Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Perioperative hyperglycaemia is associated with:
a. Impaired wound healing
b. Increased platelet aggregation
c. Depressed immune function
d. Increased perioperative myocardial infarction
e. Increased surgical site infections - Physiologic effects of insulin include:
a. Fatty acid synthesis
b. Gluconeogenesis
c. Glycogen production
d. Decreased lipolysis in adipose tissue
e. Regulation of potassium homeostasis - Long-acting insulin preparations include:
a. Aspart
b. Lispro
c. Glargine
d. Detemir
e. Regular
Key Points
- Diabetic patients are prone to significant perioperative complications
- Hyperglycaemia is associated with poor wound healing and increased surgical site infections
- Stress is the primary cause of hyperglycaemia, followed by iatrogenic discontinuation of insulin
- Maintaining perioperative glucose <180mg/dL (<10mmol/L) is associated with decreased perioperative mortality
- Diabetic patients should be scheduled as the first patients of the day
INTRODUCTION
Insulin is a hormone produced in the pancreas and secreted in response to increased levels of plasma glucose. The pancreas of Type I diabetic patients no longer manufactures insulin, whereas in Type II diabetics, the pancreas makes insulin but the body no longer (or poorly) responds to it. Insulin has many roles in maintaining physiologic homeostasis, and consequently, diabetic patients present many challenges in the perioperative period. Basal metabolic needs utilize approximately one-half of a patient’s secreted insulin, even in the fasted state. For this reason, diabetic patients presenting for surgery should not have insulin discontinued.
Surgery and anaesthesia result in a neuroendocrine stress response, which releases counter-regulatory hormones. This is summarised in the table below. (Figure 1).

Figure 1: Summary of the main neuroendocrine responses to surgery
Insulin has multiple physiologic effects; some excitatory (stimulation of glucose uptake and lipid synthesis), and others inhibitory (inhibition of lipolysis, proteolysis, glycogenolysis, gluconeogenesis, and ketogenesis).1 Whether the effect is excitatory or inhibitory depends on the target tissue.
One of the most commonly recognized effects of insulin is to decrease the concentration of glucose in the blood.
Hyper and hypo glycaemia are both associated with numerous adverse perioperative events (Figure 2). In 2012, a study published by the NICE-SUGAR study group demonstrated an increase in mortality in critically ill patients with moderate to severe hypoglycaemia, particularly in the setting of distributive shock2.
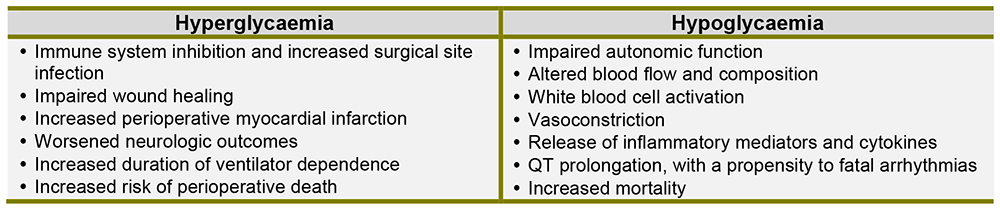
Figure 2: Adverse events associated with hyper- and hypo-glycaemia
TYPES OF INSULIN
There are numerous ways to describe the different types of insulin;
- Function (prandial vs basal)
- Biochemical structure (analog, human, porcine)
- Duration of action (short-acting, long-acting, fixed combination)
Function
Basal insulin is commonly a long-acting preparation, designed to imitate background insulin secretion, keeping blood sugar levels consistent when patients are not eating. It does not adequately cover glucose spikes after meals. Prandial insulin may be a rapid or short-acting preparation, designed to mimic the insulin surge that physiologically occurs with ingestion of a meal. Prandial insulin is taken more often during the day, preceding a meal or in response to point-of-care glucose determinations.
Biochemical Structure
There are two types of insulin structures: recombinant human insulin and analog insulin. Recombinant human insulins are essentially identical in structure to the native insulin produced in the body. Analog insulins are similar in structure but have minor modifications, which allow for rapid onset of action or peakless effect. While analogs cost more, they generally cause less hypoglycaemia and weight gain. Most diabetic patients in the US today use analog insulin.
Porcine and bovine insulins are older, animal-derived, preparations that are seldom used in current practice. Many are out-of-production.
Duration of action
Rapid-acting prandial insulin analogues include lispro, aspart, and glulisine. They are administered just prior to a meal and have an onset of action within 15 to 30 minutes. Their duration of action is 3-6 hours. The peak effect is reached with 15 to 75 minutes. These preparations are very useful for perioperative use in rapid correction of hyperglycaemia.
Short-acting prandial insulin, such as regular insulin, is a human recombinant DNA preparation with an onset of action of 30 to 60 minutes, and a peak effect at 2 to 3 hours. The duration of action is between 3 and 8 hours.
NPH (neutral protamine Hagedorn or Isophane insulin) is an intermediate-acting insulin that does peak in activity, and can therefore cause hypoglycaemia in a fasted patient. Intermediate acting insulins, like NPH or a combination insulin (such as fixed-combination 70/30 preparations), are not to be treated as basal insulins, and therefore necessitate perioperative dose adjustment.
Long-acting basal insulin analogue preparations such as detemir and glargine are dosed 1 to 2 times daily and are considered “peakless,” in that there is no discrete peak of activity (Figure 3). Consequently, they have a low association with hypoglycaemic episodes.
Insulin degludec is an ultra-long acting analogue with a duration of action of at least 42 hours, and offers daily, every other day, or three times weekly dosing. In the BEGIN Basal-Bolus Type 1 Trial, degludec was found to be non-inferior to glargine when used as basal insulin and may have less associated risk of nocturnal hypoglycaemia3. Presently, it is approved for use in Europe but not the US.
Note: The generic names of the various Insulin preparations are used here. It will be valuable to familiarise yourself with local brand names, as patients often only know them by these.
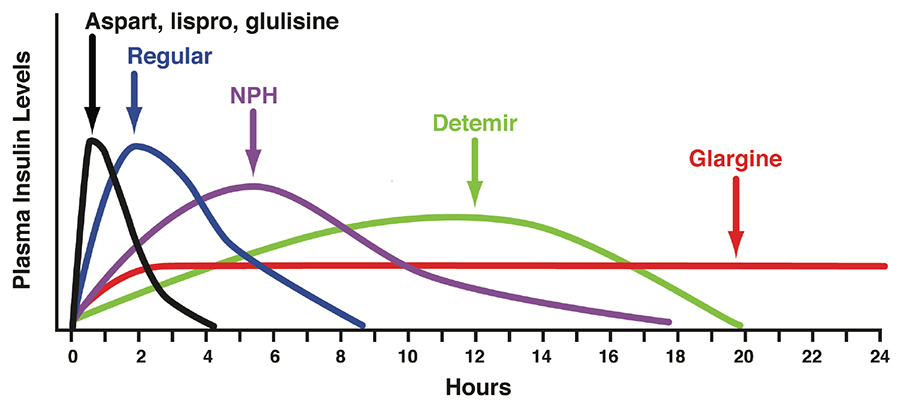
Figure 3: Insulin Profiles Reproduced with permission of Leslie DeGroot, MD, Editor-in-Chief, Endotext.org from: Hirsch, IB, Skyler, JS. The Management of Type 1 Diabetes. www.endotext.org. Version of December 18, 2015, published by MDTEXT.COM, INC, South Dartmouth, MA 02748.
PERIOPERATIVE MANAGEMENT
How is insulin dosed?
Diabetic patients frequently take a long-acting, basal, insulin preparation (such as glargine, detemir or degludec)1,2. These formulations are peakless and maintain a constant level of insulin to prevent ketosis or hyperosmolality. Basal insulin will not cause hypoglycaemia if a meal is skipped4, and should therefore be continued perioperatively at their normal dose. If a patient has a history of morning hypoglycaemia, the dose can be reduced by 20%.
Prandial insulin is usually a rapid-acting analog preparation (such as lispro, aspart or glulisine) which is injected prior to a meal. It reduces blood sugar within 15-25 minutes after injection, remains active for only three or four hours, and closely matching the normal surge of insulin secreted after a meal. (Figure 4)
Insulin pumps are portable devices that provide continuous subcutaneous insulin infusion. It uses an ultra-rapid-acting insulin continuously as a basal insulin and can be bolused prior to meals. A majority of patients can have their pump continued intraoperatively, but this decision is practitioner-dependent. Recommendations are to restrict pump use to surgical procedures less than 2 hours4. For longer procedures, the pump should be discontinued and replaced with an intravenous insulin infusion.
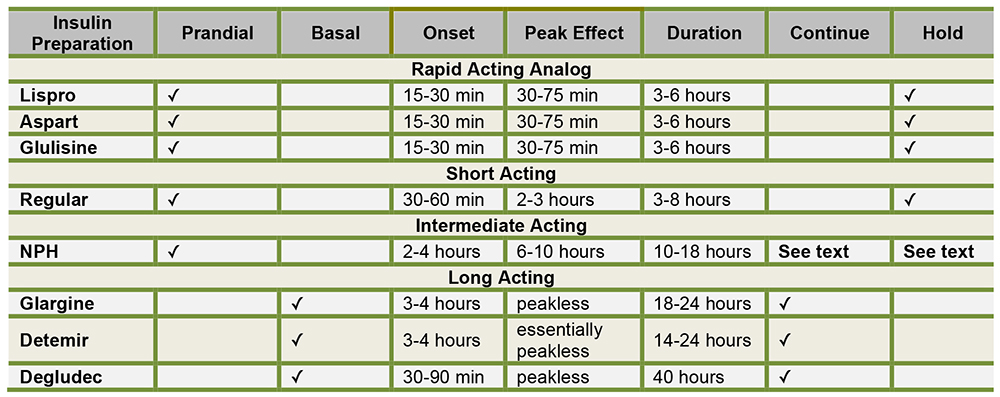
Figure 4: Table showing insulin characteristics & preoperative recommendations
Why should insulin be continued when the patient is fasted?
Patients require a constant source of insulin (whether or not they are diabetic), and discontinuing or decreasing insulin for fear of hypoglycaemia is not appropriate.
While insulin inhibits both gluconeogenesis and conversion of glycogen to glucose, it also inhibits degradation of fats to fatty acids and of fatty acids to ketones. Inadequate insulin dosing can lead to uncontrolled hyperglycaemia and even ketoacidosis, which is why long-acting insulin must be continued, even in a fasting state5.
Perioperative Glucose Target
The Normoglycaemia in Intensive Care Evaluation—Survival using Glucose Algorithm Regulation (NICE-SUGAR) Study, is a large, international, randomized trial published in 2009. The study concluded that a blood glucose target of <180 mg/dL (<10mmol/L) resulted in lower mortality than a more restrictive target of 81-108 mg/dL(4.5-6mmol/L)6. Although a specific glucose target remains unclear, most published guidelines advocate for glucose levels between 140-180mg/dL
(7.8-10mmol/L).
Preoperative Insulin Dosing and Dosing Adjustments
Type I diabetic patients using long-acting insulins, such as glargine, should continue these as normal, when fasting. If the patient is prone to morning hypoglycaemia, the dose can be reduced by 20%. For reasons stated above, this dose should not be discontinued. Rapid acting insulins, such as aspart, should be held the morning of surgery. If their morning glucose is greater than 200mg/dL (11.1mmol/L), they may adjust the dose based on their prescribed sliding scale.
Type II diabetic patients can continue their basal insulin and maintain their insulin pump. Rapid-acting insulins should be held the morning of surgery. Patients who take intermediate-acting insulins, such as NPH, should take their usual dose on the evening prior to surgery. If they will be skipping their evening meal, the dose should be reduced by 25%. On the morning of surgery, they should take half of their scheduled dose5.
Patients taking premixed insulins or fixed-combination insulins are more of a challenge. Premixed insulins (NPH 70/30, 75/25, 50/50) are a combination of intermediate-acting insulin and either fast or short-acting insulin (effectively a basal and prandial combination). Their perioperative use is not ideal. There are three options in this situation5:
Option 1. Discontinue the premixed insulin and switch to a regimen that includes long-acting insulin. This can be done by substituting a long-acting analog, such as glargine, in place of the night time dose of 70/30. A quick formula for conversion is to add all the premixed insulin doses and give half as basal insulin the night prior to surgery.
Option 2. Adjust the dose of premixed insulin. It may not be feasible or economical to change the patient’s premixed insulin just before surgery. In these situations, the patient can take ½ – ¾ of the morning dose, followed by administration of a dextrose-containing intravenous fluid and frequent blood glucose checks.
Option 3. Simply skip the morning dose of premixed insulin entirely and have the patient present to the preoperative area early and have insulin dosed there based upon point-of-care values.
Correction doses of insulin to treat hyperglycaemia can be given subcutaneously; with a rapid-acting formulation such as lispro or aspart. There are several “off-the-cuff” formulas for determining the amount of correction insulin to administer, such as 1-4 units of rapid-acting insulin for every 50 mg/dL (2.8mmol/L) of intended glucose reduction, or the “Rule of 1800” (Figure 5)7,8. Care must be taken when administering multiple doses of subcutaneous insulin to avoid hastily stacking them and causing a subsequent hypoglycaemia. Intravenous regular insulin can also be used, but it peaks within minutes, has a duration of action of 30-40 minutes, which may result in rapid and wide swings in blood glucose4. Asking the patient how much correction insulin they take for a given glucose level is a simple and effective means of dose determination.

Figure 5: The “1800” Rule
Intraoperative management
Intraoperative glucose control will be most effective with an intravenous insulin infusion. Subcutaneously administered insulin is poorly absorbed in the perioperative setting of hypothermia or peripheral vasoconstriction. A study published in 2009 found that intraoperative insulin infusion was associated with a reduction in perioperative myocardial infarction in patients undergoing vascular surgery9. Check with your institution regarding protocols that may already be in place.
Postoperative management
Glucose levels should be checked on arrival to the Post-Anaesthesia Recovery Unit (PARU), and hyperglycaemia treated with rapid or short-acting insulin. Correction with insulin in the PARU should be administered intravenously. Many of the physiologic effects of surgery and anaesthesia, such as peripheral vasoconstriction or hypothermia, may make subcutaneously administered insulin absorption unpredictable. Patients who have undergone major surgical procedures, such as CABG or large abdominal procedures, may benefit from having intraoperative insulin infusions continued in the postoperative phase. Infusion rates can be adjusted and correction insulin administered based on frequent glucose determinations. Patients who will be without oral intake should be managed with insulin infusions and may also necessitate simultaneous dextrose infusion. Once it becomes apparent that oral intake will be tolerated, the patient can be transitioned from the insulin infusion to subcutaneous insulin. Since intravenous regular insulin has a short half-life, the first dose of subcutaneous insulin should be given prior to discontinuing the infusion. Subcutaneous short- or rapid-acting insulin should be given 1-2 hours before discontinuing an infusion11. When intermediate- or longacting insulin is to be administered, it should be given 2-3 hours prior to infusion discontinuation.
Patients who have undergone minor, or outpatient, procedures can resume their usual insulin use once normal eating and drinking is tolerated. They should be made aware that some medications administered intraoperatively (such as dexamethasone) and postoperative pain may result in higher than usual glucose readings. For patients who were administered insulin intraoperatively, or in PARU, the risk of hypoglycaemia subsides within 1.5 hours after receiving subcutaneous rapid-acting insulin and within 3-4 hours for subcutaneous short-acting insulin (regular insulin). In these patients, a longer period of observation in PARU may be beneficial prior to discharge12.
SUMMARY
- Perioperative insulin management can be challenging, but with the introduction of peakless insulin analogs, it is becoming less so.
- Diabetic patients should be scheduled as the first patient of the day, or as early as possible to minimize disruption of normal insulin dosing schedules.
- Effective perioperative insulin management results in improved surgical outcomes.
ANSWERS TO QUESTIONS
- ..
a. True.
b. True.
c. True.
d. True.
e. True. Perioperative hyperglycaemia is associated with all of these, as well as worsened neurologic outcomes and an increased risk of death10. - ..
a. True. Insulin plays a pivotal role in fatty acid synthesis.
b. False. Gluconeogenesis is the production of glucose from stored sources and results in an increase in blood glucose levels. Insulin is associated with mediating glucose storage as glycogen and a decrease in blood glucose levels.
c. True. Insulin is a potent stimulator of glycogen production in the liver.
d. True. Insulin decreases lipolysis in adipose tissue and reduces ketosis.
e. True. Insulin regulates the movement of potassium from the extracellular to intracellular environment via the sodium potassium ATPase. - ..
a. False. Aspart is a rapid-acting analog, with an onset of 5-15 mins and duration of 3-5 hrs.
b. False. Lispro is a rapid-acting analog with an onset of 5-15 mins and duration of 3-5 hrs.
c. True. Glargine is a long-acting analog with an onset of 2-4 hrs and duration of 20-24 hrs.
d. True. Detemir is a long-acting analog with an onset of 2-4 hrs and duration of 16-20 hrs.
e. False. Regular is a short-acting human insulin with an onset of 30-60 mins and duration of 5-8 hrs.
REFERENCES
- Sonksen P, Sonksen J. Insulin: understanding its action in health and disease. British Journal of Anaesthesia. 2000;85(1):69-79.
- Hypoglycaemia and risk of death in critically ill patients. New England Journal of Medicine 2012;367(12):1108-18
- Heller S, Buse J, Fisher M, Garg S, Marre M, Merker L, et al. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 1 diabetes (BEGIN Basal-Bolus Type 1): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet (London, England). 2012;379(9825):1489-97.
- Vann MA. Perioperative management of ambulatory surgical patients with diabetes mellitus. Current opinion in anaesthesiology. 2009;22(6):718-24.
- Dobri G, Lansang M. How should we manage insulin therapy before surgery? CCJM. 2013;80(11):702-4.
- Intensive versus Conventional Glucose Control in Critically Ill Patients. New England Journal of Medicine. 2009;360(13):1283-97.
- Inzucchi SE. Clinical practice. Management of hyperglycemia in the hospital setting. The New England journal of medicine. 2006;355(18):1903-11.
- Rhodes ET, Ferrari LR, Wolfsdorf JI. Perioperative management of pediatric surgical patients with diabetes mellitus. Anesthesia and analgesia. 2005;101(4):986-99, table of contents.
- Subramaniam B, Panzica PJ, Novack V, Mahmood F, Matyal R, Mitchell JD, et al. Continuous perioperative insulin infusion decreases major cardiovascular events in patients undergoing vascular surgery: a prospective, randomized trial. Anesthesiology. 2009;110(5):970-7.
- Kotagal M, Symons RG, Hirsch IB, Umpierrez GE, Dellinger EP, Farrokhi ET, et al. Perioperative hyperglycemia and risk of adverse events among patients with and without diabetes. Annals of surgery. 2015;261(1):97-103.
- Khan, NA, Ghali, WA, Cagliero, E. Perioperative management of blood glucose in adults with diabetes mellitus. In: UpToDate. Post, TW (Ed), UpToDate, Waltham, MA, 2016.
- Joshi GP, Chung F, Vann MA, Ahmad S, Gan TJ, Goulson DT, et al. Society for Ambulatory Anesthesia consensus statement on perioperative blood glucose management in diabetic patients undergoing ambulatory surgery. Anesthesia and analgesia. 2010;111(6):1378-87.



