opioid-sparing regimes are desirable (eg, bariatric surgery).
- Dexmedetomidine has a developing role as an adjunct in regional and neuraxial anaesthesia.
INTRODUCTION
Dexmedetomidine is a highly selective a2-adrenoceptor agonist. It exerts sympatholytic, analgesic and sedative effects which make it an attractive option for the maintenance of sedation in the intensive care unit (ICU) and for the conscious sedation of adults and children during diagnostic or surgical procedures. This tutorial will cover basic and practical pharmacology as well as its clinical applications in the perioperative context. Please see the recent tutorial on the use of dexmedetomidine in the ICU (ATOTW 454) for its use in critical care sedation.1
STRUCTURE, PRESENTATION AND DOSING
Dexmedetomidine is the active dextro-isomer of medetomidine, an imidazole compound used as a veterinary sedative and analgesic. It is presented as a clear, colourless solution of 100 μg-ml-1 dexmedetomidine hydrochloride, which is usually diluted2 with normal saline to a concentration of either 4 μg-ml-1 or 8 μg-ml-1. Dosing varies depending on the application however the most common dosing regimen for conscious sedation involves an infusion of 0.2 – 1.4 μg-kg-1-h-1 with or without an initial loading dose of 1 μg-kg-1 over 10 minutes.
PHARMACOKINETICS
Administration and Absorption
Although only licenced for intravenous (IV) use, multiple routes of administration of dexmedetomidine have been described, including oral, intranasal, intramuscular, buccal and as an additive in neuraxial and peripheral nerve blocks. Oral use is limited by its extensive first-pass metabolism.
Distribution
On intravenous administration, dexmedetomidine exhibits 2-compartment kinetics with rapid redistribution, including across the blood-brain barrier and placenta.2 It is highly protein bound, and at steady state, the volume of distribution in healthy adults is approximately 1 – 2l-kg-1 however this is increased in neonates and children and can vary widely in critically ill patients.
Metabolism and Elimination
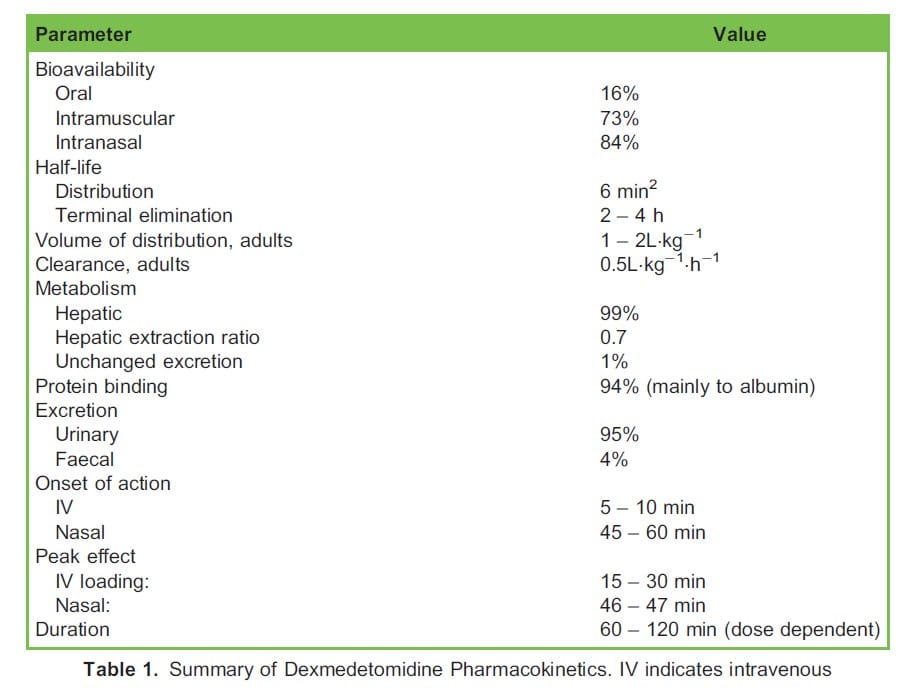
Metabolism takes place in the liver via N-glucuronidation, N-methylation and cytochrome P450 catalysed oxidation to inactive metabolites which are mainly excreted in the urine. The estimated context-sensitive half time of dexmedetomidine is favourably short at between 1 and 4 hours.3 Thus, the elimination half-life is similar following a single dose or infusions of more than 24 hours. A summary of the pharmacokinetics of dexmedetomidine can be found in Table 1.
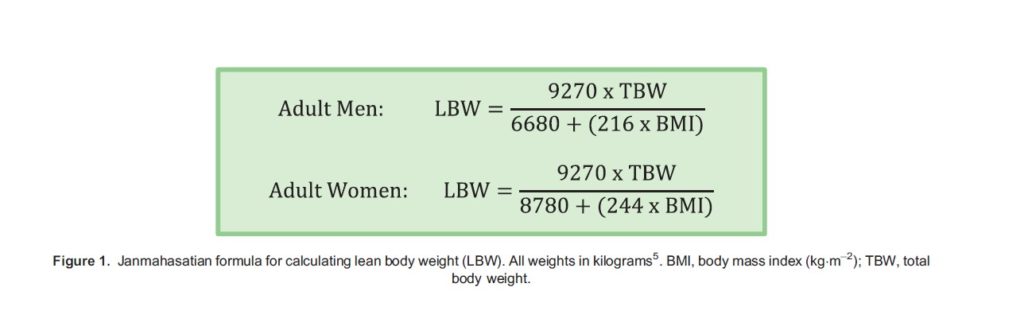
No major differences in the pharmacokinetics of dexmedetomidine have been observed in renal impairment however half-life prolongation and reduced clearance have been observed in hepatic impairment.2 With regard to obesity, infusions based on actual body weight can produce supratherapeutic plasma concentrations. In a recent study involving obese patients undergoing abdominal surgery, a dosing regimen scaled to lean body weight (LBW) adequately described dexmedetomidine pharmacokinetics.4 In our institution, we use LBW as calculated by the Janmahasatian formula5 (Figure 1) for dosing of dexmedetomidine during bariatric surgery.
MECHANISM OF ACTION
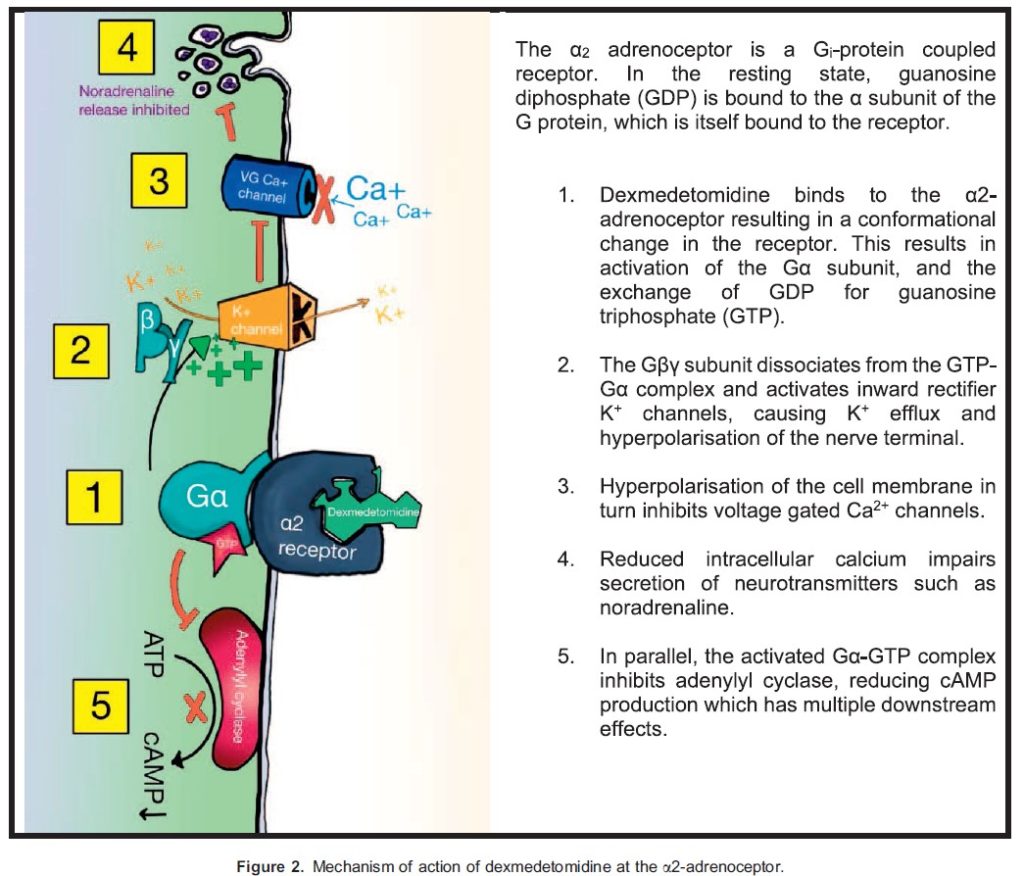
Dexmedetomidine is a highly selective a2-adrenoceptor agonist (a1:a2 ratio 1:1620). Dexmedetomidine is approximately 4 times more selective for a2-adrenoceptors than clonidine (a1:a2 ratio 1:400). The mechanism of action of dexmedetomidine at the a2-adrenoceptor is summarised in Figure 2.
 Centrally, a2-adrenoceptors are found on the presynaptic membrane of noradrenergic neurons. Thus, agonism results in the inhibition of noradrenaline release. In the peripheral vasculature, a2-adrenoceptor agonism causes vasoconstriction. Dexmedetomidine also appears to have some activity at imidazoline or ‘‘I’’ receptors, which also contributes to its sympatholytic, sedative and analgesic effects however knowledge of this mechanism is limited.
Centrally, a2-adrenoceptors are found on the presynaptic membrane of noradrenergic neurons. Thus, agonism results in the inhibition of noradrenaline release. In the peripheral vasculature, a2-adrenoceptor agonism causes vasoconstriction. Dexmedetomidine also appears to have some activity at imidazoline or ‘‘I’’ receptors, which also contributes to its sympatholytic, sedative and analgesic effects however knowledge of this mechanism is limited.
PHARMACODYNAMICS
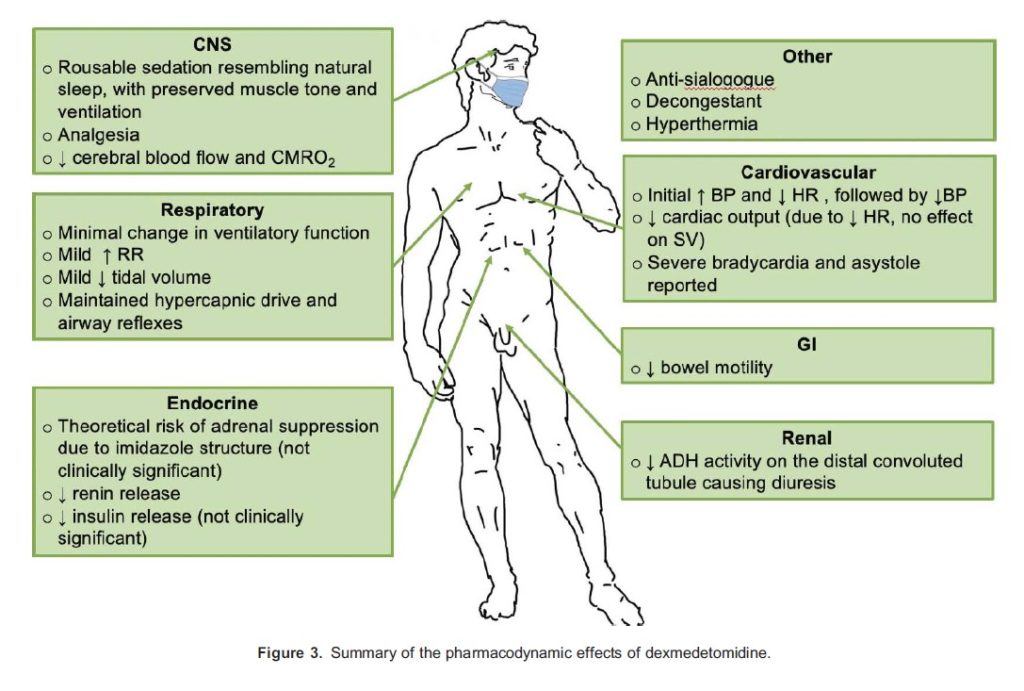
The sedative effects of dexmedetomidine are thought to arise through the activation of pre- and postsynaptic a2-adrenoceptors in the locus coeruleus, the main noradrenergic nucleus in the pons.
Its analgesic effects are thought to be mediated by central nociceptive receptor activation resulting in interneuron hyperpolarisation and the reduced release of nociceptive neurotransmitters such as glutamate and substance P.
Dexmedetomidine demonstrates concentration-dependent a-adrenoceptor selectivity. Clinically this results in a biphasic haemodynamic response. Following a bolus dose, high plasma concentrations cause peripheral a2B-mediated vasoconstriction, causing a transient rise in blood pressure and reflex reduction in heart rate. As concentrations fall, the activation of presynaptic a2 adrenoceptors in the locus coeruleus reduces catecholamine release resulting in a fall in blood pressure. A similar effect is also observed during infusions. At low infusion rates hypotension predominates however as infusion rates increase blood pressure rises and may surpass baseline.6
The pharmacodynamic effects of dexmedetomidine are summarised in Figure 3.
ADVERSE EFFECTS, INTERACTIONS AND WITHDRAWAL
The most commonly reported adverse effects of dexmedetomidine during both ICU and procedural sedation include bradycardia, hypotension and respiratory depression. Coadministration of dexmedetomidine with inhalational and intravenous anaesthetic agents, benzodiazepines and opioids will accentuate these effects. Cautious concomitant use of agents that also produce bradycardia and hypotension, such as b-blockers, is advised as there have been case reports in the literature of severe bradycardia leading to asystole.
An uncommon but significant adverse effect of dexmedetomidine is hyperthermia however the exact mechanism is not yet clear. It is an important differential diagnosis in a patient with unexplained pyrexia to avoid assumptions of infection which may lead to unnecessary antibiotic exposure.
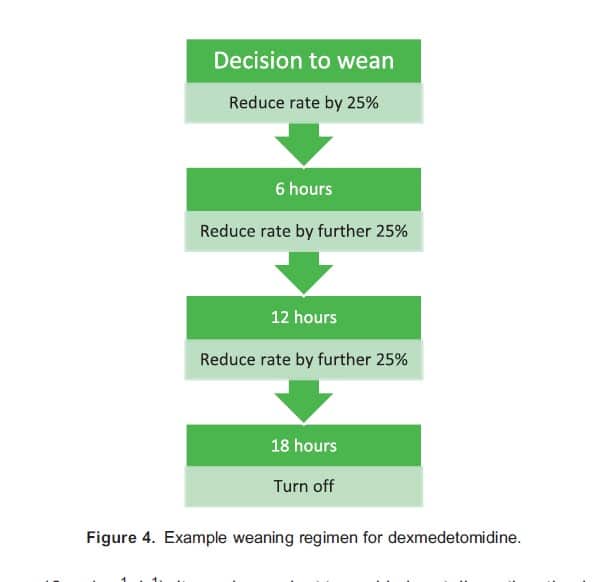
Withdrawal symptoms including hypertension, tachycardia and agitation have been described in patients receiving prolonged infusions of dexmedetomidine. These phenomena appear to be related to higher infusion rates (eg, . 0.8 μg-kg-1-h-1) and cumulative daily doses (eg, . 12 μg kg-1 d-1). It may be prudent to avoid abrupt discontinuation by weaning the infusion over several hours. An example of a weaning regimen is provided in Figure 4.
Clonidine is often used as an alternative tapering agent in this context. Existing low-quality evidence suggests that clonidine is a safe option to reduce the duration of dexmedetomidine treatment and wean and may facilitate ongoing a2-adrenoceptor agonism in a noncritical care environment.7
PROCEDURAL SEDATION
Awake Fibre-Optic Intubation
Optimal sedation is key to the success of an awake fibre-optic intubation (AFOI). Although not yet accepted into common practice, dexmedetomidine has unique pharmacologic properties that make it attractive for this purpose, notably its minimal effects on respiratory function even at high doses.6 A 2014 Cochrane review on this topic found that dexmedetomidine decreased procedural discomfort when compared with propofol target-controlled infusion (TCI) or midazolam and had similar intubation times.8 It resulted in less airway obstruction compared with propofol TCI.8 There was no significant increase in cardiovascular adverse effects when compared with fentanyl, midazolam or propofol-based techniques.8 One study comparing remifentanil to dexmedetomidine for AFOI found that the dexmedetomidine group had a higher rate of recall and required more intubation attempts but that lower oxygen saturations were observed in the remifentanil group.9 Interestingly, we came across few studies comparing dexmedetomidine to remifentanil for AFOI; this may be a consideration for future research. See Table 2 for a suggested regimen for dexmedetomidine in AFOI.
Paediatric Procedural Sedation
Undertaking magnetic resonance imaging (MRI) in young children requires planning and skill to ensure optimal images are obtained for diagnostic purposes. Conscious sedation with dexmedetomidine is gaining popularity over general anaesthesia for this purpose.


A 2020 meta-analysis of intranasal dexmedetomidine for procedural sedation in paediatrics found that it provides effective sedation for both painful and nonpainful procedures and in fact may be superior to midazolam and chloral hydrate.10 An additional advantage of intranasal dexmedetomidine is its palatability compared with oral midazolam, which can be challenging to administer due to its unpleasant taste. See Table 3 for a suggested regimen for use in this context.
Premedication
Dexmedetomidine is a useful tool for the premedication of children and adults before general anaesthesia. In our institution, we use 2 μg kg-1 of undiluted dexmedetomidine (100 μg ml-1) via a mucosal atomisation device (MAD) intranasally for children. For adults, a reduced dose is advised (i.e, 1.5 μg kg-1). We have found that using the smallest possible syringe (e.g. 1ml) allows for better atomisation and that a minimum of 40 minutes is required for optimal effect.
INTRAOPERATIVE USE
Dexmedetomidine has shown promise for use during the intraoperative period, notably for its favourable effects on the stress response to surgery, as an analgesic adjunct and as a possible prophylactic agent for postoperative delirium and agitation.
Attenuation of Stress Response to Surgery
Use of perioperative dexmedetomidine is associated with reduced levels of adrenaline, noradrenaline and cortisol in patients undergoing a variety of surgeries11. Furthermore, dexmedetomidine appears to have a favourable effect on immune cells and inflammatory cytokine levels11. It has been shown that concomitant use of dexmedetomidine alongside general anaesthesia for major abdominal surgery may blunt the stress response to an extent comparable to combined epidural and general anaesthesia and thus may be an option in patients in whom a neuraxial technique is not possible12. Although there is a clear benefit from the use of dexmedetomidine in terms of inflammation and markers thereof, the association with perioperative stress, inflammation and clinical outcomes is not yet clear.
Postoperative Delirium and Agitation
Dexmedetomidine modifies several contributors to delirium, including inflammation, pain, promotion of natural sleep-wake cycles and use of delirium inducing agents such as opioids. Recent meta-analyses demonstrated a reduction in postoperative delirium among a variety of patients treated with dexmedetomidine intraoperatively13. Timing, dosing and duration of infusion varied significantly however and there was insufficient evidence to draw conclusions on the effect of dexmedetomidine on the duration of mechanical ventilation or length of ICU or hospital stay.
Analgesic Adjunct
When compared with remifentanil, intraoperative dexmedetomidine appears to be superior for reducing pain scores and opioid requirement up to 24 hours postoperatively, with less hypotension, shivering and nausea.14 Overall, the current evidence is weak, which may explain the lack of universal adoption of this technique however in cases in which postoperative opioid use is undesirable for example during bariatric surgery, intraoperative dexmedetomidine may have a role as part of a multimodal opioid-free or -sparing regimen. Dexmedetomidine forms part of the standard bariatric anaesthetic in our institution; we use a loading dose of 0.6 μg kg-1 given over 15 minutes followed by 0.5 – 1 μg kg-1 h-1 (max 1.5 μg kg-1 h-1) for the duration of the case. See Table 4 for suggested dosing regimen.
Regional Anaesthesia
Dexmedetomidine has shown some utility as an adjunct in peripheral and neuraxial nerve blocks. The addition of dexmedetomidine to local anaesthetics in peripheral nerve blockade has been shown to significantly prolong the duration of analgesia by approximately 5 hours. Motor blockade is also prolonged and while perineural dexmedetomidine does not appear to cause neurotoxicity, hypotension and bradycardia may be more common when it is used.15 The optimal dose of dexmedetomidine as an adjunct in peripheral nerve blockade has not been fully elucidated however doses in the range of 25 – 150 μg will prolong sensory blockade. The risk of hypotension and bradycardia is increased in patients treated with perineural dexmedetomidine. A dose of 100 μg has been suggested by the authors of 1 small randomised controlled trial as the best compromise for both efficacy and safety.16

When given intrathecally, dexmedetomidine prolongs the duration of neuraxial block and may reduce onset time, analgesic requirement and the incidence of postoperative shivering. Doses from 2 – 20μg have been used without significant differences in duration of analgesia or adverse effects.17 Similarly, epidural dexmedetomidine has been shown to prolong analgesia and reduce pain scores and time to onset in a variety of patients when combined with traditional local anaesthetics, either as a bolus or continuous infusion.18 Bolus doses in the literature range from 0.5 – 1 μg kg-1 in 10 – 20ml of local anaesthetic. As an increased incidence of motor block.19
Maternofoetal Surgery
Following the publication of several studies in animals, including non-human primates, concerns have been raised over the possible harmful effects of inhalational anaesthetics on brain development. Fortunately, most of the preclinical trials suggesting harmful effects on the developing brain employed high concentrations of or prolonged exposure to volatile anaesthetics far beyond what occurs in routine clinical practice. Early experimental studies have shown that dexmedetomidine exerts an anti- apoptotic effect in animal models of perinatal brain injury. Thus dexmedetomidine has been proposed as part of a neuroprotective strategy to reduce harm to the foetus during maternal general anaesthesia.20
CONCLUSION
Dexmedetomidine is a unique and versatile drug with multiple applications in critical care and anaesthesia. A thorough understanding of its mechanism of action and potential side effects will allow practitioners to use it safely and effectively. Further research is required to explore its use in novel areas such as AFOI and as part of opioid-sparing analgesia regimes.
REFERENCES
- Bonfield S, Garland A, English W. ATOTW 454: Use of Dexmedetomidine for Sedation in Adult Critical Care. ATOTW. August 2021. Accessed September 2021. https://resources.wfsahq.org/atotw/use-of-dexmedetomidine-for-sedation-in- adult-critical-care/
- Orion Pharma (UK) Ltd. Dexmedetomidine summary of product characteristics. Accessed November 26, 2020. https:// medicines.org.uk/emc/product/4783/smpc#gref
- Iirola T, Ihmsen H, Laitio R, et Population pharmacokinetics of dexmedetomidine during long-term sedation in intensive care patients. Br J Anaesth. 2012;108(3):460-468. doi:10.1093/bja/aer441
- Rolle A, Paredes S, Cortı´nez LI, et Dexmedetomidine metabolic clearance is not affected by fat mass in obese patients. Br J Anaesth. 2018;120(5):969-977. doi:10.1016/j.bja.2018.01.040
- Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM, Green Quantification of lean bodyweight. Clin Pharmacokinet. 2005;44(10):1051-1065. doi:10.2165/00003088-200544100-00004
- Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93(2):382-394. doi:10.1097/00000542-200008000-00016
- Bhatt K, Thompson Quan A, Baumgartner L, et Effects of a clonidine taper on dexmedetomidine use and withdrawal in adult critically ill patients—a pilot study. Crit Care Explor. 2020;2(11):e0245. doi:10.1097/CCE.0000000000000245
- He XY, Cao JP, He Q, Shi Dexmedetomidine for the management of awake fibreoptic intubation. Cochrane Database Syst Rev. 2014;(1):CD009798. doi:10.1002/14651858.CD009798.pub2
- Hagberg C, Lam N, Abramson S, Vahdat K, Craig Dexmedetomidine vs. remifentanil for sedation in awake intubations—a randomized, double-blind trial [abstract]. Anesthesiology. 2008;109:A14.
- 10 Poonai N, Spohn J, Vandermeer B, et al. Intranasal dexmedetomidine for procedural distress in children: a systematic review. Pediatrics. 2020;145(1):e20191623. doi:10.1542/peds.2019-1623
- Wang K, Wu M, Xu J, et al. Effects of dexmedetomidine on perioperative stress, inflammation, and immune function: systematic review and meta-analysis. Br J Anaesth. 2019;123(6):777-794. doi:10.1016/j.bja.2019.07.027
- Li Y, Wang B, Zhang LL, et Dexmedetomidine combined with general anesthesia provides similar intraoperative stress response reduction when compared with a combined general and epidural anesthetic technique. Anesth Analg. 2016;122(4):1202-1210. doi:10.1213/ANE.0000000000001165
- Duan X, Coburn M, Rossaint R, Sanders RD, Waesberghe JV, Kowark Efficacy of perioperative dexmedetomidine on postoperative delirium: systematic review and meta-analysis with trial sequential analysis of randomised controlled trials. Br J Anaesth. 2018;121(2):384-397. doi:10.1016/j.bja.2018.04.046
- Grape S, Kirkham KR, Frauenknecht J, Albrecht E. Intra-operative analgesia with remifentanil vs. dexmedetomidine: a systematic review and meta-analysis with trial sequential analysis. Anaesthesia. 2019;74(6):793-800. doi:10.1111/anae. 14657
- Schnabel A, Reichl SU, Weibel S, et al. Efficacy and safety of dexmedetomidine in peripheral nerve blocks: a meta- analysis and trial sequential Eur J Anaesthesiol. 2018;35(10):745-758. doi:10.1097/EJA.0000000000000870
- Keplinger M, Marhofer P, Kettner SC, Marhofer D, Kimberger O, Zeitlinger M. A pharmacodynamic evaluation of dexmedetomidine as an additive drug to ropivacaine for peripheral nerve blockade: a randomised, triple-blind, controlled study in Eur J Anaesthesiol. 2015;32(11):790-796. doi:10.1097/EJA.0000000000000246
- Paramasivan A, Lopez-Olivo MA, Foong TW, Tan YW, Yap Intrathecal dexmedetomidine and postoperative pain: a systematic review and meta-analysis of randomized controlled trials. Eur J Pain. 2020;24(7):1215-1227. doi:10.1002/ejp. 1575
- Zhang X, Wang D, Shi M, Luo Y. efficacy and safety of dexmedetomidine as an adjuvant in epidural analgesia and anesthesia: a systematic review and meta-analysis of randomized controlled trials. Clin Drug Investig. 2017;37(4):343- doi:10.1007/s40261-016-0477-9
- Wangping Z, Ming optimal dose of epidural dexmedetomidine added to ropivacaine for epidural labor analgesia: a pilot study. Evid Based Complement Alternat Med. 2017;2017:7924148. doi:10.1155/2017/7924148
- Andropoulos DB. Effect of anesthesia on the developing brain: Infant and fetus. Fetal Diagn Ther. 2018;43(1):1-11. doi:10. 1159/000475928



