Paediatric Anaesthesia
KEY POINTS
- Appreciation of patient and procedural risk factors for hemorrhage, development of system-level patient blood management programs, and case-specific, intraoperative goal directed care are important strategies to decrease morbidity and mortality associated with pediatric hemorrhage.
- Primary goals in hemorrhage management are to maintain hemodynamic stability, support oxygen delivery/end organ perfusion, prevent coagulopathy, and reduce harm associated with blood product transfusion including transfusion associated circulatory overload (TACO) and transfusion associated acute lung injury (TRALI).
- The ratios of blood components used in massive transfusion, the use of adjunctive care strategies including anti- fibrinolytics, and ongoing post-operative resuscitation are key components of patient blood management.
- The development of a multidisciplinary patient blood management system is important to effectively and safely address the demands and strain on a system caused by a patient with massive hemorrhage.
INTRODUCTION
The management of massive hemorrhage and massive transfusion has been described in adults through studies and experience in combat situations. It has been shown that tourniquet use, damage control resuscitation strategies, balanced transfusion ratios, and anti-fibrinolytic therapy have important implications in decreasing trauma related mortality. (1) Treatment of massive hemorrhage in children is less well defined and many regimens related to its management have been extrapolated from the adult trauma literature and techniques. (2) Children are not small adults and evidence-based strategies specifically designed for management of massive hemorrhage and transfusion in children should be utilized to help decrease preventable morbidity and mortality. (3) These strategies should include awareness of patient or procedural risk factors, system and provider preparation for potential hemorrhage in high risk situations, and intraoperative goal-directed care with a particular emphasis on prevention of the well-described lethal triad (coagulopathy, acidosis, and hypothermia) associated with massive transfusion. This tutorial acts as a guideline for managing blood loss in the pediatric patient specifically in the perioperative period.
DEFINING HEMORRHAGE IN CHILDREN
Definition and Etiology
In the pediatric population, massive hemorrhage can be defined as blood loss exceeding one circulating blood volume (CBV) within 24-hours, blood loss of 50% of CBV within 3 hours, or transfusion at a rate of 10% of total blood volume (TBV) every 10 minutes. (4) This definition requires that the provider calculate the weight-based estimated circulating blood volume (EBV) of the patient.
Massive transfusion is less well defined. One possible definition is from the combat literature. In injured pediatric patients, transfusion of greater than 40 ml/kg in the first 24 hours post injury is considered a massive transfusion and is associated with an increased risk for in-hospital death. (6)
Hemorrhage can occur in any surgical situation in children but is most notable in trauma, liver transplant, cardiac surgery, major spinal surgery, cranial vault surgery, neurosurgical procedures (arteriovenous malformations, Vein of Galen), and minimally invasive procedures where direct control of bleeding can be problematic. Hemorrhage risk can also be elevated due to patient related factors including inherited bleeding disorders such as Von Willebrand’s Disease and hemophilia or acquired bleeding disorders such as dilutional coagulopathy. (4) A high level of vigilance for perioperative bleeding should be maintained in patients with these hemostatic risk factors.
GOALS OF HEMORRHAGE MANAGEMENT IN THE PEDIATRIC PATIENT
The management of hemorrhage should employ goal directed therapy that seeks:
- To promote hemodynamic stability as measured by vital signs
- To maintain end organ perfusion and oxygen delivery
- To reduce harm and side effects associated with transfusion (4)
- To avoid over-transfusion through the use of point of care and laboratory testing and appropriate utilization of blood components.
- To prevent the lethal triad of coagulopathy, acidosis, and hypothermia. (3)
While efforts are being made to stop and control the bleeding, these hemorrhage management goals can be achieved by goal- directed therapy which may include the transfusion of appropriate blood products.
However, until blood products are available, initial therapy may require boluses of crystalloid or colloid solutions. This must be done with care as large quantities may contribute to dilutional coagulopathy. (3) Maintaining an appropriate blood pressure during massive hemorrhage may require the use of temporizing measures such as vasopressor support. (4) Appropriate blood pressure goals vary by age and tend to increase with advancing age. It is recommended that mean systolic blood pressure (SBP) be maintained at an average of >55 mm Hg in premature neonates and >110 in adolescents. (5) Lactate, measurement of base deficit, and urine output can help to monitor end organ perfusion. (3) Non-invasive cerebral oxygenation (near infrared spectrometry; NIRS) monitoring may also play a role as increases in brain tissue oxygen tension have been noted after RBC transfusion. (3)
Transfusion Risk
There are a number of risk factors and side effects that should be considered during hemorrhage management. Transfusion of blood products can lead to hemolytic transfusion reactions as well as non-hemolytic reactions such as transfusion related acute lung injury (TRALI), transfusion associated circulatory overload (TACO), and transfusion related immunomodulation (TRIM). Banked blood products can also cause citrate toxicity and electrolyte abnormalities such as hyper- and hypokalemia, hypocalcemia, and hypomagnesemia. (3, 4) There should be a heightened awareness of these derangements during high volume transfusion situations as can significantly alter outcome and early treatment is critical.
It is recommended that hyperkalemia risk be reduced in children under 1-year-old or < 10 kilograms by blood washing or the use of ‘‘fresh’’ red blood cells collected within 7 days of transfusion. (7) This population has high risk of transfusion-associated hyperkalemic arrest compared to adults due to low circulating volume and renal immaturity. (5)
Patient Blood Management (PBM)
Patient Blood Management is the timely application of evidence-based medical and surgical concepts designed to maintain hemoglobin concentration, optimize hemostasis and minimize blood loss to improve patient outcomes. (8) The development of a multidisciplinary Patient Blood Management (PBM) program within an institution can help to effectively and safely address the demands and strain on the system caused by a patient with massive hemorrhage. This program should include protocols to direct the availability of blood products after initiation of a large-scale transfusion event, transfusion triggers to activate such an event, and the quantity/ratio of blood products provided in the event. This allows for safe and effective optimization of blood products usage while reducing unnecessary transfusions. This can potentially decrease morbidity and mortality for the patient and decrease health care costs for the institution. (8)

Table 1. Weight based calculation of circulating blood volume (CBV) (5)
PREOPERATIVE MANAGEMENT
In surgical cases with a high risk of massive hemorrhage or in cases where the patient’s comorbidities increase the risk of massive hemorrhage, careful preoperative planning is essential. Care should be taken to obtain a complete history, physical, and appropriate screening laboratory tests.
Preoperative laboratory studies should be utilized to assess for any existing coagulopathy or anemia. If present, these conditions should be treated in a timely fashion, and, if possible, elective surgery delayed for medical optimization. Iron supplementation should be considered for iron deficiency anemia (4) Treatment of pre-existing conditions of hemostasis such as hemophilia and Von Willebrand’s disease should be performed.
Autologous blood donation is another preoperative strategy to consider in specific situations such as when the patient is difficult to cross match due to antibodies or has a rare blood type. However, this practice is not routinely recommended given the unfavorable risk-benefit ratio. Risks include inducing preoperative anemia and clerical error associated with autologous donation. (4)
The use of preoperative embolization may be beneficial in specific situations such as AVMs or tumors whose surgical resection has a high risk of massive hemorrhage. However, embolization has the associated risk of additional anesthesia for the procedure and risk of injury during the embolization procedure which may in itself necessitate hemorrhage management. It is recognized that the ability to embolize at risk lesions may not be readily available in many locations. (4)
The blood bank should be consulted early and blood type screening initiated prior to high risk procedures so that appropriate blood products can be available for the patient. Cross-matched blood products should be readily available in high risk situations. Emergency release blood may be necessary in some situations.
INTRAOPERATIVE MANAGEMENT
Intraoperative hemorrhage may occur suddenly, unexpectedly, and, at times, rapidly. Medical systems’ PBM programs should have a tested massive hemorrhage management plan (including massive transfusion protocol (MTP)) in place in the event of such a situation. This plan should be adapted to meet the capabilities of the institution. Things to consider in developing massive hemorrhage guidelines are intra-operative blood conservation strategies, types and quantities of blood products to be utilized at the time of hemorrhage, and adjunct therapies to be used in association with transfusion.
Blood Volume Calculation
In all surgical cases it is advisable to calculate the weight based estimated blood volume (EBV) in accordance with Table 1. This can be used to calculate a maximal allowable blood loss volume (MABL) via the following calculation:
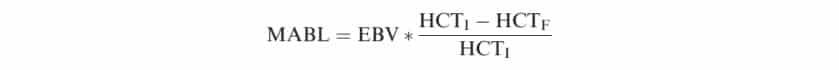
HCTI = the initial HCT (hematocrit) and HCTF = the accepted minimal HCT for patient (5)
Example: A 1 year old (9 kg) male presenting for craniosynostosis repair has a HCT of 34%(0.34). His EBV would be approximately 720 ml (9 kg x 80 ml/kg). His MABL would be calculated as follows assuming accepted minimal HCT of 25%:
MABL = 720ml ∗ (0.34 – 0.25) / 0:34 = 720ml ∗ 9/0.34 = 190ml
The accepted minimal HCT may vary depending on comorbidities but 21-25% (with a corresponding restrictive hemoglobin target of 7 g/dL in a hemodynamically stable patient or 8 g/dL in an actively bleeding patient) is often used in otherwise healthy patients. (4,9) This number weighs the risk and benefits of poor outcome from blood loss versus risk of blood product transfusion and supports a restrictive transfusion methodology.
Hemorrhage Management
Good venous and, if possible, arterial access are essential in hemorrhaging patients. In potential hemorrhage situations, appropriate large bore IV access to allow for rapid infusion of products should be obtained prior to procedure. Large bore is an ambiguous term in the pediatric population as a 22 gauge IV would be appropriate for transfusion in an infant due to the relatively low amount of volume required for resuscitation but would likely not provide enough flow to rapidly resuscitate an 18- year-old man. Central venous access, cut-down or intraosseous device may be useful in the event of hemorrhage but should be placed based on provider comfort. A long, 18-gauge central cannula has a similar flow rate to a 24-gauge IV. (5) Central access as well as arterial access can be utilized for rapid sampling of blood for transfusion monitoring. Central access may be a safer mechanism to instill temporizing vasopressors. Central access sites (femoral vein, subclavian vein, femoral vein) each have their own set of risk which should be considered before access is obtained. (5)
In most circumstances, a PRBC volume of 10-20 ml/kg should be utilized for initial transfusion. (3) In massive, sudden hemorrhage higher volume may be needed.
With massive hemorrhage requiring massive transfusion, red blood cell transfusion should be balanced with ‘‘yellow’’ blood product transfusion in the form of 1:1:1:1 volume ratio of PBRC: fresh frozen plasma (FFP): platelets: cryoprecipitate to prevent coagulopathy while maintaining adequate oxygen delivery to tissues. (3,4,11) Transfusion of fresh frozen plasma (FFP), platelets, and cryoprecipitate should be considered in managing massive hemorrhage when EBV loss is greater than 50% of total blood volume. (3,4). Preferably blood product administration should be goal directed and guided by point of care and laboratory testing such as thromboelastography (TEG or ROTEM), if available. (3,4)
Blood transfusion creates laboratory derangements due to dilution as well as from red cell lysis and blood preservative components. Electrolyte and coagulation testing should be used if available to evaluate for abnormalities. However, empiric treatment of some abnormalities may be utilized if testing is unavailable (i.e. acidosis and hypocalcemia). (4) Calcium can be replaced with calcium gluconate (30-100 mg/kg) or calcium chloride (20 mg/kg) (5) and acidosis can be improved with sodium bicarbonate (1 mEq/kg). (5) Calcium chloride can cause tissue necrosis if there is line infiltration. It should only be administered via central access. Calcium gluconate is a safer medication for peripheral administration. Attention must also be made to levels of magnesium and potassium and should be treated. Hyperkalemia, hypokalemia and hypomagnesemia can all be associated with high volume transfusion. (4)
Minimal platelet count transfusion thresholds in children have not been determined but a platelet count of 50 × 109 platelets/L is currently considered adequate during ongoing bleeding in the adult population. (10) A transfusion of 5-10 ml/kg of platelets will increase platelet count by 50-100 × 109 platelets/L. (4) This has been similarly noted in the pediatric population. FFP in a dose of 10-15 ml/kg should be used for transfusion. (4) This can help to support fibrinogen levels which may impair coagulation when <150-200 mg/dL. (4) Fibrinogen levels should be maintained >150 mg/dL during massive transfusion to optimize hemostasis. Cryoprecipitate and fibrinogen concentrate are the best sources of fibrinogen.
Massive Hemorrhage Protocol
Unforeseen hemorrhage presents a challenge to clinicians as blood products may not be immediately available, patients may be in extremis, and intravenous access poor. It is in these situations that following a massive hemorrhage protocol which also incorporates a massive transfusion protocol (MTP) can be incredibly beneficial. (8) It helps the healthcare facility to have an established protocol to allow for the immediate release of blood products in a set ratio. (11) Many pediatric institutions provide units based on the weight of the patient in order to conserve blood products for the institution (i.e. a blood volume for a 5 kg child may only require replacement of 1 unit of blood while a 25 kg adolescent would need several units of blood for one volume). In addition to decreasing the stress on the system, the immediate availability of blood products can allow the anesthesiologist to use goal-directed approach to transfusion upon receiving the products and can potentially avoid the overuse of crystalloid solutions thus preventing an unnecessary coagulopathy. (11)
In massive and especially unforeseen hemorrhage, MTPs should be activated early. Any massive transfusion system should be coordinated among provider and auxiliary care groups (i.e. blood bank, nursing, ICU). In an activated massive transfusion, the goal should be for blood bank teams to deliver products ahead of need so that they can be effectively transfused by direct patient care providers. (11)
Adjunct Considerations
It is essential during massive hemorrhage to consider physical and pharmacological adjunct therapies to help reduce the physiological effects of massive hemorrhage. Specifically, these therapies should aim to prevent the lethal triad of coagulopathy, acidosis, and hypothermia. (4) Temperature should be closely monitored and active warming using forced-air warmers, heat lamps, warmed blankets, room warming, and appropriate warming of recently thawed blood products. (4)
Cell salvage techniques can be used to collect autologous blood from the surgical field for processing and transfusion. In infants, salvageable cells can be difficult to collect in adequate quantities for transfusion. Overall cell salvage may be best utilized in patients > 10 kg and with > 40% blood loss. (4) There is controversy about the use of cell salvage in tumor surgery or in circumstances where blood cell lysis occurs. It is also recognized that this may not be a reasonable management strategy in many locations. (4)
The use of antifibrinolytic therapy as an adjunct to decrease surgical bleeding has been studied in both the adult and pediatric population. (12) Tranexamic acid (TXA), an inhibitor of plasmin activation, is one of the common antifibrinolytics utilized. [Further information about TXA can be found in ATOTW #406.] Numerous studies have shown its utility in the pediatric population during foreseen blood loss procedures. (13) The PED-TRAX trial established that TXA is associated with reduced mortality in pediatric trauma (14) and stated that ‘‘the timely administration of TXA to injured patients is associated with a survival advantage and this advantage seems to extend to the injured pediatric population.’’ (14) The ideal TXA therapeutic dosage range is a loading dose of 10 to 30 mg/kg (max 2 grams) given over 15 minutes followed by an infusion of 5 to10 mg/kg/ h. (4,15) These doses have been shown to maximize effect while reducing harmful side effects including seizures. (15) TXA has been safely used in pediatrics but is contraindicated in patients with active thromboembolic disease or conditions with consumption coagulopathy. It is relatively contraindicated in patients with renal dysfunction and acquired thrombotic disease. (15)
POSTOPERATIVE MANAGEMENT
After leaving the operating room, patients often remain critically ill after a massive hemorrhage and its associated management. They may require ongoing resuscitation with blood products and adjunct therapy. It is not uncommon that children develop transfusion associated circulatory overload (TACO) and transfusion associated acute lung injury (TRALI) and may require further cardiac and respiratory support. (4) This will require an intensive level of care and may continue to burden the healthcare system. Further intensive support can be administered to the best of the system’s ability with the aid of physicians, nurses, and ancillary staff.
CONCLUSION
Massive hemorrhage in the pediatric population poses a high risk of morbidity and mortality. In many situations, this risk can be reduced through appropriate preoperative planning, intraoperative management, and postoperative care. Proper blood management strategies, including use of a multidisciplinary massive transfusion protocol, can reduce risk of harm to patients while alleviating some of the stress that these critical events pose to a healthcare system. These should be developed and adapted to fit the resources available in the system. In addition, it is essential that these strategies are frequently simulated and that they be adapted as newer research becomes available regarding the safest way to manage hemorrhage in this unique population.
REFERENCES
- Daniel Y, Habas S, Malan L, et al. Tactical damage control resuscitation in austere military environments. J R Army Med Corp. 2016 Dec; 162(6): 419-427.
- Hendrickson JE, Shaz BH, Periera G, et al. Implementation of a pediatric trauma massive transfusion protocol: on institution’s experience. Transfusion. 2012 Jun; 52(6): 1228-36.
- Nystrup KB, Stensballe J, Bøttger M, et al. Transfusion therapy in paediatric trauma patients: a review of the literature. Scand J Trauma Resusc Emerg Med. 2015 Feb 15; 23: 21.
- Goobie SM, Haas T. Perioperative bleeding management in pediatric patients. Curr Opin Anaesthesiol. 2016 Jun; 29(3): 352-8
- Nemergut ME, Haile DT, Mauermann WJ, et al. Chapter 20: Blood Conservation and Transfusion Medicine. In: Davis PJ, Cladis FP, eds. Smith’s Anesthesia for Infants and Children. 9th ed. St. Louis, Missouri: Elsevier; 2017: 399–422.
- Neff LP, Cannon JW, Morrison JJ, et al. Clearly defining pediatric massive transfusion: cutting through the fog and friction with combat data. J Trauma Acute Care Surg. 2015 Jan; 78(1): 22-8; discussion 28-9.
- Tyler D. WAKE UP SAFE® The Pediatric Anesthesia Quality Improvement Initiative. 2015. http://wakeupsafe.org/wp-content/uploads/2018/10/Hyperkalemia_ statement.pdf [Accessed 15 February 2019]
- Goobie SM, Gallagher T, Gross I, et al. Society for the advancement of blood management administrative and clinical standards for patient blood management programs. 4th edition (pediatric version). Paeditr Anaesth. 2019 Mar; 29(3): 231- 236. https://onlinelibrary.wiley.com/doi/10.1111/pan.13574
- Valentine SL, Bembea MM, Muszynski JA, et al. Consensus Recommendations for RBC Transfusion Practice in Critically Ill Children from the Pediatric Critical Care Transfusion and Anemia Expertise Initiative. Pediatr Crit Care Med. 2018 Sep; 19(9): 884-898.
- American Society of Anesthesiologist Task Force on Perioperative Blood M. Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologist Task Force on Perioperative Blood Management. Anesthesiology. 2015 Feb; 122: 241-275.
- Dressler A, Finck C, Carroll C, et al. Use of a massive transfusion protocol with hemostatic resuscitation for severe intraoperative bleeding in a child. J Pediatr Surg. 2010 Jul; 45(7): 1530-33.
- Goobie SM, Frank SM. Tranexamic Acid: What is known and unknown, and where do we go from here? Anesthesiology. 2017 Sep; 127(3): 405-407.
- Goobie SM, Zurakowski D, Glotzbecker MP, et al. Tranexamic acid is efficacious at decreasing the rate of blood loss in adolescent scoliosis surgery: a randomized placebo-controlled trial. J Bone Joint Surg Am. 2018 Dec 5; 100(23): 2024- 2032.
- Eckert MJ, Wertin TM, Tyner SD, et al. Tranexamic acid administration to pediatric trauma patients in a combat setting: the pediatric trauma and tranexamic acid study (PED-TRAX). J Trauma Acute Care Surg. 2014 Dec; 77(6): 852-8; discussion 858.
- Goobie SM, Faraoni D. Tranexamic acid and perioperative bleeding in children: What do we still need to know? Curr Opin Anesthesiol. 2019 Jun;32(3):343-352.



